LUM GmbH
Filter
Showing all 2 results
| Brand | LUM GmbH |
|---|---|
| Origin | Germany |
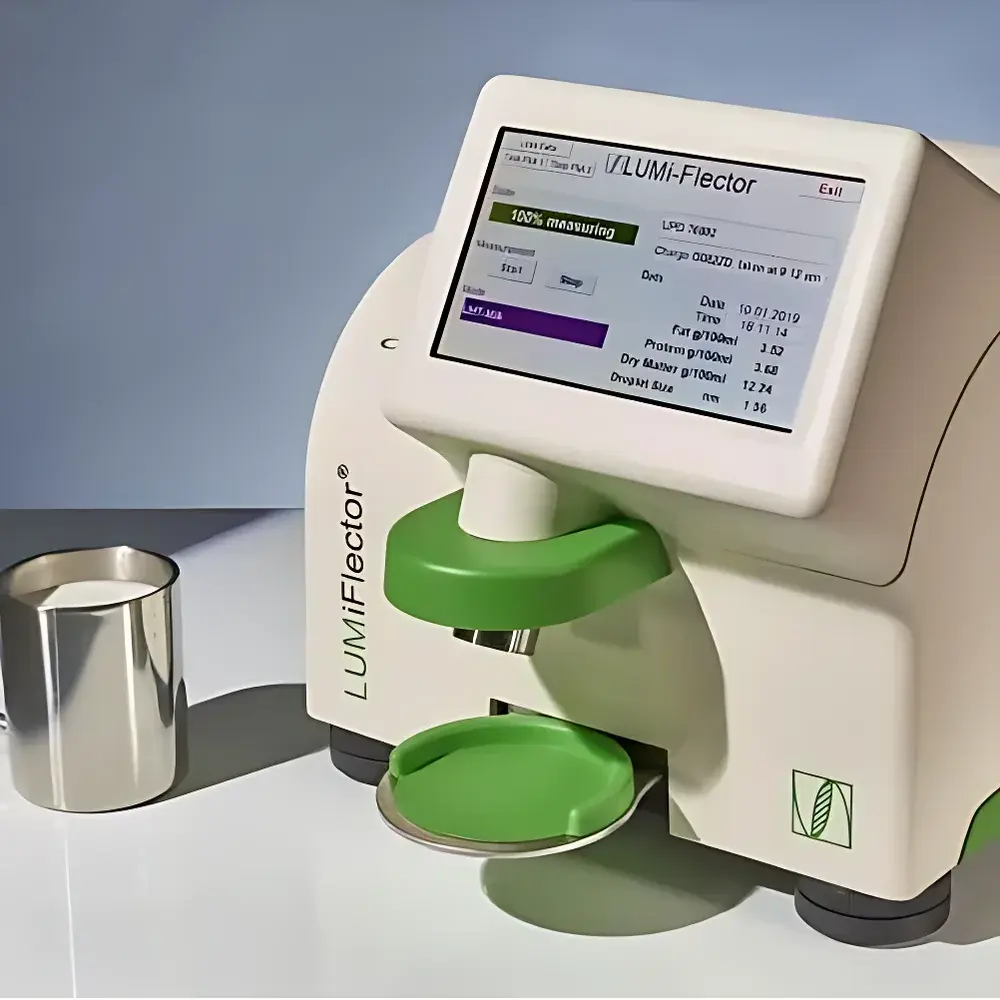
| Model | LUMiFlector |
| Application Principle | Near-Infrared and UV-Visible Multi-Reflection Spectroscopy (MRS®) |
| Measurement Time | <20 s (lab), <8 s (inline) |
| Sample Handling | Non-contact, no sample preparation, no reagents |
| Output Parameters | Up to 4 simultaneous quantitative attributes (e.g., fat, protein, dry matter, total solids) |
| Calibration | Product-specific multivariate calibration models |
| Interface | 4× 4–20 mA analog I/O, 4× 24 V digital outputs, VARINLINE® process housing (inline version) |
| Compliance Context | Designed for GLP/GMP-aligned QA/QC environments |
| Brand | LUM GmbH |
|---|---|
| Origin | Germany |
| Model | LUMiSizer 65X |
| Measurement Principle | STEP-Technology (Spatial and Temporal Extinction Profiles) |
| Sample Throughput | 12 positions simultaneously |
| Temperature Control Range | 4 °C to 60 °C (±1 K) |
| Particle Size Range (ISO 13318-2) | 20 nm – 100 µm |
| Stability & Sedimentation Velocity Range | 10 nm – 1000 µm |
| Concentration Range | 0.00015 % – 90 % w/w |
| Density Range | up to 22 g/cm³ |
| Viscosity Range | 0.8 – 10⁸ mPa·s |
| Acceleration Force | 6 × g to 2300 × g |
| Optical Wavelength Options | 470 nm (blue) and 865 nm (NIR) |
| Sample Volume | 0.05 – 2.0 mL |
| Compliance | ISO/TR 13097, ISO 13318-2, FDA 21 CFR Part 11 (via SEPView software audit trail), GLP/GMP-ready |