Azenta FreezerPro® Biological Sample Management Software
| Brand | Azenta |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | FreezerPro® |
| Pricing | Upon Request |
Overview
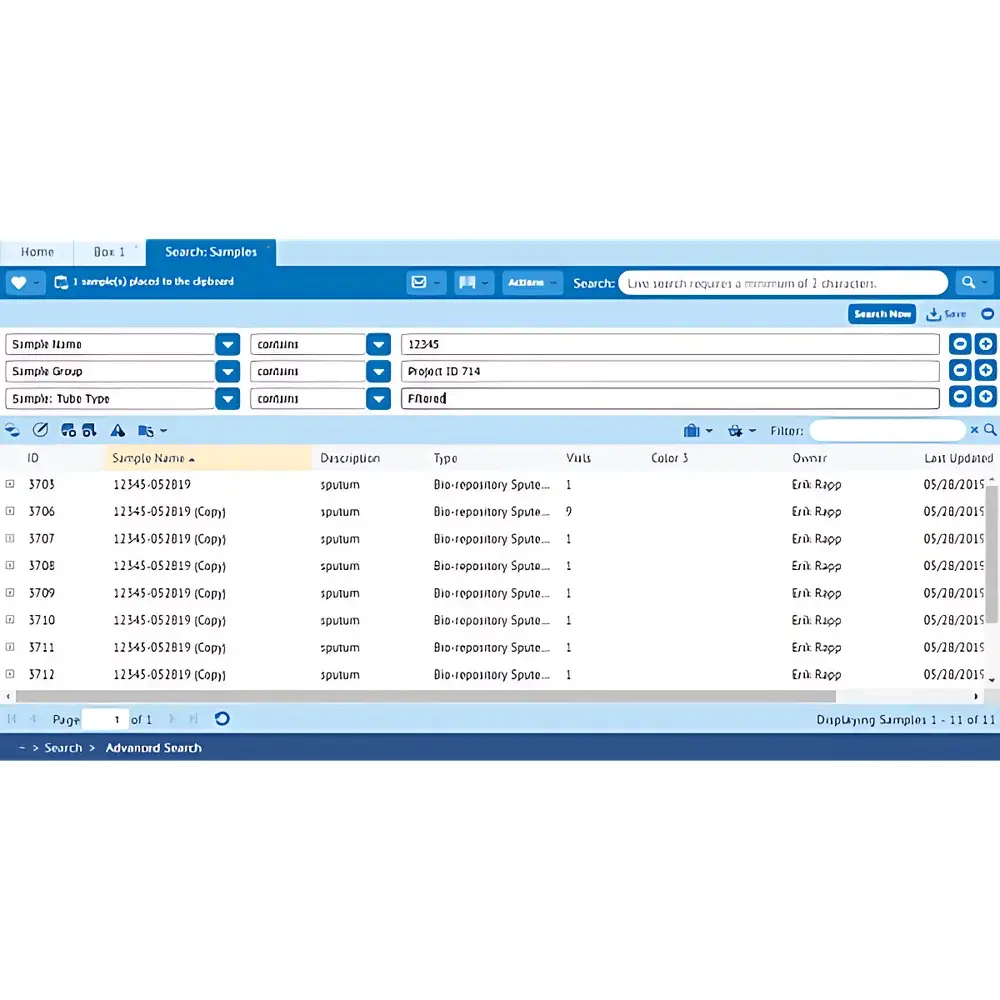
FreezerPro® by Azenta is a validated, web-based Laboratory Information Management System (LIMS) purpose-built for biorepository operations, clinical biobanks, translational research cores, and pharmaceutical sample stewardship. Engineered for precision and regulatory rigor, it implements a metadata-driven architecture that decouples sample data models from underlying database schema—enabling dynamic configuration without code modification. The system operates on a client-server model with HTTPS-secured browser access, supporting concurrent multi-site usage across geographically distributed repositories. Its core functionality centers on lifecycle traceability of biospecimens—from accessioning and cryopreservation (vial, rack, box, freezer hierarchy) through aliquoting, distribution, and disposition—while enforcing audit-ready data integrity at every transactional layer.
Key Features
- Zero-Code Configuration: SOP-aligned workflow setup via intuitive drag-and-drop interface; no scripting or developer involvement required for field definitions, status transitions, or permission rules.
- Unbounded Scalability: Hierarchical storage modeling supports unlimited freezer racks, boxes, positions, and specimen records—validated in production environments exceeding 50 million vials.
- Intelligent Alerting Engine: Configurable real-time notifications for critical thresholds including volume depletion, freeze-thaw cycle count, expiration date proximity, and container occupancy limits.
- Derivative Sample Tracking: Native support for parent-child relationships, lineage mapping, and derived assay metadata (e.g., DNA extraction batches, RNA sequencing runs, histopathology annotations).
- Visual Inventory Navigation: Interactive 2D/3D freezer map rendering with color-coded status overlays, barcode-scanned position validation, and drag-to-move vial relocations.
- Multi-Language & Accessibility Compliance: UI localization for English, Spanish, French, German, Japanese, and Simplified Chinese; fully compliant with WCAG 2.1 AA and U.S. Section 508 standards.
Sample Compatibility & Compliance
FreezerPro® accommodates heterogeneous biospecimen types—including whole blood, PBMCs, FFPE blocks, tissue sections, plasma, serum, saliva, and microbial isolates—with customizable attribute schemas per collection protocol. It natively supports common biobanking standards: ISBER Best Practices (2023), NCI Biorepository Guidelines, and caBIG®-aligned data elements. Regulatory compliance is embedded—not bolted on: full implementation of FDA 21 CFR Part 11 (electronic signatures, audit trails, role-based e-signature workflows), cGMP/cGLP-aligned electronic record retention policies, HIPAA-compliant PHI handling with field-level encryption, and ISO 9001:2015 quality management system documentation templates.
Software & Data Management
The platform provides bidirectional data exchange via RESTful APIs and scheduled batch import/export using CSV, TSV, and HTML formats. All reports are generated from a unified metadata ontology, enabling consistent cross-collection analysis. Custom report templates can be authored using built-in WYSIWYG editor or exported as PDF, Excel, or XML. Audit logs capture immutable timestamps, user IDs, IP addresses, and before/after field values for every create/update/delete action—retained for configurable durations meeting institutional retention policies. Integration modules exist for Thermo Fisher SampleManager LIMS, LabVantage, and major ELN systems via HL7 FHIR and ASTM E1578 interfaces.
Applications
- Academic and government biorepositories managing longitudinal cohort studies (e.g., All of Us, UK Biobank)
- Pharma-sponsored clinical trial biobanks requiring chain-of-custody documentation for IND submissions
- Hospital-based pathology archives digitizing legacy FFPE inventory with slide-level annotation
- Contract research organizations (CROs) operating multi-client sample hosting facilities under ISO/IEC 17025 scope
- Regenerative medicine labs tracking MSC expansion batches with donor eligibility flags and release testing integration
FAQ
Does FreezerPro® support 21 CFR Part 11-compliant electronic signatures?
Yes—signature workflows include dual authorization, biometric challenge questions, and timestamped certificate binding aligned with FDA guidance.
Can existing freezer inventories be migrated into FreezerPro®?
Yes—structured migration services include legacy data cleansing, ontology mapping, and post-load validation reports.
Is on-premise deployment available, or is it cloud-only?
Both options are supported: AWS-hosted SaaS instances (HIPAA-eligible) and validated on-premise deployments with annual infrastructure certification.
How frequently are regulatory updates incorporated (e.g., new ISBER editions, FDA guidance revisions)?
Compliance patches are released quarterly as part of the standard maintenance subscription, with change logs published in the Customer Portal.
What training and validation support is provided for GxP environments?
Azenta offers IQ/OQ/PQ protocol templates, UAT facilitation, and third-party validation consulting accredited to ISO/IEC 17020.