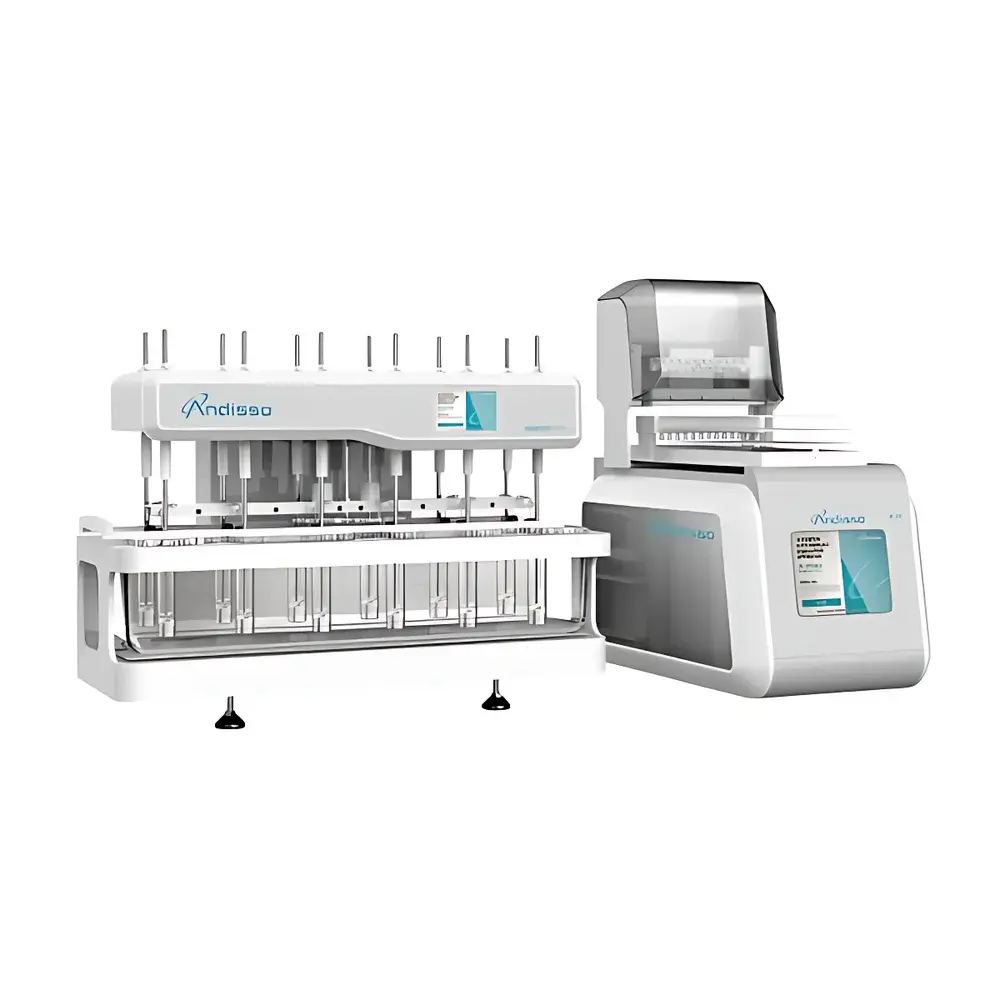
ANDISSO D1200 PRO Immersion Cell Dissolution Auto-Sampling System
| Brand | ANDISSO |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Regional Classification | Domestic (China) |
| Model | D1200 PRO Immersion Cell |
| Instrument Type | Online Dissolution Sampling System |
| Dissolution Vessels | 12-position |
| Temperature Range | Ambient to 50 °C |
| Temperature Accuracy | ±0.2 °C |
| Water Bath Volume | 46 L |
Overview
The ANDISSO D1200 PRO Immersion Cell Dissolution Auto-Sampling System is an online, 12-vessel dissolution testing platform engineered for precise and reproducible release profiling of semi-solid pharmaceutical dosage forms—including topical gels, creams, ointments, and transdermal patches—using the immersion cell method (USP ). Unlike conventional paddle or basket apparatuses optimized for oral solid dosage forms, this system implements a vertically oriented, temperature-controlled immersion cell assembly submerged directly into each dissolution medium. The principle relies on passive diffusion across a defined membrane interface under sink conditions, enabling quantitative assessment of drug release kinetics from viscous or non-homogeneous matrices where traditional hydrodynamic methods yield poor discrimination or mechanical interference. Designed in accordance with USP General Chapter and aligned with ICH Q5C stability guidance, the D1200 PRO provides a validated, GLP-ready architecture for regulatory-compliant dissolution method development and quality control.
Key Features
- 12-channel parallel immersion cell configuration with independent temperature monitoring per vessel position, ensuring uniform thermal equilibrium across all test units.
- Integrated water bath with 46 L capacity and high-efficiency circulation system, maintaining stable thermal conditions (±0.2 °C) from ambient up to 50 °C—critical for simulating physiological skin surface temperatures during topical product evaluation.
- Automated sampling module with programmable timing, volume selection (0.1–10 mL), and sequential aspiration via inert PTFE tubing to prevent adsorption or cross-contamination.
- Rugged stainless-steel chassis with corrosion-resistant internal components, compliant with ISO 9001-certified manufacturing practices and suitable for continuous operation in regulated QC laboratories.
- Modular design supporting both standalone operation and seamless integration with external HPLC/UHPLC autosamplers or UV/Vis spectrophotometers via RS-232 or Ethernet interfaces.
Sample Compatibility & Compliance
The D1200 PRO is specifically validated for use with semi-solid formulations that exhibit low mobility, high viscosity, or heterogeneous phase distribution—such as hydrogels, emulsion-based creams, and polymer-matrix patches—where conventional dissolution methods fail to generate discriminatory profiles. Each immersion cell accommodates standard USP-compliant membranes (e.g., cellulose ester, polysulfone) with defined molecular weight cutoffs and thickness specifications. The system supports full compliance with pharmacopoeial requirements including USP , Ph. Eur. 2.9.40, and JP 17 , and is routinely deployed in laboratories undergoing FDA pre-approval inspections and EMA GMP audits. All thermal and timing parameters are traceable to NIST-traceable calibration standards, and the system architecture supports 21 CFR Part 11-compliant electronic records when paired with validated acquisition software.
Software & Data Management
Control and data acquisition are managed through ANDISSO Dissolution Suite v3.2—a Windows-based application offering protocol-driven method definition, real-time temperature and sampling event logging, and automated calculation of cumulative release percentages. The software enforces audit trail functionality with user-level access controls, electronic signatures, and immutable record retention. Raw data exports comply with ASTM E2500-17 guidelines for analytical instrument data integrity, supporting CSV, PDF, and XML formats compatible with LIMS integration. Optional validation packages include IQ/OQ documentation, risk assessments per ISO 14971, and computerized system validation (CSV) templates aligned with Annex 11 and PIC/S PI 011-3.
Applications
- Development and optimization of in vitro release testing (IVRT) methods for generic topical products under FDA’s “Guidance for Industry: In Vitro Release Testing for Topical Semi-Solids” (2022).
- Comparative dissolution profiling of innovator vs. generic transdermal systems under discriminatory hydrodynamic conditions.
- Stability-indicating release studies supporting shelf-life determination per ICH Q1A(R3) and Q5C.
- Supporting biowaiver submissions by establishing in vitro–in vivo correlation (IVIVC) for semi-solid formulations where systemic absorption is limited and local bioavailability is the critical quality attribute.
- QC batch release testing in compliance with pharmacopoeial monographs requiring immersion cell methodology.
FAQ
What types of dosage forms is the D1200 PRO specifically designed for?
It is engineered for semi-solid and topical formulations—including creams, gels, ointments, pastes, and transdermal patches—where immersion cell methodology is pharmacopoeially mandated or scientifically justified.
Does the system support 21 CFR Part 11 compliance?
Yes, when operated with the validated Dissolution Suite v3.2 software and appropriate administrative controls, it meets electronic signature, audit trail, and data integrity requirements specified in 21 CFR Part 11.
Can the immersion cells be calibrated independently per vessel position?
Each vessel position includes a dedicated Pt100 sensor with individual calibration offset storage; full system calibration is performed using NIST-traceable reference thermometers per USP verification protocols.
Is the water bath volume sufficient for extended dissolution runs (e.g., 24–72 hours)?
The 46 L capacity ensures minimal evaporation-induced concentration drift over multi-day tests, especially when operated under humidity-controlled laboratory environments and with optional bath cover accessories.
How is sample carryover prevented during automated aspiration?
The fluid path uses chemically inert PTFE tubing with programmable rinse cycles between samples; all wetted surfaces are validated for extractables and leachables per USP .