Avestin
Filter
Showing all 19 results
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Model | EF-B15 |
| Instrument Type | High-Pressure Homogenizer |
| Dimensions (W×D×H) | 335 × 280 × 650 mm |
| Sample Volume per Cycle | 3–15 mL |
| Construction Material | 316L Stainless Steel |
| Sterilization | Steam-Sterilizable |
| Cooling Option | Optional Heat Exchanger |
| Pressure Adjustment | Continuously Variable |
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Model | EmulsiFlex |
| Instrument Type | High-Pressure Homogenizer |
| Dimensions (W×D×H) | 335 × 280 × 650 mm |
| Processing Volume Range | 15–1,000,000 mL |
| Wetted Materials | Metal-to-Metal & Metal-to-Ceramic Seals Only |
| Sterilization | SIP-Compatible (Steam-in-Place), All Wetted Parts Autoclavable |
| Temperature Control | Stainless Steel Heat Exchanger Included |
| Regulatory Compliance | Designed for GLP/GMP Environments, Supports 21 CFR Part 11 Audit Trail Configuration (via optional software package) |
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Model | B15 |
| Instrument Type | High-Pressure Homogenizer |
| Dimensions (W×D×H) | 335 × 280 × 650 mm |
| Sample Volume Range | 3–15 mL |
| Operating Pressure | 45,000 psi (3,100 bar) |
| Air Supply | 5–10 bar |
| Residual Volume | < 0.1 mL |
| Drive Mechanism | Pneumatic Single Plunger |
| Weight | 37 kg |
| Construction Material | ASTM-certified 316L stainless steel |
| Compliance | FDA-compliant materials, GMP-compatible design, no elastomeric seals in product path |
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Model | EF-C3 |
| Instrument Type | High-Pressure Homogenizer |
| Dimensions (W×D×H) | 530 mm × 460 mm × 760 mm |
| Processing Capacity | 0.07–3 L/h |
| Maximum Pressure | 30,000 psi |
| Minimum Sample Volume | 7 mL |
| Sterilization | Autoclavable & SIP-Compatible (Steam-in-Place) |
| Construction | Metal-to-Metal & Metal-to-Ceramic Seals, O-Ring-Free Fluid Path |
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Model | EmulsiFlex-C3 |
| Instrument Type | High-Pressure Homogenizer |
| Dimensions (W×D×H) | 530 × 460 × 760 mm |
| Processing Volume Range | 7–3000 mL |
| Maximum Operating Pressure | 200 MPa (30,000 psi) |
| Sterilization | Direct Steam-in-Place (SIP) |
| Construction Materials | FDA-compliant 316L stainless steel |
| Compliance | Designed for GMP environments, SIP-capable, seal-free fluid path |
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Model | EF-C5 |
| Instrument Type | High-Pressure Homogenizer |
| Dimensions (W×D×H) | 430 × 160 × 320 mm |
| Processing Capacity | 7 mL – 5 L/h |
| Maximum Pressure | 30,000 psi |
| Sterilization | Fully Autoclavable & SIP-Compatible |
| Valve Design | Metal-to-Metal / Metal-to-Ceramic Sealing |
| Power Requirement | None (Hydraulically Actuated) |
| Temperature Control | Integrated Jacketed System |
| Optional Integration | In-Line Extrusion Module with 50/100/200/400 nm Track-Etched Membranes |
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Manufacturer | Avestin Inc., Ottawa, Canada |
| Model | EmulsiFlex-C5 |
| Instrument Type | High-Pressure Homogenizer |
| Operating Pressure Range | 500–30,000 psi (3.4–207 MPa) |
| Sample Volume Range | 7–5,000 mL |
| Flow Rate | 1–5 L/h |
| Dimensions (W×D×H) | 100 × 200 × 300 mm |
| Drive System | Pneumatic Single Plunger |
| Homogenizing Valve | Wear-Resistant Ceramic |
| Temperature Control Range | −25 to 200 °C (via external bath) |
| Sterilization | SIP (Steam-in-Place) and CIP (Clean-in-Place) compliant |
| Materials | Electropolished 316L stainless steel and FDA-compliant ceramic components |
| Regulatory Compliance | Designed and validated per GMP, FDA 21 CFR Part 11 (audit trail capable), ISO 13485, and USP <1043> for sterile processing equipment |
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Manufacturer | Avestin Inc., Ottawa, Canada |
| Model | EmulsiFlex-C50 |
| Max Operating Pressure | 30,000 psi (207 MPa) |
| Flow Rate | 15–50 L/h |
| Minimum Sample Volume | 25 mL |
| Drive Type | Pneumatic Dual-Piston |
| Safety | Auto-circuit cutoff with 20% transient overpressure tolerance |
| Construction Material | ASTM-certified 316L stainless steel |
| Sterilization | Direct steam-in-place (SIP), O-ring-free fluid path |
| Compliance | FDA 21 CFR Part 11-ready architecture, GMP-compliant design |
| Optional Accessories | Extrusion filter, heat exchanger |
| Instrument Category | High-Pressure Cell Disruptor |
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Model | EmulsiFlex-C50 |
| Maximum Operating Pressure | 30,000 psi (200 MPa) |
| Flow Rate | 15–50 L/h |
| Minimum Sample Volume | 25 mL |
| Drive Type | Pneumatic Dual-Piston |
| Chamber Material | ASTM-certified 316L stainless steel |
| Sterilization | In-line steam sterilization (SIP) |
| Compliance | FDA-compliant materials, GMP-compatible design |
| Dimensions (W×D×H) | 450 × 760 × 500 mm |
| Sample Compatibility | Aqueous & organic suspensions, liposomes, emulsions, nanocrystals, cell lysates |
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Manufacturer | Avestin Inc., Ottawa, Canada |
| Model | EmulsiFlex-C55 |
| Pressure Range | 0–30,000 psi (0–207 MPa) |
| Flow Rate | 15–50 L/h |
| Minimum Sample Volume | 7 mL |
| Drive Type | Pneumatic Dual-Piston |
| Compliance | GMP, FDA 21 CFR Part 11–ready architecture, ASME BPE–compatible materials |
| Construction Material | Electropolished 316L stainless steel |
| Sterilization | SIP/CIP-capable, O-ring–free fluid path |
| Safety | Auto-cutoff circuit protection, transient overpressure tolerance up to 20% |
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Manufacturer | Avestin Inc., Ottawa, Canada |
| Model | EmulsiFlex-C55 |
| Instrument Type | High-Pressure Homogenizer |
| Dimensions (W×D×H) | 600 × 430 × 540 mm |
| Processing Volume Range | 30–55,000 mL |
| Maximum Operating Pressure | 30,000 psi (207 MPa) |
| Flow Rate | Up to 55 L/h |
| Minimum Sample Volume | 7 mL |
| Drive Mechanism | Pneumatic Dual-Piston |
| Material Construction | Electropolished ASTM F899 316L Stainless Steel |
| Sterilization | SIP/CIP-Compatible Design |
| Regulatory Compliance | FDA 21 CFR Part 11–ready architecture, GMP-compliant construction, USP <1043> and <1207> aligned |
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Manufacturer | Avestin Inc., Ottawa, Canada |
| Model | EmulsiFlex-D20 |
| Instrument Type | High-Pressure Homogenizer |
| Operating Pressure | 35,000 psi (2,413 bar), typical working pressure up to 30,000 psi (2,070 bar) |
| Flow Rate | 20 L/h |
| Minimum Sample Volume | 50 mL |
| Residual Volume | < 1 mL |
| Drive System | Electric single-plunger pump |
| Dimensions | 730 × 600 × 640 mm |
| Weight | 120 kg |
| Power Supply | 3-phase, 50/60 Hz, 230/400 V |
| Safety Features | Automatic circuit cutoff, transient overpressure tolerance up to +20% |
| Construction Material | 316L stainless steel (entire fluid path) |
| Compliance | GMP-compliant design, FDA-compliant materials per 21 CFR Part 11 and USP <1058>, zero elastomeric seals in product-contact zones |
| Optional Accessories | Sterile filtration extruder, integrated heat exchanger |
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Manufacturer | Avestin Inc., Ottawa, ON |
| Model | EmulsiFlex-D20 |
| Operating Pressure | 35,000 psi (2,413 bar) / Max Continuous: 30,000 psi (2,070 bar) |
| Flow Rate | 20 L/h |
| Minimum Sample Volume | 50 mL |
| Sample Hold-up Volume | <1 mL |
| Drive Type | Electric Single-Plunger Pump |
| Dimensions (W×D×H) | 730 × 600 × 640 mm |
| Weight | 120 kg |
| Power Supply | 3-phase, 50/60 Hz, 230/400 V |
| Construction Material | ASTM A240 316L stainless steel + ceramic wear components |
| Sterilization | Direct steam-in-place (SIP) compatible |
| Compliance | GMP-compliant design, FDA 21 CFR Part 11–ready software architecture, USP <1043> and ISO 22442-1 applicable for bioprocess equipment qualification |
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Model | LF-1 |
| Type | Manual Piston-Driven Extrusion Device |
| Dimensions (W×D×H) | 280 × 40 × 40 mm |
| Sample Volume Range | 0–1 mL |
| Membrane Compatibility | Polycarbonate (PC) Track-Etched Filters (common pore sizes: 100 nm, 200 nm, 400 nm, 800 nm, 1 µm) |
| Construction | Stainless Steel Housing & Anodized Aluminum Components |
| Residual Volume | < 5 µL |
| Regulatory Compliance | Designed for GLP-compliant lab environments |
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Model | LF-50 |
| Instrument Type | High-Pressure Extrusion System |
| Dimensions (W×D×H) | 280 × 460 × 260 mm |
| Sample Volume Range | 5–50 mL per run |
| Compatible Membrane Pore Sizes | 10 nm – 1.0 µm (polycarbonate track-etched membranes) |
| Maximum Operating Pressure | 10,000 psi (69 MPa) |
| Material Contact Path | 316 Stainless Steel, PTFE, and Anodized Aluminum |
| Compliance | Designed for GLP-compliant lab environments |
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Model | LF-50 |
| Maximum Sample Volume | 50 mL per run |
| Operating Pressure | Up to 600 psi (41 bar) |
| Membrane Stack Configuration | Up to 3 polycarbonate membranes with support liners |
| Temperature Control | Jacketed stainless steel sample chamber compatible with external thermostatic circulators |
| Compliance | Designed for GMP-compliant processing and cleanability |
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Model | LF1 |
| Sample Volume | 0.1–1 mL |
| Membrane Pore Sizes | 50, 100, 200, 400, 800, 1000 nm |
| Operating Principle | Manual pressure-driven extrusion through polycarbonate track-etched membranes |
| Weight | <2 kg |
| Temperature Compatibility | Compatible with water bath (ambient to 60 °C) |
| Includes | Two 1 mL glass syringes, one extrusion module, fifty 100 nm membranes |
| Optional Accessories | Stabilizing base clamp, nitrogen-driven automated extrusion system |
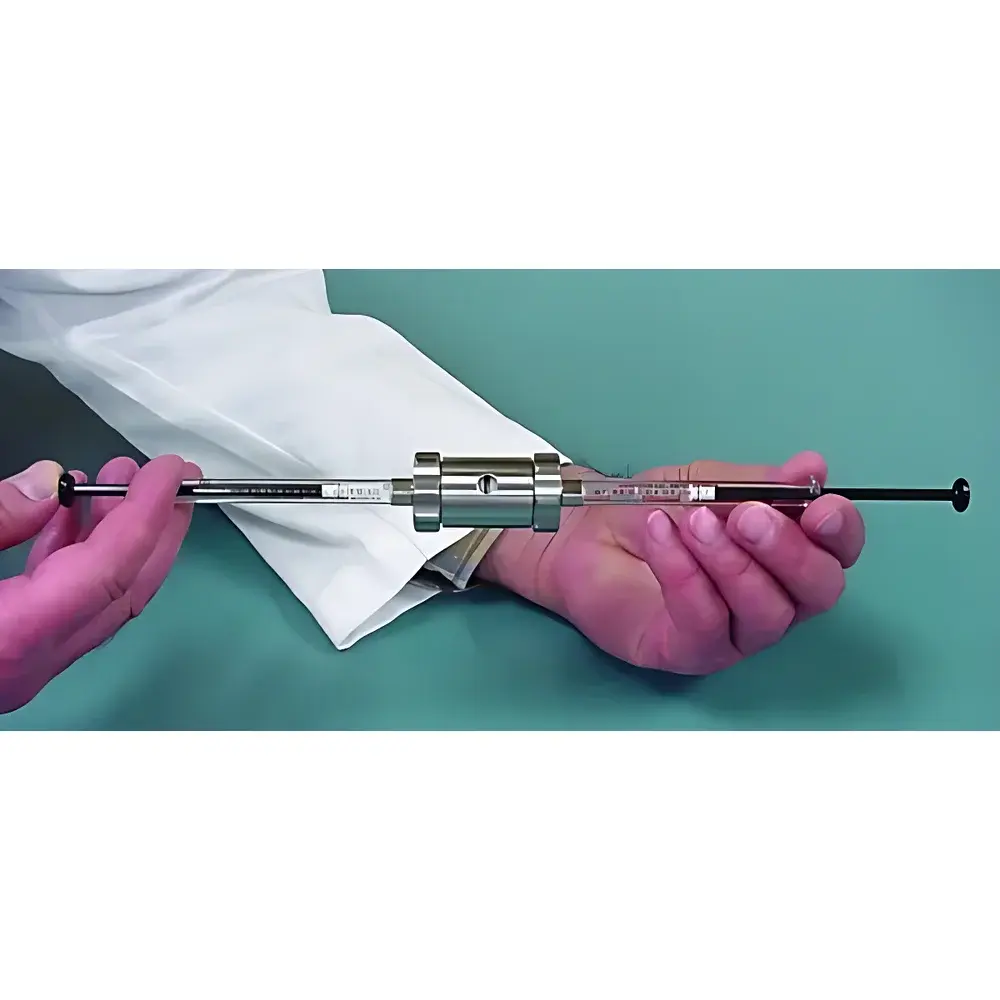
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Model | LiposoFast LF1 |
| Actuation | Manual |
| Sample Volume per Extrusion | 1 mL (2 × 500 µL syringes) |
| Minimum Sample Volume | 0.1 mL |
| Residual Volume | 0 µL |
| Available Membrane Pore Sizes | 50, 100, 200, 400, 800, and 1000 nm |
| Optional Temperature Control | Water-bath compatible |
| Weight | <2 kg |
| Compliance | Designed for GLP-compliant lab environments |
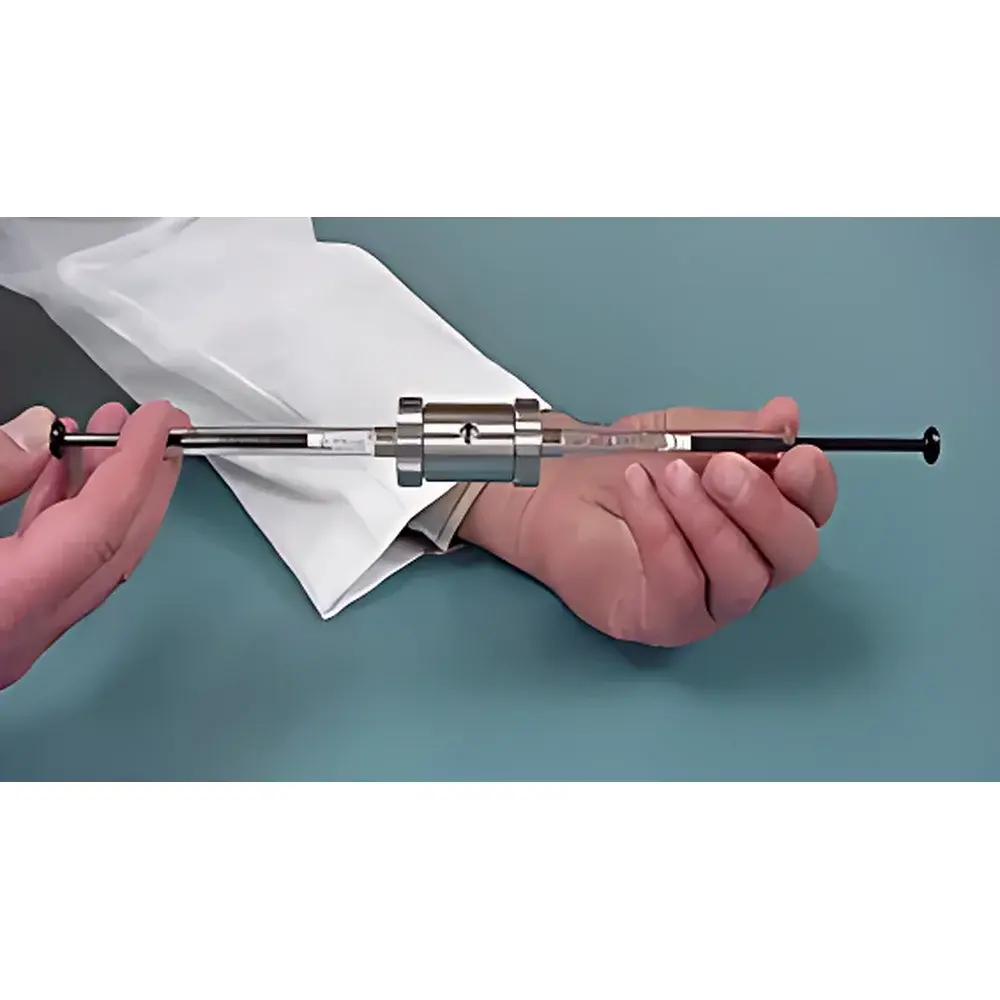
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Model | LipoSofast |
| Sample Volume per Cycle | 1 mL (2 × 500 µL syringes) |
| Minimum Sample Volume | 0.1 mL |
| Residual Volume | 0 µL |
| Available Polycarbonate (PC) Membrane Pore Sizes | 50, 100, 200, 400, and 800 nm |
| Temperature Compatibility | Water-bath compatible for thermal control (e.g., 4–40 °C) |
| Weight | < 2 kg |
| Construction | Borosilicate glass syringes, stainless steel housing, chemically resistant seals |