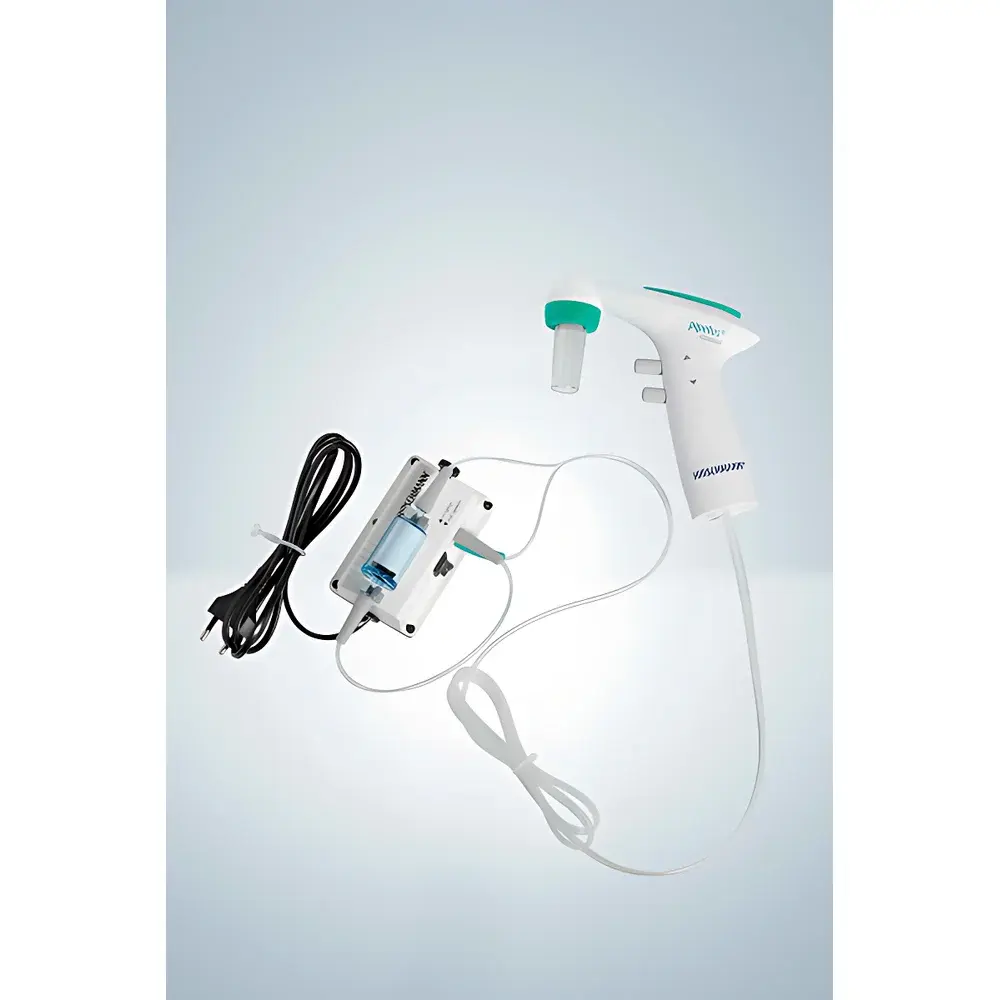
Hirschmann Pipetus-Standard Biosafety-Grade Electronic Pipette Controller
| Brand | Hirschmann (Germany) |
|---|---|
| Origin | Germany |
| Model | Pipetus-Standard |
| Type | Single-Channel Electric Pipette Controller |
| Volume Range | 0.1–200 mL |
| Maximum Capacity | 200 mL |
| Accuracy | ±0.5% |
| Weight (Handheld Unit) | 130 g |
| Housing Material | Polyamide 6 (PA6) |
| Sterilization Compatibility | 121 °C autoclaving (wet heat) for liquid/aerosol-contact components (excluding membrane filters) |
| Filter Option | Integrated bidirectional high-efficiency air filter (HEPA-grade, optional) |
| Catalog Numbers | 9903000 (without filter), 9904000 (with filter) |
Overview
The Hirschmann Pipetus-Standard Biosafety-Grade Electronic Pipette Controller is an engineered solution for precise, contamination-controlled liquid handling in biosafety level 2 (BSL-2) and BSL-3 laboratories. Unlike conventional electric pipette controllers with integrated electronics in the handheld unit, the Pipetus-Standard adopts a split architecture: the motorized aspiration and dispensing mechanism resides in a sterile, autoclavable handheld body, while the vacuum source is external—driven via a dedicated pump equipped with a bidirectional high-efficiency particulate air (HEPA) filter. This design eliminates internal electronic components from direct exposure to aerosols or splashes, enabling full 121 °C steam sterilization of all wetted and aerosol-contact surfaces (per ISO 17664 and EN 285 standards), except for the disposable hydrophobic membrane filter. The device operates on the principle of controlled vacuum generation and pressure modulation, delivering reproducible volumetric transfer across its full 0.1–200 mL range without compromising sterility integrity.
Key Features
- Autoclavable handheld unit: All liquid- and aerosol-contact components—including housing, control shaft, and ceramic-tipped volume adjustment knob—rated for repeated 121 °C, 20-minute steam sterilization cycles per DIN EN 285.
- External HEPA-filtered pump system: Bidirectional filtration ensures both intake and exhaust air streams are filtered to ≥99.97% efficiency at 0.3 µm particle size (EN 1822-1), preventing cross-contamination between samples and protecting the pump from biological load.
- Ceramic-tipped precision control lever: Chemically inert aluminum oxide ceramic pin provides long-term resistance to corrosion from acids, bases, and organic solvents—critical for repeated decontamination with ethanol, hydrogen peroxide vapor, or sodium hypochlorite.
- Adjustable aspiration/dispense speed: Independent control over suction and blow-out rates enables optimization for viscous media, volatile solvents, or low-volume transfers requiring meniscus-level accuracy.
- Universal silicone adapter: Compliant with Class A and Class B glass pipettes (ISO 648, ISO 1042) and polypropylene/PS plastic pipettes across 0.1–200 mL nominal volumes; no adapter changes required.
- Lightweight ergonomic design: 130 g handheld mass minimizes operator fatigue during extended use; non-porous PA6 polymer surface resists microbial adhesion and supports validated wipe-down protocols (e.g., 70% IPA, 0.5% NaOCl).
Sample Compatibility & Compliance
The Pipetus-Standard is validated for use with aqueous buffers, cell culture media, viral lysates, clinical specimens, and low-viscosity reagents commonly handled in diagnostic, vaccine development, and molecular biology workflows. It complies with IEC 61010-1 (Safety Requirements for Electrical Equipment for Measurement, Control, and Laboratory Use) and meets essential requirements of the EU Medical Device Regulation (MDR 2017/745) when used as an ancillary device in GLP- and GMP-regulated environments. Its sterilization protocol aligns with WHO TRS 961 Annex 5 and USP for sterilizing reusable laboratory devices. Documentation includes traceable autoclave validation reports and filter integrity test certificates (DOP/NaCl challenge per ISO 14644-3).
Software & Data Management
As a standalone analog-electromechanical instrument, the Pipetus-Standard does not incorporate embedded firmware, touchscreen interfaces, or digital data logging—intentionally eliminating cybersecurity risks and software validation burdens. All operational parameters are set manually via tactile controls; no electronic records are generated. This architecture supports strict adherence to FDA 21 CFR Part 11 requirements where electronic records are *not* mandated, and facilitates seamless integration into paper-based SOPs and audit-ready lab notebooks. Optional pump units may include analog pressure gauges calibrated per ISO/IEC 17025-accredited procedures.
Applications
- Safe aspiration of infectious samples inside Class II A2/B2 biosafety cabinets without compromising cabinet airflow integrity.
- Serial dilution of high-titer viral stocks under containment conditions with minimized aerosol generation.
- Transfer of primary human cells or organoid cultures where surface sterility and chemical residue absence are critical.
- Preparation of NGS library inputs in cleanroom-adjacent workflows requiring frequent equipment decontamination.
- QC testing of disinfectants and antimicrobial agents where instrument carryover must be eliminated between replicates.
FAQ
Can the entire handheld unit be autoclaved?
Yes—the handheld body, control lever assembly, and silicone adapter are fully autoclavable at 121 °C for 20 minutes. The membrane filter (if installed) must be removed prior to sterilization.
Is the device suitable for use in explosion-hazard areas?
When paired with an ATEX-certified external pump (e.g., Hirschmann P-Ex series), the system meets Zone 2/22 classification requirements for non-incendive operation.
Does it support regulatory audit trails?
No electronic logs are generated. Full traceability is maintained through manual logbook entries, autoclave cycle records, and filter replacement documentation per ISO 9001 and ISO/IEC 17025.
What pipette types are compatible?
All standard taper (ISO 648) and fast-fit (ISO 1042) glass and plastic serological pipettes from 0.1 mL to 200 mL, including borosilicate, soda-lime, and polypropylene variants.
How often should the HEPA filter be replaced?
Filter service intervals depend on usage intensity and aerosol load; typical replacement is recommended every 6–12 months or after 200 autoclave cycles—validated via differential pressure monitoring per EN 1822-5.