CSChrom Plus Chromatography Data System (CDS) Workstation
| Brand | Laballiance |
|---|---|
| Origin | USA |
| Model | CSChrom Plus |
| Regulatory Compliance | FDA 21 CFR Part 11, GLP/GMP Audit Trail |
| Deployment Architecture | Single-user to Client/Server Network |
| Software Category | Chromatography Data System (CDS) for HPLC, GC, and UHPLC |
Overview
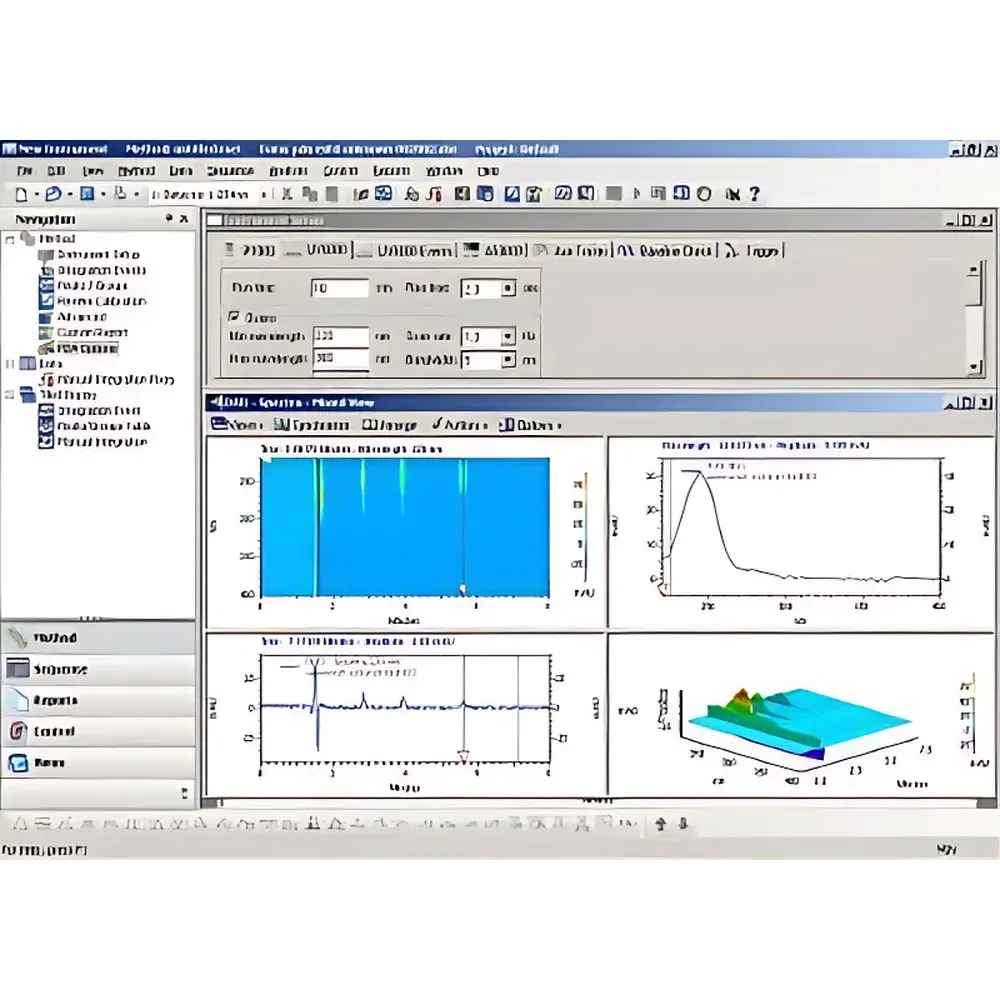
CSChrom Plus is a validated, enterprise-grade Chromatography Data System (CDS) engineered for regulatory-compliant chromatographic analysis in pharmaceutical, biotechnology, clinical, and contract research laboratories. Built on a modular, scalable architecture, it supports instrument control, real-time data acquisition, quantitative and qualitative peak processing, system suitability assessment, electronic signature management, and secure long-term data archiving. The software implements core chromatographic measurement principles—including retention time alignment, peak integration via adaptive baseline algorithms, spectral deconvolution (for PDA/DAD), and multi-wavelength absorbance ratio analysis—enabling traceable, reproducible interpretation of complex elution profiles across HPLC, UHPLC, and GC platforms.
Key Features
- Regulatory-Ready Electronic Records & Signatures: Fully compliant with FDA 21 CFR Part 11 and Annex 11 requirements, including role-based electronic signatures, cryptographic hash integrity checks, and immutable audit trail logging for all user actions, method modifications, reprocessing events, and report generation.
- Comprehensive Audit Trail: Time-stamped, user-identified, and context-aware event logging—including file creation, parameter edits, integration rule changes, peak reintegration, and report exports—stored separately per project and retained in tamper-evident format for GLP/GMP audits.
- Intuitive Instrument Control & Monitoring: Unified driver interface supporting major HPLC, GC, and autosampler manufacturers; real-time pressure, flow rate, detector signal, and oven temperature monitoring with configurable alarm thresholds and visual status indicators.
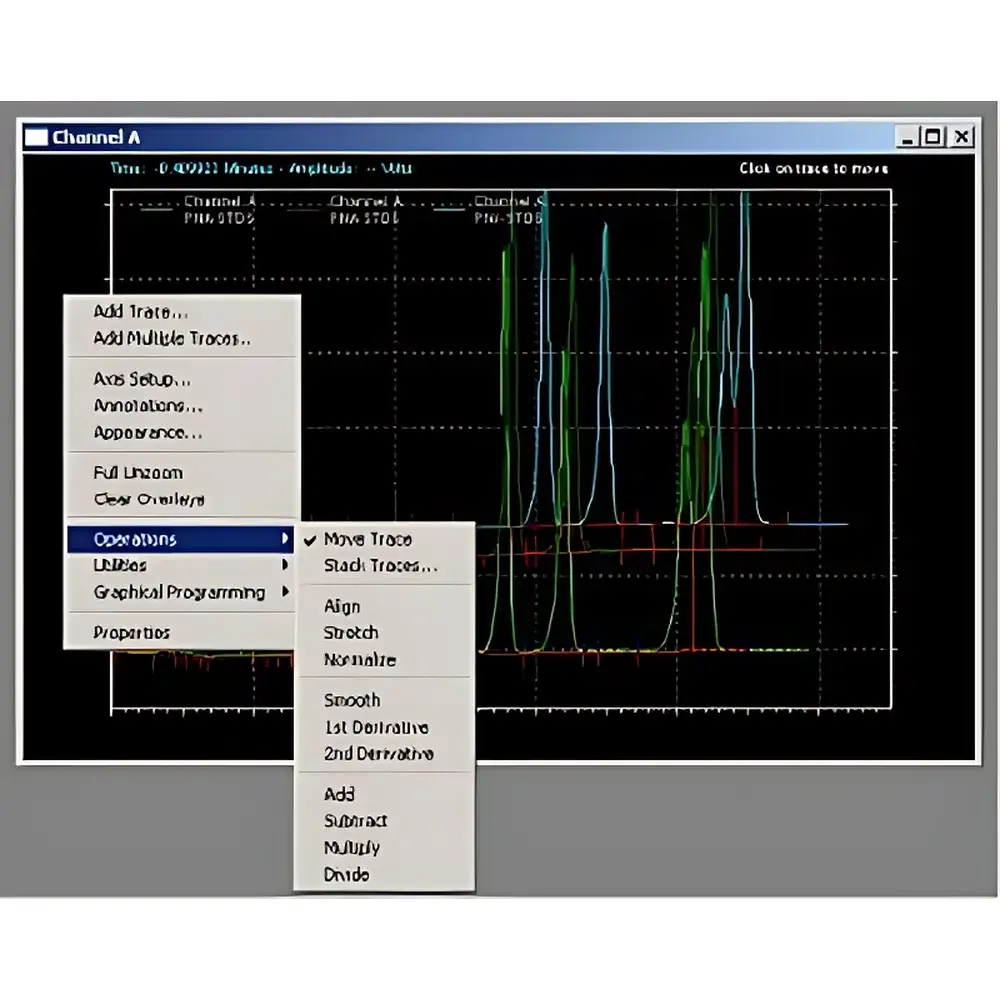
- Advanced PDA/DAD Processing: 3D contour plots, spectral overlay, peak purity assessment (via spectral correlation and peak tracking), library searching (NIST, Wiley, custom), and wavelength-ratio mapping for co-elution detection and compound identification support.
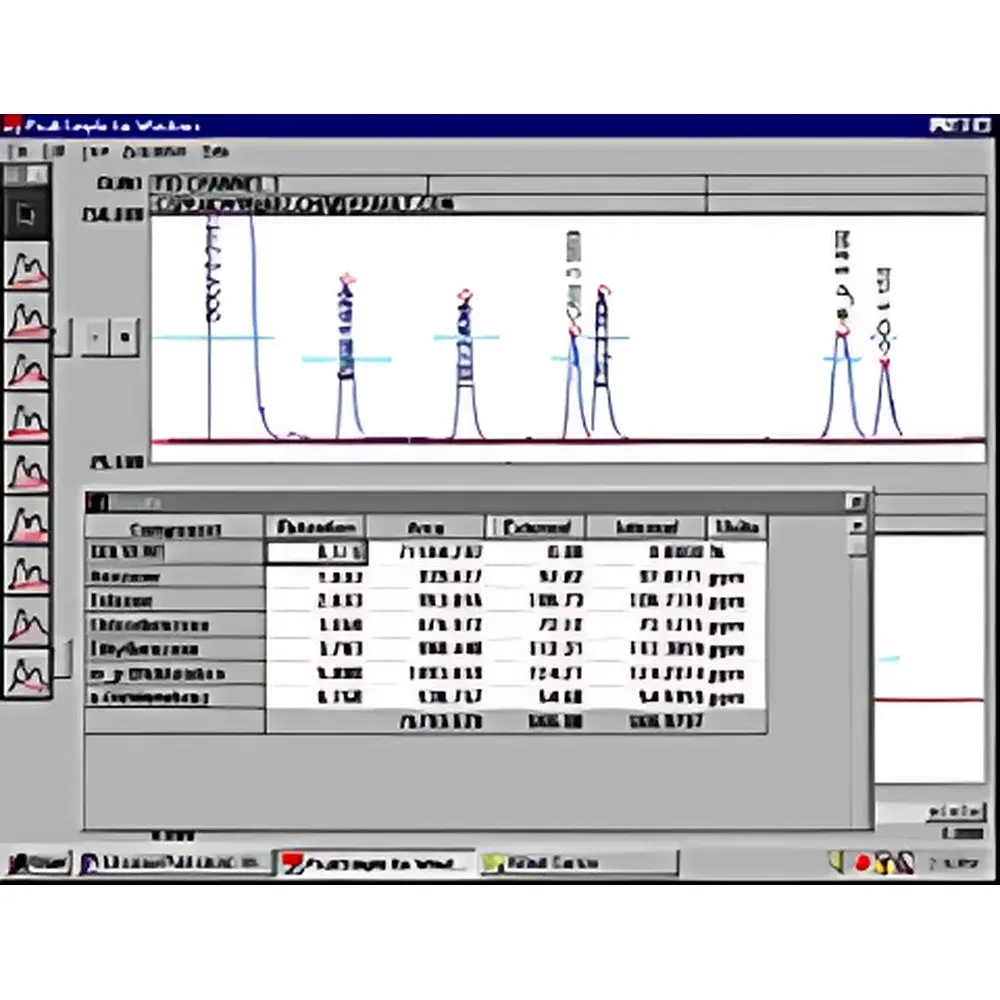
- Graphical & Scriptable Data Integration: Drag-and-drop baseline manipulation, manual peak splitting/merging, tangent skim, valley-to-valley, and exponential skimming algorithms; integration rules can be saved, versioned, and applied across batches.
- Flexible Sample Sequence Management: Visual sample rack editor with vial position mapping; dynamic sequence modification (insert, skip, repeat, requeue); automated re-injection triggers based on system suitability failure or retention time drift thresholds.
Sample Compatibility & Compliance
CSChrom Plus natively acquires and processes analog and digital signals from UV-Vis, fluorescence, conductivity, mass spectrometry (via vendor-neutral .mzML import), and electrochemical detectors. It supports raw data formats from Agilent, Waters, Thermo Fisher, Shimadzu, and PerkinElmer systems through certified drivers. All data objects—including methods, sequences, results, and reports—are stored in a structured, relational database (Microsoft SQL Server) with ACID compliance. The system undergoes periodic validation per ICH Q2(R2) and ASTM E2500-22 guidelines, and includes built-in tools for IQ/OQ documentation templates aligned with GAMP 5 practices.
Software & Data Management
- Multi-Tier Deployment: Operates as standalone desktop application or scales to client/server topology with centralized database, load-balanced acquisition nodes, and web-based remote monitoring dashboard.
- Search & Retrieval Engine: Full-text indexing of sample IDs, analyst names, injection dates, method versions, and custom metadata fields; cross-project queries with Boolean logic and export to CSV, PDF, or XML.
- Report Generation Framework: WYSIWYG report designer with drag-and-drop layout controls, dynamic field binding, conditional formatting, embedded Excel-style calculations, and template-based batch reporting (e.g., QC release summaries, stability study reports).
- Security Model: Hierarchical user roles (Administrator, Supervisor, Analyst, Reviewer) with granular permissions per module (acquisition, processing, reporting, audit trail review); password policies, session timeouts, and Windows Active Directory integration supported.
Applications
CSChrom Plus is routinely deployed in quality control laboratories for pharmacopeial testing (USP <621>, EP 2.2.46), stability-indicating assay development, impurity profiling, dissolution testing, residual solvent analysis (ICH Q3C), and extractables/leachables studies. Its system suitability engine automates evaluation of resolution, tailing factor, theoretical plates, and %RSD for replicate injections—generating pass/fail flags aligned with USP <621> criteria. In regulated bioanalysis, the software supports PK/PD data reduction workflows compliant with FDA Bioanalytical Method Validation Guidance (2018), including LLOQ determination, carryover assessment, and matrix effect correction.
FAQ
Does CSChrom Plus support 21 CFR Part 11-compliant electronic signatures?
Yes. It provides tiered signature types (initials, full name, biometric-ready), signature justification fields, and dual-control approval workflows for critical operations such as result approval and method validation.
Can audit trail data be exported for regulatory inspection?
Yes. Audit trail records are exportable in PDF/A-2 or CSV format with digital signature verification metadata, preserving chronological integrity and user attribution without modification.
Is CSChrom Plus qualified for use in GMP manufacturing environments?
Yes. The software includes preconfigured qualification protocols (IQ/OQ/PQ), change control documentation templates, and version-controlled method libraries meeting EU GMP Annex 11 and PIC/S PI 011-3 requirements.
How does CSChrom Plus handle data backup and disaster recovery?
It integrates with enterprise backup solutions via VSS-compatible SQL Server backups, supports point-in-time recovery, and optionally replicates the database to geographically redundant sites using SQL Always On Availability Groups.
Can third-party instruments be added post-deployment?
Yes. New instrument drivers are certified and deployed via controlled software updates; each driver undergoes functional testing against manufacturer specifications and documented in the system’s validation master file.