Bio-Rad CFX Opus 96 Real-Time PCR Detection System
| Brand | Bio-Rad |
|---|---|
| Origin | Singapore |
| Model | CFX Opus 96 |
| Instrument Type | Real-Time Fluorescent Quantitative PCR System |
| Sample Throughput | 96-well format |
| Heating Rate | 5°C/sec |
| Temperature Accuracy | ±0.2°C |
| Well-to-Well Temperature Uniformity | ±0.3°C |
| Optical Configuration | Individual well excitation and detection with FRET-capable channels |
Overview
The Bio-Rad CFX Opus 96 Real-Time PCR Detection System is an engineered platform for high-fidelity nucleic acid quantification, designed to meet the rigorous demands of academic research, pharmaceutical development, and diagnostic assay validation laboratories. It operates on the principle of real-time fluorescence monitoring during thermal cycling—detecting accumulation of amplicon-specific signals (e.g., SYBR Green, hydrolysis probes, or FRET-based assays) with cycle-by-cycle resolution. The system integrates a thermally optimized reaction block with a precision optical engine that eliminates cross-well signal bleed-through and path-length variability—critical for reproducible ΔΔCq calculations and low-abundance target detection. Its architecture complies with fundamental requirements for GLP-aligned workflows, including audit-trail–capable software logging, user-access controls, and data integrity safeguards aligned with ALCOA+ principles.
Key Features
- Patented lightweight thermal block with dynamic gradient capability—enabling rapid empirical optimization of annealing temperatures across 12 distinct zones in a single run.
- Individual-well optical excitation and emission detection—each of the 96 wells is sequentially illuminated and measured using dedicated LED/photodiode pairs, eliminating inter-well crosstalk and obviating the need for spatial fluorescence normalization.
- FRET-compatible optical channels—supporting dual-labeled probe chemistries such as molecular beacons and adjacent hybridization probes without hardware modification.
- High-fidelity temperature control: ±0.2°C accuracy and ±0.3°C well-to-well uniformity across the full 96-well plate—minimizing edge effects and ensuring inter-run comparability.
- 5°C/sec heating rate—reducing total run time without compromising amplification efficiency or specificity, particularly beneficial for fast-cycling protocols.
- Adjustable 12–55° tilt-angle touchscreen interface—optimized for glove-compatible operation (single- or double-layer nitrile or latex) and ergonomic benchtop use.
Sample Compatibility & Compliance
The CFX Opus 96 accommodates standard 0.2 mL skirted or semi-skirted 96-well PCR plates and compatible optical seals. It supports all major qPCR chemistries—including intercalating dyes (SYBR Green I), hydrolysis probes (TaqMan), hybridization probes, and scorpions—without recalibration. The system’s firmware and CFX Maestro Software v5.2+ are designed to support 21 CFR Part 11–compliant configurations when deployed with appropriate IT infrastructure (e.g., electronic signatures, role-based access, and immutable audit trails). While not intended for clinical diagnostics, its performance characteristics align with ISO/IEC 17025 method validation expectations for research-grade nucleic acid quantification.
Software & Data Management
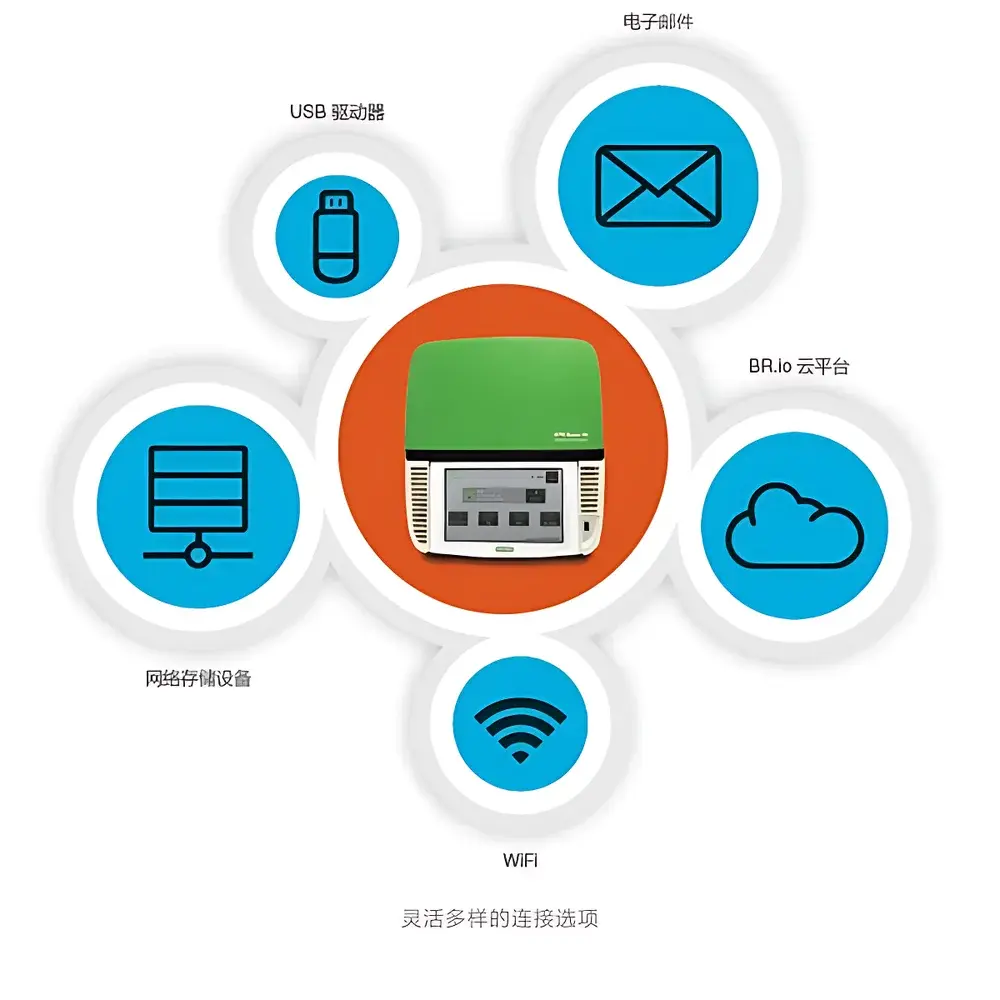
CFX Maestro Software provides a validated, modular environment for experimental design, acquisition, analysis, and reporting. It enables automated statistical evaluation—including unpaired t-tests, one-way ANOVA with post-hoc Tukey correction, and reference gene stability ranking (geNorm and NormFinder algorithms). Data visualization tools support annotation of p-values, custom axis labeling, vector-based export (SVG, TIFF, PNG), and direct integration with publication-ready figure assembly. The software natively imports PrimePCR Assay metadata—validating primer specificity, amplicon size, and efficiency curves against Bio-Rad’s curated database. Data persistence options include local storage (USB 3.0), network-attached storage (SMB/CIFS), and encrypted cloud synchronization via BR.io—a zero-client web platform requiring no local software installation. All instrument logs, user actions, and result exports are timestamped and cryptographically hashed to support traceability.
Applications
The CFX Opus 96 is routinely deployed in gene expression profiling (including low-input RNA samples), miRNA quantification, SNP genotyping, viral load determination, and host-pathogen interaction studies. Its thermal precision and optical fidelity make it suitable for validating CRISPR editing efficiency, measuring splice variant ratios, and supporting QC in oligonucleotide synthesis workflows. In bioprocess development, it serves as a release assay tool for plasmid DNA purity assessment and residual host-cell DNA quantification per ICH Q5A guidelines. The system also supports multiplexed detection in food safety testing (e.g., pathogen screening) and environmental microbiology (e.g., 16S rRNA gene copy number estimation).
FAQ
Is the CFX Opus 96 compliant with FDA 21 CFR Part 11?
Yes—when configured with CFX Maestro Software in secure mode, enabled audit trail logging, electronic signatures, and appropriate network authentication, it satisfies core Part 11 requirements for electronic records and signatures.
Can the system operate without a connected PC?
Yes—it features an embedded Linux OS and fully functional touchscreen interface; all protocol setup, run initiation, and basic analysis can be performed standalone.
Does the optical system require daily calibration or background subtraction?
No—its fixed-optics design with per-well excitation/detection eliminates drift-related recalibration needs; background fluorescence is subtracted algorithmically during baseline determination.
What file formats does CFX Maestro support for data export?
CSV (for statistical import), RDML (for MIQE-compliant exchange), SVG/TIFF/PNG (publication graphics), and PDF (annotated reports with embedded metadata).
Is BR.io cloud storage encrypted and GDPR-compliant?
Data in transit and at rest is AES-256 encrypted; Bio-Rad maintains ISO/IEC 27001 certification and offers Data Processing Agreements (DPAs) for EU-based institutions.