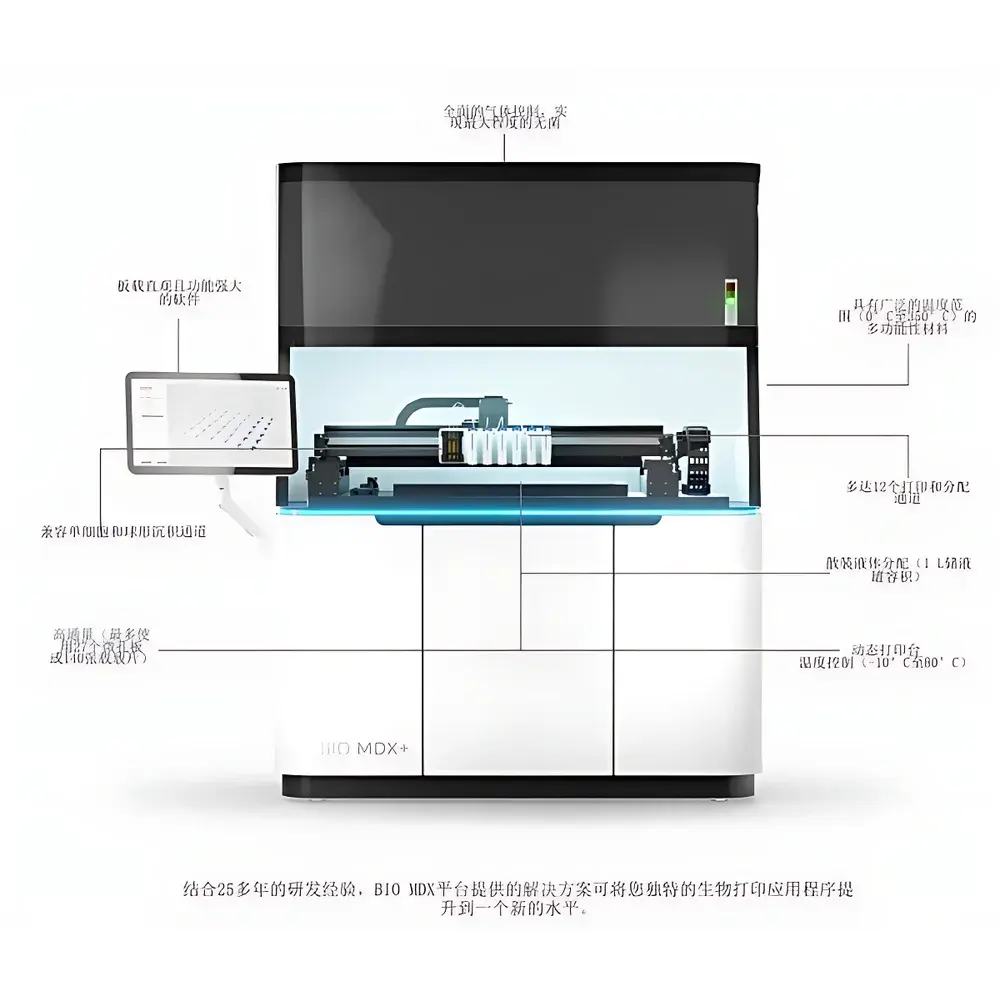
Cellink BIO MDX Biological 3D Bioprinter
| Brand | CELLINK |
|---|---|
| Origin | Sweden |
| Model | BIO MDX |
| Maximum Microplate Capacity | 27 standard microplates or 140 microscope slides |
| Droplet Volume Resolution (with sciDROP PICO) | <200 pL |
| Nozzle Temperature Range | 0–350 °C |
| Build Platform Temperature Range | −10–80 °C |
| Ambient Control | HEPA-filtered laminar airflow, programmable humidity, temperature, and dew point |
| Positioning Accuracy | ≤1 µm |
Overview
The Cellink BIO MDX is an industrial-grade biological 3D bioprinter engineered for reproducible, scalable, and GxP-aligned biofabrication. It operates on a precision motion-controlled Cartesian architecture, integrating thermal regulation, environmental containment, and multi-modal dispensing technologies to support both extrusion-based and drop-on-demand bioprinting modalities. Designed for translational research labs, contract development and manufacturing organizations (CDMOs), and academic core facilities, the BIO MDX enables standardized production of 3D tissue constructs—including spheroids, organoids, and scaffold-free co-cultures—under controlled aseptic conditions. Its closed-system design complies with ISO 14644-1 Class 5 cleanroom performance standards when operated with integrated HEPA filtration and real-time environmental monitoring.
Key Features
- High-throughput platform supporting up to 27 standard microplates or 140 glass slides per print run—enabling parallelized assay development and large-scale matrix screening.
- Sub-micron positioning accuracy (≤1 µm) achieved via high-resolution stepper motors and optical encoder feedback, ensuring geometric fidelity across printed biomaterial layers.
- Dual-temperature control architecture: independent regulation of print head (0–350 °C) and build platform (−10–80 °C) to accommodate thermosensitive bioinks, melt-electrospun polymers, and crosslinking-sensitive hydrogels.
- sciDROP PICO integration for picoliter-scale liquid dispensing (<200 pL resolution), enabling precise spatial patterning of growth factors, antibodies, or small-molecule therapeutics within 3D constructs.
- Patented laminar-flow enclosure with HEPA filtration, dynamic humidity control (10–95% RH), temperature stability (±0.5 °C), and dew point management—minimizing evaporation and microbial contamination during extended print sessions.
- Onboard nozzle imaging system with real-time visual feedback and auto-alignment calibration—critical for spheroid placement accuracy and multi-material registration.
Sample Compatibility & Compliance
The BIO MDX accommodates a broad spectrum of biomaterials, including natural hydrogels (e.g., collagen, fibrin, alginate), synthetic polymers (e.g., Pluronic F127, PCL, PLA), decellularized extracellular matrix (dECM) bioinks, and cell-laden suspensions at viscosities ranging from 10 to 10⁶ mPa·s. Its modular tooling interface supports interchangeable print heads for extrusion, inkjet, and microvalve-based deposition. From a regulatory perspective, the system’s audit-trail-capable software, user-access controls, and electronic signature functionality align with FDA 21 CFR Part 11 requirements. Routine operation adheres to ISO/IEC 17025 principles for measurement traceability, and environmental logs satisfy GLP documentation standards for preclinical tissue model generation.
Software & Data Management
BIO MDX is operated via CELLINK’s BIOCAD™ software suite—a validated, version-controlled platform supporting CAD import (STL, 3MF), slice-based path planning, parameter scripting, and batch protocol execution. All print jobs generate immutable metadata logs including ambient conditions, thermal profiles, motor step counts, and imaging timestamps. Data export conforms to FAIR principles (Findable, Accessible, Interoperable, Reusable), with native support for HDF5 and CSV formats. Integration with LIMS and ELN systems is enabled through RESTful API endpoints and configurable ODBC connectors—facilitating automated data ingestion into enterprise quality management systems (QMS).
Applications
- High-content 3D cell culture model development for oncology, neurobiology, and immunology research.
- Automated fabrication of patient-derived tumor organoids for drug sensitivity profiling and biomarker discovery.
- Multi-material bioprinting of vascularized tissue constructs with embedded endothelial channels.
- Spheroid array generation for high-throughput toxicity screening and intercellular communication studies.
- Process validation and tech transfer in biomanufacturing environments requiring repeatable, documented biofabrication workflows.
FAQ
Does the BIO MDX support sterile, closed-loop operation for GMP-compliant processes?
Yes—the system’s HEPA-filtered enclosure, glove-port access design, and autoclavable print modules enable integration into ISO Class 5 cleanrooms; full validation packages are available upon request.
Can I use third-party bioinks with the BIO MDX?
Yes—the open-architecture fluidics system accepts syringes and cartridges from multiple vendors; viscosity and crosslinking compatibility must be verified per material safety data sheet (MSDS) and rheological characterization.
Is remote monitoring and troubleshooting supported?
Yes—BIOCAD™ includes secure cloud-enabled diagnostics, live camera feed streaming, and role-based remote desktop access compliant with HIPAA and GDPR data handling policies.
What level of technical support and service coverage is provided?
CELLINK offers global field service engineering, annual preventive maintenance contracts, and application specialist consultation—aligned with ISO 9001-certified service delivery protocols.