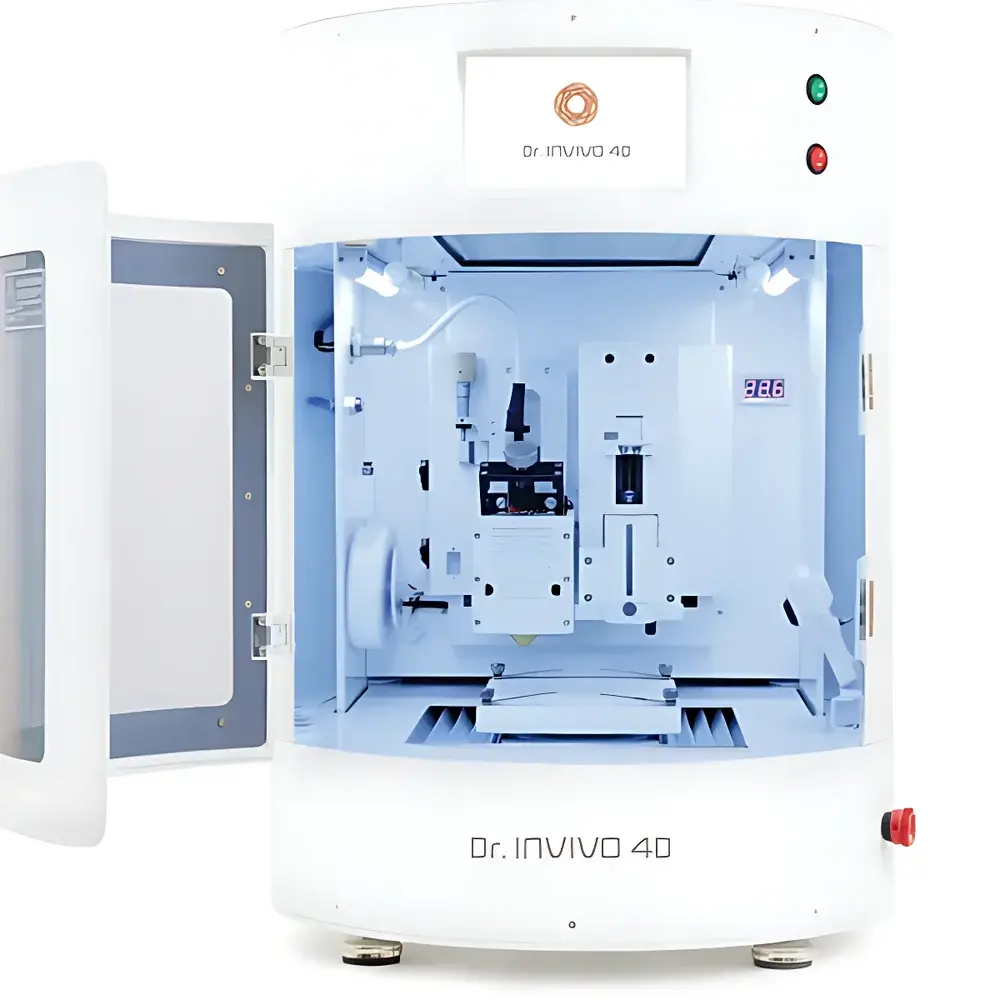
INVIVO 4D Biological 3D Printer by Newton
| Brand | Newton |
|---|---|
| Origin | South Korea |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | INVIVO 4D |
| Pricing | Upon Request |
Overview
The INVIVO 4D Biological 3D Printer by Newton is an advanced bioprinting platform engineered for high-fidelity, spatially controlled deposition of living cells and biomaterials under physiologically relevant conditions. Unlike conventional extrusion-based 3D printers, the INVIVO 4D integrates real-time environmental modulation—including temperature, humidity, CO₂ concentration, and substrate pre-conditioning—to support cell viability throughout the printing process. Its core architecture leverages pneumatic-assisted microextrusion combined with dynamic printhead positioning and synchronized stage motion to achieve sub-100 µm feature resolution in X-Y-Z axes, enabling reproducible fabrication of complex 3D tissue architectures. Designed specifically for translational research environments, the system operates within Class II biosafety-compliant enclosures and supports sterile workflow integration via ISO 5 (Class 100) laminar flow compatibility. It is not intended for clinical manufacturing or GMP production but serves as a validated tool for preclinical tissue model development, regulatory-grade assay prototyping, and academic discovery.
Key Features
- Pneumatic microextrusion system with pressure-controlled dispensing (0.1–800 kPa range) for precise bioink rheology adaptation
- Integrated environmental chamber maintaining 36.5 ± 0.3°C, 5% CO₂, and >95% RH during printing
- Multi-material capability: simultaneous handling of hydrogels, thermoresponsive bioinks, filamentous scaffolds, microparticulate suspensions, and paste-like composites
- Modular printhead configuration supporting up to four independent dispensing units (e.g., cell-laden hydrogel + crosslinker + support bath + fluorescent tracer)
- Automated calibration suite with optical feedback for nozzle alignment, bed leveling, and z-height optimization
- Open-architecture firmware allowing custom G-code injection and third-party sensor integration (e.g., force transducers, impedance monitors)
Sample Compatibility & Compliance
The INVIVO 4D accommodates a broad spectrum of biologically active feedstocks, including natural hydrogels (alginate, gelatin-methacryloyl, hyaluronic acid derivatives), synthetic polymers (PEGDA, Pluronic F127), decellularized extracellular matrix (dECM) bioinks, and hybrid composite pastes containing ceramic microparticles or drug-loaded microspheres. All material handling pathways are constructed from medical-grade stainless steel and autoclavable polymer components compliant with ISO 10993-5 cytotoxicity standards. The system meets CE marking requirements under Directive 2014/30/EU (EMC) and 2014/35/EU (LVD), and its software architecture supports audit trails aligned with GLP documentation practices. While not certified under FDA 21 CFR Part 11, it provides timestamped log export functionality suitable for internal validation protocols and ISO/IEC 17025-accredited laboratory workflows.
Software & Data Management
Control and monitoring are executed via BioPrint Studio v4.x—a Windows-based application built on Qt framework with dual-mode operation: guided wizard interface for novice users and scriptable command-line mode for advanced automation. The software includes native support for STL, 3MF, and AMF file formats, with embedded mesh repair tools and layer-by-layer simulation preview. Real-time telemetry logging captures extrusion pressure, stage position, environmental setpoints, and thermal gradients at 10 Hz sampling frequency. Export options include CSV, HDF5, and JSON-structured datasets compatible with MATLAB, Python (NumPy/Pandas), and commercial analysis platforms such as Imaris or Arivis Vision4D. All user actions, parameter changes, and error events are recorded in encrypted SQLite databases with SHA-256 hashing for traceability.
Applications
- Development of patient-specific tumor organoids for oncology drug screening
- Fabrication of vascularized cardiac patches with embedded endothelial progenitor cells
- Microfluidic organ-on-chip devices incorporating printed epithelial barriers and stromal compartments
- Controlled-release implant prototypes using gradient-doped hydrogel matrices
- Stem cell niche engineering via topographical and biochemical cue patterning
- Educational modules for undergraduate and graduate biomanufacturing curricula
FAQ
Is the INVIVO 4D suitable for Good Manufacturing Practice (GMP) environments?
No—it is designed for research use only and does not meet current GMP infrastructure or documentation requirements.
Can I integrate my own bioink formulation into the system?
Yes; the open fluidics interface allows full customization of dispensing parameters and compatibility testing with non-Newtonian and shear-thinning materials.
Does the system support live-cell imaging during printing?
It features standardized C-mount and M42 optical ports for integration with inverted fluorescence microscopes, though real-time imaging requires external hardware synchronization.
What sterilization methods are validated for the printhead assembly?
Autoclaving at 121°C for 20 minutes is validated for all metal and PEEK components; silicone tubing and syringe barrels require gamma irradiation or ethylene oxide treatment.
Is remote operation supported?
Yes—via secure RDP or VNC over local network; cloud connectivity is disabled by default to comply with institutional data governance policies.