Elite Chromatography Data Workstation
| Brand | Elite |
|---|---|
| Origin | Liaoning, China |
| Manufacturer Type | Manufacturer |
| Product Category | Domestic |
| Model | Chromatography Data Workstation |
| Price Range | USD 1,400 – 7,200 (based on configuration) |
| Software Type | Chromatography-Specific Data Acquisition & Management System |
| Regulatory Compliance | GLP/GMP, FDA 21 CFR Part 11, Chinese NMPA Guidelines for Chromatographic Data Integrity |
| Optional Modules | System Suitability Testing (SST), Gel Permeation Chromatography (GPC) Analysis, Photodiode Array (PDA) Data Processing, Multi-System Instrument Control |
| Audit Trail | Full, time-stamped, immutable, user-attributed event logging (login/logout, parameter changes, report generation, method edits, data deletion/reprocessing) |
Overview
The Elite Chromatography Data Workstation is a validated, regulatory-compliant software platform engineered for acquisition, processing, analysis, and archival of chromatographic data generated by HPLC, UHPLC, GC, and GPC systems. Built upon a relational database architecture, it implements deterministic data handling protocols aligned with international data integrity principles—specifically designed to meet the technical and procedural requirements of Good Laboratory Practice (GLP), Good Manufacturing Practice (GMP), and FDA 21 CFR Part 11. The workstation operates as a centralized control and reporting hub, supporting real-time instrument monitoring, method-driven sequence execution, and automated post-run evaluation. Its core architecture enforces data immutability: raw detector signals are stored in binary format with hash-verified integrity; all subsequent processing steps—including baseline correction, peak integration, calibration curve fitting, and purity assessment—are recorded as traceable, timestamped, and user-identified events within a tamper-evident audit trail.
Key Features
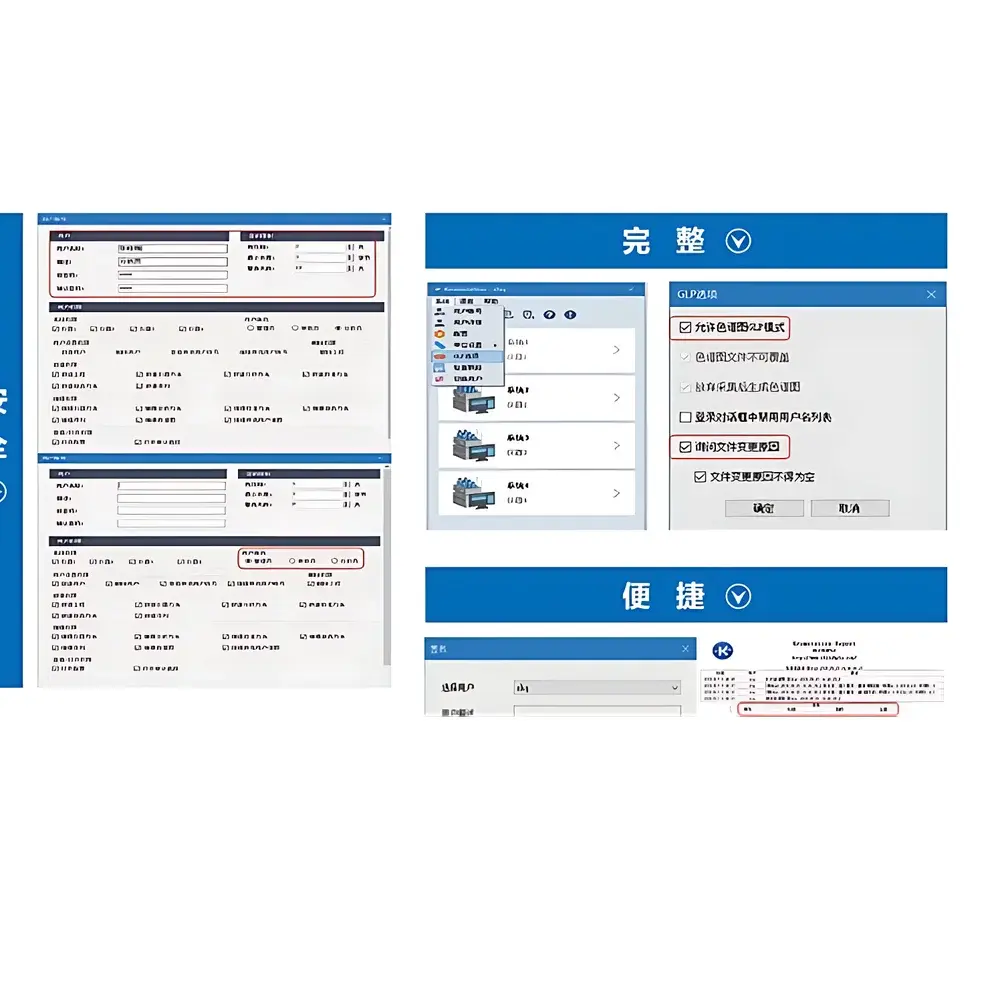
- Full 21 CFR Part 11 compliance: Electronic signatures, role-based access control (RBAC), password policies, and biometric or token-based authentication support.
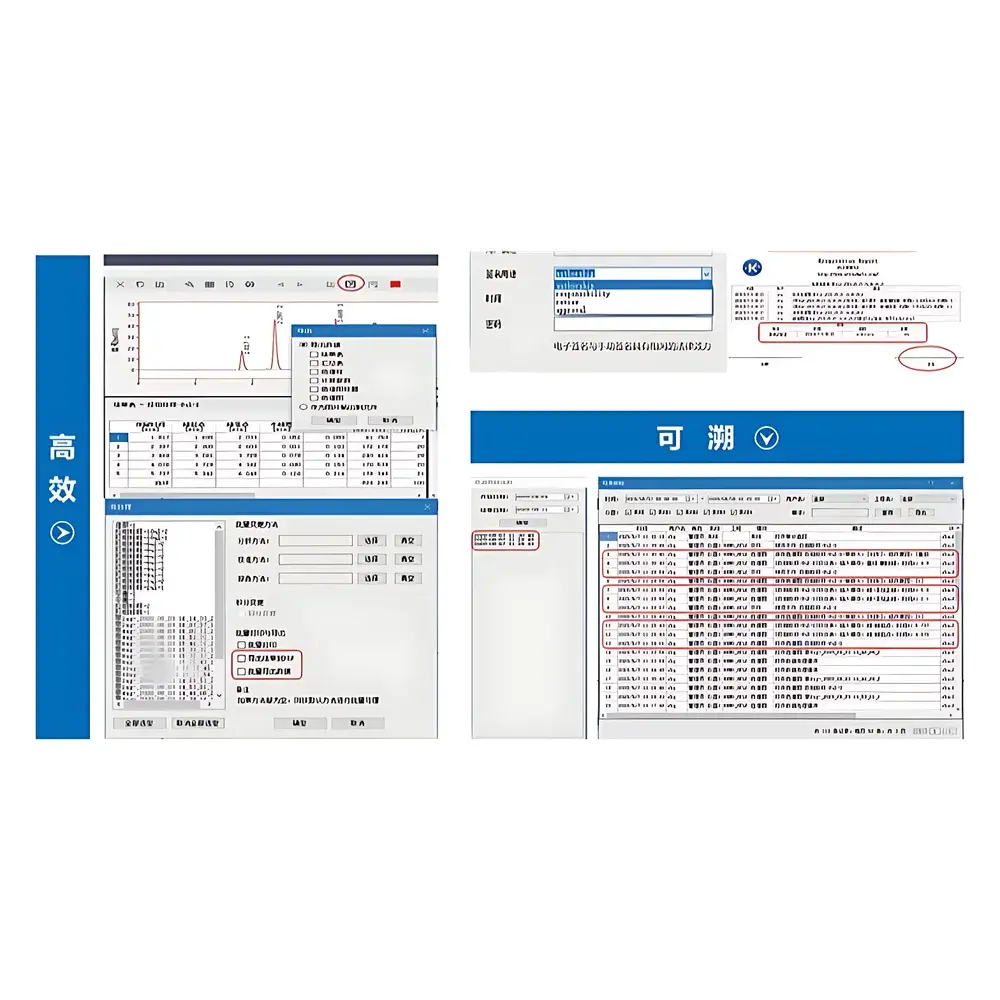
- Comprehensive audit trail: Captures every system-level and user-level action—including login/logout timestamps, method modifications, reprocessing events, report exports, and data deletion—with immutable storage and exportable XML/CSV logs for regulatory review.
- Modular architecture: Base platform supports optional licensed modules—SST (System Suitability Testing) for automated pass/fail evaluation against pre-defined criteria; PDA (Photodiode Array) module enabling multi-view spectral visualization (chromatogram, spectrum, contour plot, 3D surface, peak purity heatmaps, spectral library matching); and GPC module for automated molecular weight distribution calculation, including flow rate correction and multi-detector delay compensation.
- Multi-instrument control: Simultaneous acquisition and synchronization from up to four independent chromatographic systems (e.g., dual HPLC + GC + GPC), each with dedicated method templates and hardware abstraction layers.
- Database-backed data management: All raw data, processed results, methods, sequences, and audit logs reside in an embedded SQL Server Express or configurable Microsoft SQL Server instance—enabling structured querying, version-controlled method archiving, and long-term retention per ALCOA+ principles.
Sample Compatibility & Compliance
The workstation accepts analog voltage inputs (±5 V, 16-bit resolution) and digital signals (RS-232, USB-HID, Ethernet TCP/IP) from major OEM detectors and pumps. It supports standard chromatographic file formats (e.g., .CDF, .RAW, .D) via vendor-neutral import filters and exports analytical reports in PDF/A-1b, CSV, and XML formats compliant with ISO 19005-1. Validation documentation—including IQ/OQ/PQ protocols, risk assessments, and configuration specifications—is provided for GxP-regulated environments. The system has been successfully deployed in pharmaceutical QC labs undergoing FDA, EMA, and NMPA inspections, with documented evidence of audit trail completeness, electronic signature enforcement, and data lifecycle control across 10+ years of operational history.
Software & Data Management
Data integrity is enforced at the architectural level: raw acquisitions are written directly to the database without intermediate file caching; all processing parameters are versioned and linked to their associated raw dataset. The integrated reporting engine generates fully traceable analytical certificates—including peak tables with retention time, area%, height%, purity metrics, and calibration status—each digitally signed and archived with cryptographic hashes. Backup and recovery procedures follow NIST SP 800-33 guidelines, with optional integration into enterprise backup infrastructure via VSS-compatible interfaces. Data retention policies are configurable per project, sample type, or regulatory jurisdiction, supporting automatic archival to network-attached storage or cloud object storage (AWS S3, Azure Blob) with AES-256 encryption.
Applications
The Elite Chromatography Data Workstation is deployed in pharmaceutical development (method validation, stability studies), biologics characterization (SEC-MALS, IEX, RP-HPLC), polymer analytics (GPC/SEC for Mn, Mw, PDI), food safety testing (pesticide residue analysis), and environmental monitoring (PAHs, PCBs). Its PDA module enables rigorous peak purity assessment per USP and ICH Q2(R2), while the SST module automates system suitability checks per USP , EP 2.2.46, and JP 6.03. In regulated QC environments, the GPC module delivers reproducible molecular weight distributions under ISO 16014 and ASTM D5296 standards—critical for excipient qualification and polymer release testing.
FAQ
Does the workstation support electronic signatures compliant with 21 CFR Part 11?
Yes—electronic signatures are implemented with two-factor authentication, signature justification fields, and binding to specific actions (e.g., “Approve Report”, “Release Batch”).
Can audit trail records be exported for regulatory submission?
Yes—audit logs are exportable in XML and CSV formats, retaining full metadata (user ID, timestamp, action type, before/after values, IP address, session ID).
Is the GPC module compatible with third-party gel permeation chromatography systems?
Yes—the GPC module ingests analog outputs from any GPC system with ±5 V signal capability and supports custom detector response calibration.
What database options are supported for long-term data archiving?
Embedded SQL Server Express (default) or customer-provided Microsoft SQL Server 2016+, with support for AlwaysOn Availability Groups and Transparent Data Encryption (TDE).
How is software validation handled for GMP environments?
Elite provides a complete validation package—including URS, FRS, IQ/OQ/PQ protocols, traceability matrices, and change control documentation—aligned with GAMP 5 and Annex 11 requirements.