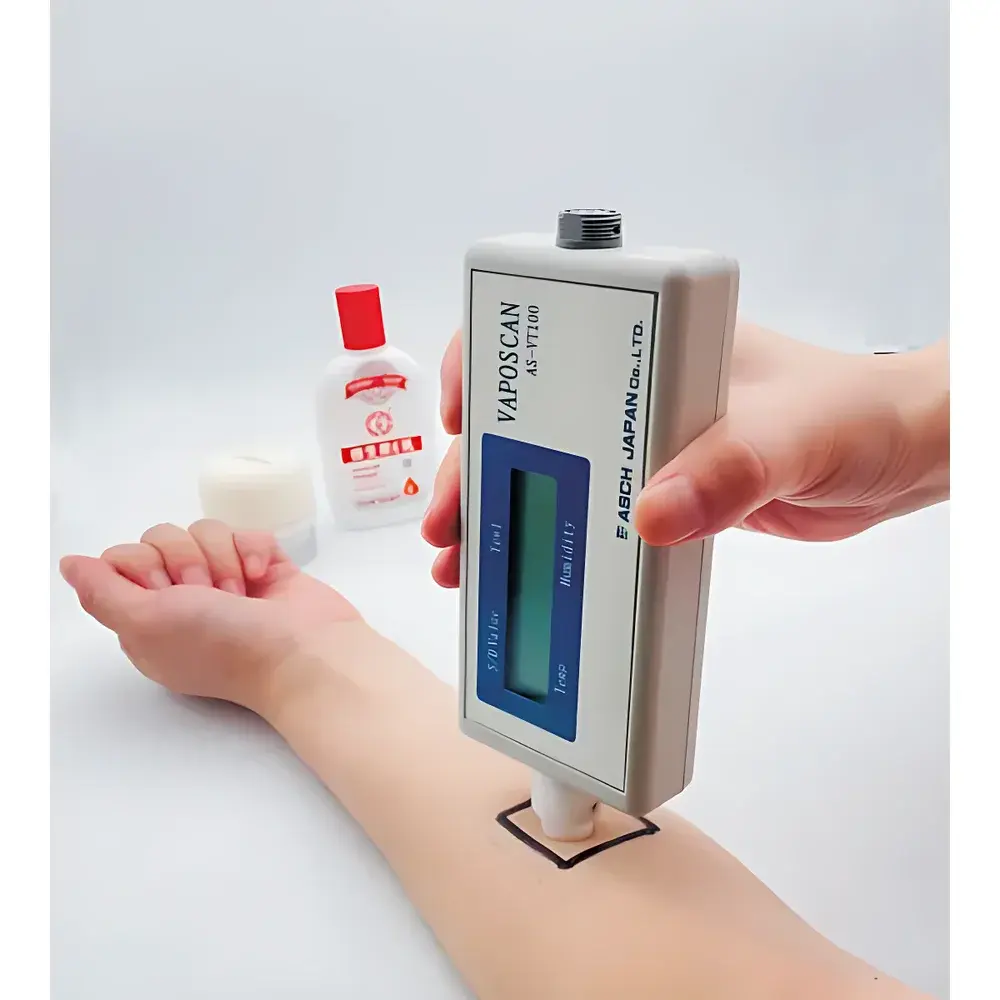
ASCH VAPOSCAN AS-VT100RS Transepidermal Water Loss (TEWL) Meter
| Brand | ASCH |
|---|---|
| Origin | Japan |
| Model | AS-VT100RS |
| Measurement Principle | Closed-chamber dynamic humidity gradient analysis |
| TEWL Range | 0–300 g/m²/h |
| Resolution | 0.1 g/m²/h |
| Measurement Time | 5–14 s |
| Probe Aperture Options | 6, 8, 10, or 12 mm (DIP-switch configurable) |
| Dimensions | 165 × 70 × 28 mm |
| Weight | 200 g |
| Power Supply | AC adapter or AA batteries |
| Environmental Monitoring | Integrated temperature & relative humidity sensors |
| Data Output | RS-232 serial interface |
| Sample Types | Human skin (in vivo), excised animal skin (rat, pig, rabbit), 3D reconstructed human epidermis (RHE), and cultured skin models |
Overview
The ASCH VAPOSCAN AS-VT100RS is a precision-engineered transepidermal water loss (TEWL) meter designed for quantitative, non-invasive assessment of stratum corneum barrier integrity. It operates on the closed-chamber principle—measuring the rate of water vapor flux across the skin surface by detecting real-time humidity gradients within a thermally stabilized, sealed measurement cavity. This method complies with internationally recognized physical principles defined in ISO 13732-3 and ASTM E1965-22 for cutaneous evaporimetry. Unlike open-chamber systems, the VAPOSCAN’s sealed architecture eliminates interference from ambient air turbulence, drafts, or uncontrolled humidity fluctuations—ensuring high reproducibility under both clinical and preclinical laboratory conditions. Its microprocessor-controlled environmental compensation engine continuously monitors internal chamber temperature and relative humidity, enabling automatic validation of measurement readiness and termination criteria per ISO 24444:2019 guidelines for instrumental skin testing.
Key Features
- High-speed acquisition: Delivers stable TEWL readings within 5–14 seconds—optimized for throughput in high-volume IVPT/IVRT studies and longitudinal dermatological monitoring.
- Modular probe system: Interchangeable apertures (6 mm, 8 mm, 10 mm, and 12 mm) configured via onboard DIP switches—enabling anatomically appropriate measurements on facial skin, scalp, lips, volar forearm, or ex vivo diffusion cell membranes.
- Dual-power architecture: Seamless transition between AC adapter operation and four AA alkaline batteries—supporting uninterrupted field use in controlled environment rooms, animal facilities, and Good Laboratory Practice (GLP)-compliant labs.
- Integrated environmental sensing: Onboard temperature and RH sensors provide concurrent dermal microclimate data—critical for normalization and statistical correction per FDA Guidance for Industry (October 2022) on topical drug product permeation testing.
- RS-232 digital output: Enables direct integration with DAQVAPOSCAN software for time-stamped data logging, automated report generation, and audit-trail-compliant storage aligned with 21 CFR Part 11 requirements.
Sample Compatibility & Compliance
The VAPOSCAN AS-VT100RS supports validated measurement across diverse biological substrates: human volunteers (in vivo), excised porcine, murine, and rabbit skin (ex vivo), as well as 3D human epidermal equivalents (e.g., EpiDerm™, SkinEthic™). Its performance aligns with regulatory expectations outlined in multiple pharmacopeial and agency documents—including USP Semisolid Drug Products—Performance Tests, EMA Guideline on Quality of Transdermal Patches (2014), PMDA’s BE guidance for topical generics (2006), and CDE’s July 2024 draft technical guidance on IVRT/IVPT studies. For barrier integrity qualification prior to IVPT, the instrument meets the FDA-recommended acceptance threshold of ≤15 g/m²/h for intact human trunk/thigh skin—verified using standardized calibration protocols traceable to NIST-maintained hygrometric references.
Software & Data Management
DAQVAPOSCAN software provides native support for real-time visualization, batch export (CSV, Excel-compatible), and metadata tagging (operator ID, subject ID, anatomical site, probe size, ambient lab conditions). All measurement events are timestamped and logged with full environmental parameter history—facilitating retrospective GLP/GMP audit readiness. The software enforces user-level access control, electronic signature capture, and immutable record retention—fully compliant with FDA 21 CFR Part 11 Subpart B requirements for electronic records and signatures. Exported datasets include raw humidity delta curves, calculated TEWL slopes, SD of baseline stability, and pass/fail flags against configurable acceptance criteria.
Applications
- Pharmaceutical development: Barrier integrity screening before and after IVPT experiments; formulation comparison of topical corticosteroids, retinoids, and transdermal patches.
- Cosmetic science: Objective evaluation of moisturizer efficacy, occlusive agent performance, and irritancy potential of surfactants or preservatives per ISO 11948-1.
- Dermatotoxicology: Longitudinal monitoring of TEWL elevation in murine atopic dermatitis, psoriasis-like, and UVB-induced photoaging models.
- Regulatory submission support: Generation of TEWL datasets meeting CDE, FDA, EMA, and PMDA documentation standards for ANDA, NDA, and MAA dossiers.
- In vitro skin model qualification: Routine assessment of barrier maturity in reconstructed epidermis used for OECD TG 439 and 442D assays.
FAQ
What is the recommended calibration frequency for the VAPOSCAN AS-VT100RS?
Calibration verification should be performed daily using a certified reference evaporation source (e.g., porous ceramic disc with known vapor flux), with full recalibration conducted quarterly or after any mechanical impact or firmware update.
Can the device measure TEWL on hairy or uneven skin surfaces?
Yes—when paired with the optional 6 mm probe and gentle manual stabilization, it achieves reliable readings on scalp, eyebrows, and flexural regions; however, dense terminal hair should be trimmed to avoid cavity seal disruption.
Does the instrument meet ISO 24444:2019 requirements for TEWL instrumentation?
Yes—the closed-chamber design, real-time environmental compensation, and documented measurement uncertainty (<±3.2% at 10 g/m²/h) satisfy all critical clauses of ISO 24444:2019 Annex A.
Is RS-232 the only data interface available?
Yes—no USB or Bluetooth interface is provided; RS-232 ensures deterministic timing and electromagnetic compatibility in regulated laboratory environments.
How does the VAPOSCAN handle temperature drift during extended measurement sessions?
The embedded thermal sensor triggers automatic hold-and-wait logic if chamber temperature deviates >±0.3°C from setpoint—preventing erroneous slope calculation due to thermal lag artifacts.