COMECAUSE IN-Y300 Automated Antibiotic Zone-of-Inhibition Analyzer and Potency Measurement System
| Brand | COMECAUSE |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | IN-Y300 |
| Price | USD 7,200 (FOB) |
Overview
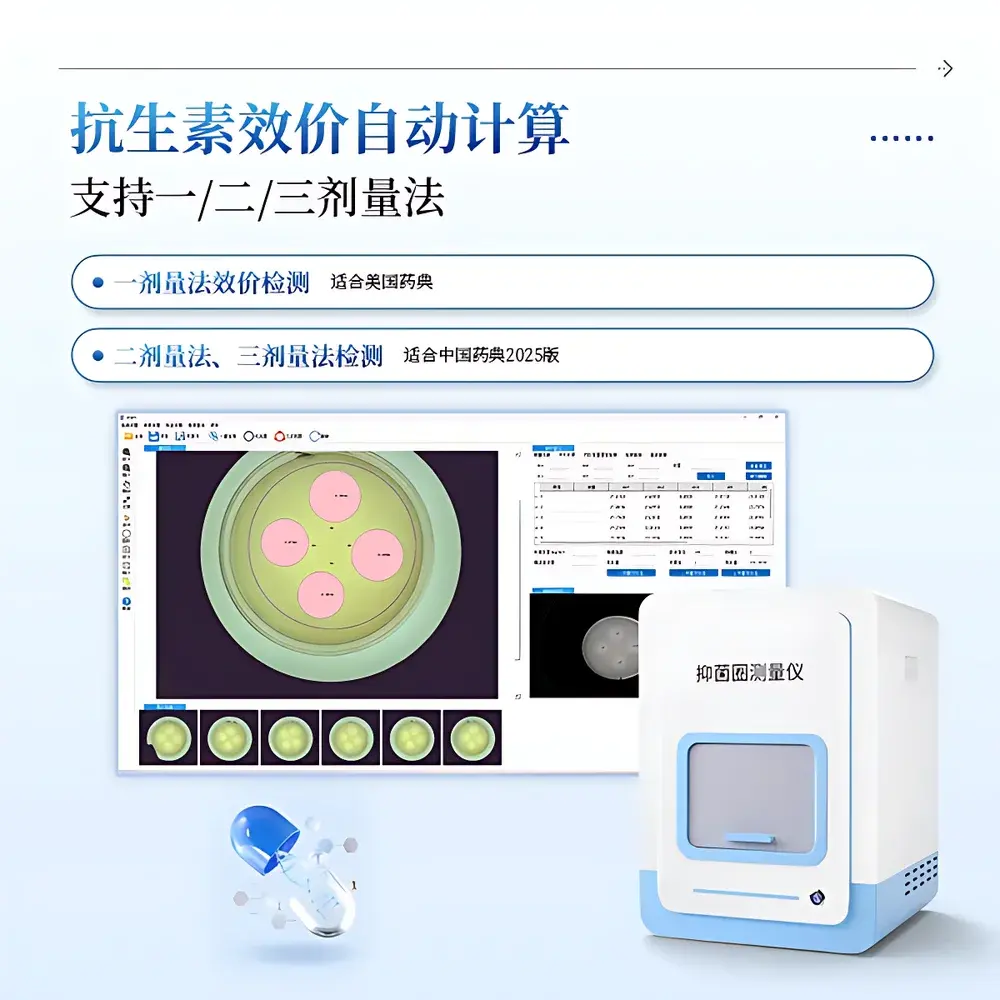
The COMECAUSE IN-Y300 is a fully automated, image-based zone-of-inhibition (ZOI) analyzer engineered for quantitative antibiotic potency assessment in compliance with pharmacopoeial standards. It operates on the principle of digital image metrology—capturing high-resolution grayscale or color images of agar diffusion plates under controlled illumination, then applying edge-detection algorithms and geometric fitting to determine the precise diameter (in mm) of inhibition zones surrounding antibiotic-impregnated discs or wells. This measurement serves as the primary input for calculating microbial growth inhibition response, enabling dose–response modeling required by USP , EP 2.6.6, and ChP 2025 Volume IV (Antibiotic Microbiological Assays). Designed for routine QC/QA laboratories, pharmaceutical stability testing units, and academic microbiology research facilities, the IN-Y300 replaces manual caliper-based measurements with traceable, operator-independent analysis—reducing inter-operator variability and supporting GLP/GMP-aligned workflows.
Key Features
- Fully enclosed dark chamber with electromagnetic shielding to eliminate ambient light interference and ensure optical consistency across repeated assays.
- Dual independent illumination system: top-mounted 360° quad-color (RGB+white) LED ring light for shadow-free surface illumination; bottom-transmitted white LED array for enhanced contrast of translucent zones; integrated 254 nm UV-C lamp for in-chamber sterilization between runs.
- 12 MP color CMOS sensor paired with a 12 mm focal-length macro lens (f/2.0), optimized for uniform resolution across 90–110 mm Petri dishes; compatible with smaller plates via central placement and software-defined ROI cropping.
- Three-tier ZOI detection algorithm: (1) Auto-edge detection for sharp, circular zones; (2) Circle-fitting mode for elliptical or irregular zones using least-squares optimization; (3) Manual three-point circle definition for low-contrast or diffused boundaries.
- Pharmacopoeia-compliant potency calculation engine supporting one-dose (USP), two-dose (ChP 2025), and three-dose assay designs—including parallel-line analysis, slope-ratio computation, and confidence interval estimation per ICH Q5C guidelines.
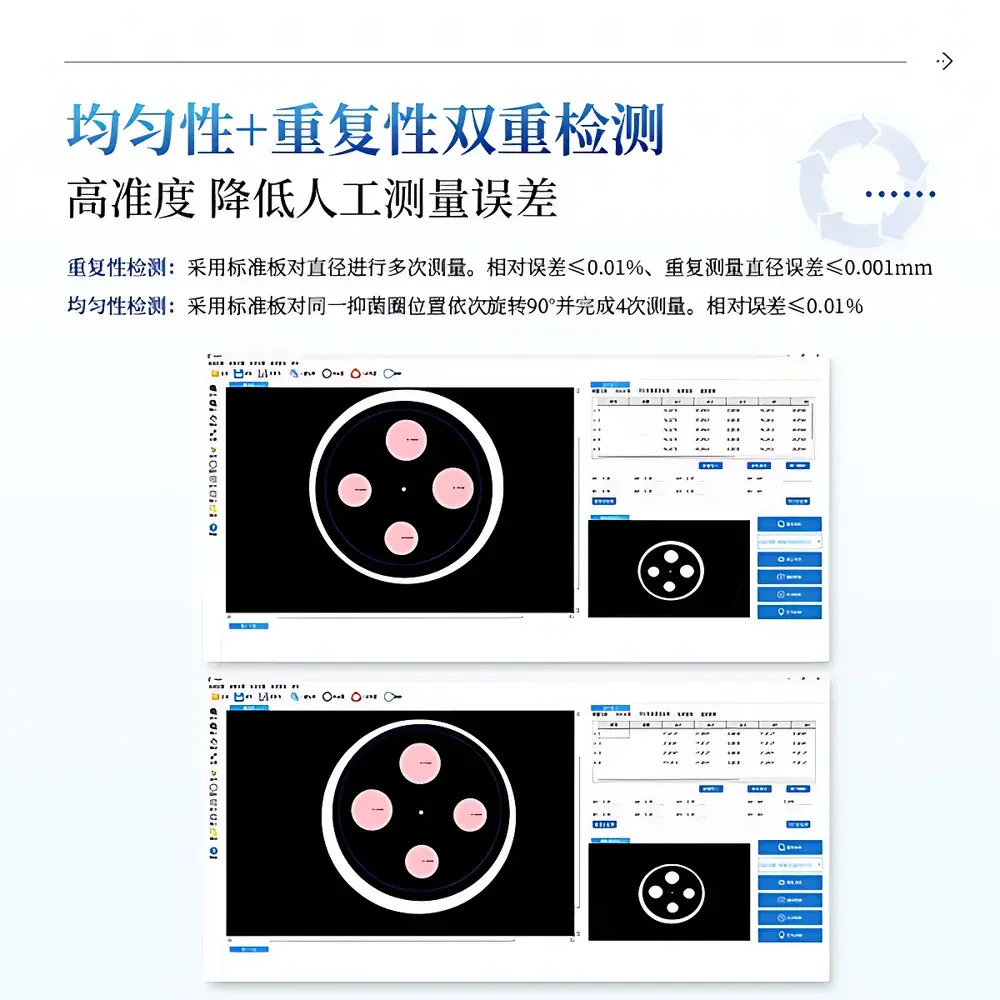
- Traceable calibration framework: dual-mode calibration (automatic via intrinsic camera parameters + manual via NIST-traceable stainless-steel standard plate); pixel-to-mm conversion validated at ±0.01 mm repeatability (n = 20, CV ≤ 0.02%).
Sample Compatibility & Compliance
The IN-Y300 accommodates standard Mueller–Hinton, Sabouraud dextrose, and nutrient agar plates (90–110 mm diameter), as well as custom-sized plates down to 35 mm when centrally positioned. It supports all common antibiotic disc types (6 mm, 8 mm), well diffusion formats (e.g., 6-mm punch wells), and gradient strip assays (e.g., E-test). The system meets ISO/IEC 17025 requirements for measurement uncertainty estimation and complies with data integrity principles outlined in FDA 21 CFR Part 11 (electronic records/signatures), including role-based access control, immutable raw image archiving, and full audit trail logging (user ID, timestamp, action type, parameter changes). All potency reports include metadata required for regulatory submission: assay date, analyst ID, reference standard lot number, dilution series, and statistical residuals.
Software & Data Management
The proprietary IN-Analysis Suite v4.2 provides bilingual (English/Chinese) GUI with configurable dashboards and real-time preview overlays. It features embedded statistical modules for uniformity-of-measurement validation (4-point rotational test per ChP Annex XIX), outlier rejection based on user-defined SD thresholds, and customizable annotation layers (text, arrows, scale bars). Raw images are stored in lossless TIFF format with embedded EXIF metadata; processed results export to PDF (with digital signature support), CSV (for LIMS integration), and XML (for CDISC SEND compliance). Cloud synchronization is implemented via TLS 1.3-encrypted REST API to COMECAUSE’s ISO 27001-certified data center, enabling remote review, multi-site collaboration, and version-controlled assay protocol deployment.
Applications
- Antibiotic potency release testing in pharmaceutical manufacturing (API and finished dosage forms).
- Stability-indicating method development for degradation product interference assessment.
- Microbial susceptibility profiling in clinical microbiology labs (CLSI M02/M07 implementation support).
- Disinfectant efficacy validation per EN 13697 and ASTM E2149.
- Teaching applications in undergraduate and graduate microbiology curricula—demonstrating quantitative bioassay design and statistical validation principles.
- Environmental monitoring of antimicrobial resistance (AMR) hotspots in wastewater and soil samples.
FAQ
Does the IN-Y300 support FDA 21 CFR Part 11 compliance?
Yes—the system implements electronic signature workflows, audit trail logging with tamper-evident hashing, and role-based permission tiers (Admin, Analyst, Reviewer) meeting Part 11 Subpart B requirements.
Can it process plates with overlapping or fused inhibition zones?
The software includes semi-automated zone separation tools using watershed segmentation and user-guided boundary refinement, though fused zones require manual intervention per USP Section 5.2.
Is calibration traceable to national standards?
Manual calibration uses a certified 10-mm pitch stainless steel standard plate (certified to ISO 6988:2022); certificate of calibration is provided with each instrument shipment.
What is the maximum throughput per hour?
With batch processing enabled, the system analyzes up to 48 standard 90-mm plates per hour (including auto-focus, capture, detection, and report generation).
Does it integrate with laboratory information management systems (LIMS)?
Yes—via HL7 v2.5 and ASTM E1384-compliant APIs; pre-built connectors available for Thermo Fisher SampleManager, LabVantage, and STARLIMS.