Laboratory Services
Filter
Showing 61–90 of 179 results
| Brand | KHLAB |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Domestic (China) |
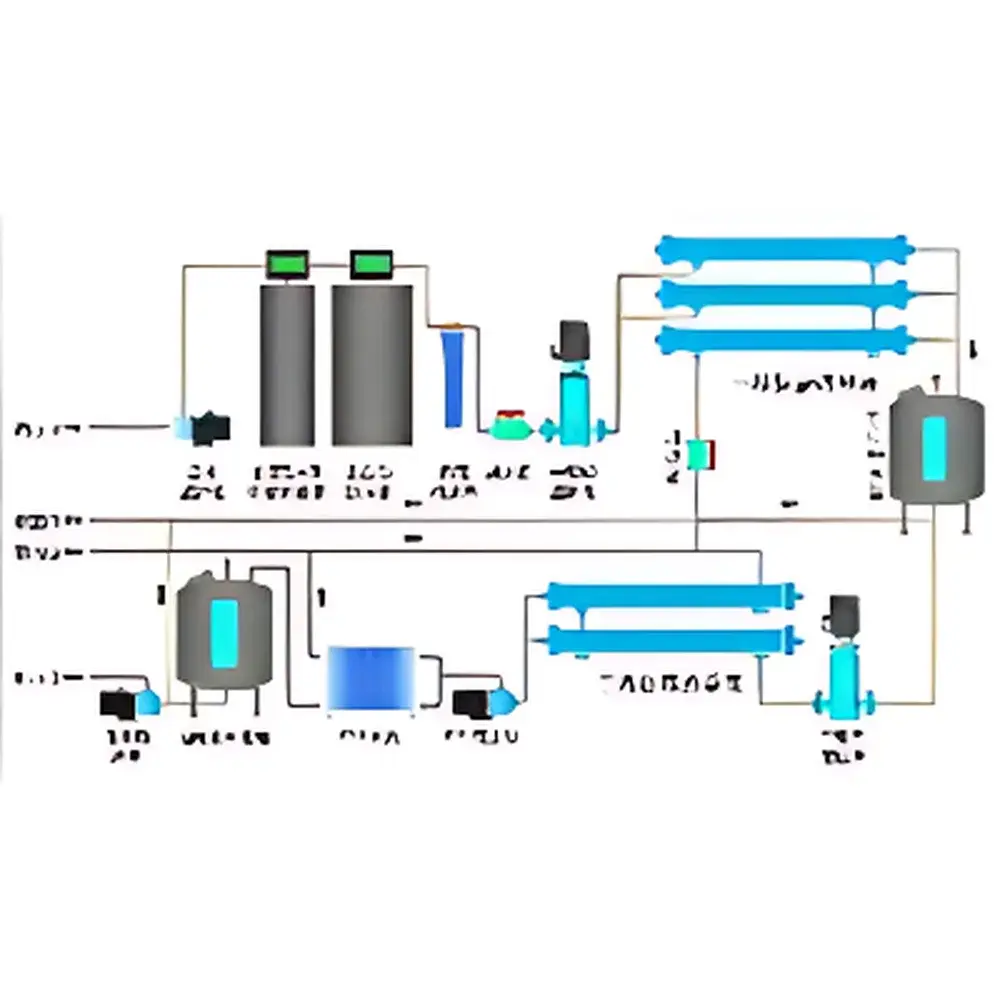
| Model | PIVAS (Pharmacy Intravenous Admixture Services) Facility |
| Quotation | Upon Request |
| Design Experience | 10+ Years |
| Brand | KHLAB |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | Precision Medicine Laboratory |
| Quotation | Upon Request |
| Design Experience | 10+ Years |
| Brand | Kingfar |
|---|---|
| Model | ErgoLAB |
| Type | Cloud-Based Laboratory Information Management System (LIMS) for Human Factors & Ergonomics Research |
| Deployment | SaaS |
| Compliance Framework | Supports GLP/GMP-aligned audit trails, ISO 9241-210 (Human-Centered Design), ASTM F2500 (VR/AR Human Factors), USP <1058> (Analytical Instrument Qualification), FDA 21 CFR Part 11 (Electronic Records & Signatures) |
| Data Synchronization Precision | Sub-millisecond (<1 ms) via hardware-triggered TTL and software-based LSL (Lab Streaming Layer) integration |
| Supported Signal Modalities | 35+ concurrent streams including EEG, fNIRS, eye-tracking, EDA/GSR, EMG, ECG, HRV, RESP, PPG, SpO₂, SKT, EOG, facial expression (AU-coded), motion capture, VR/AR interaction logs, web/mobile UI telemetry, environmental sensors (temp, humidity, noise, illuminance, PM2.5), and spatial trajectory data (indoor/outdoor/VR) |
| Brand | Kingfar |
|---|---|
| Origin | Imported |
| Manufacturer Type | Authorized Distributor |
| Pricing | Available Upon Request |
| Brand | Kyocera |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Domestic (China) |
| Model | JK-WB |
| Service Fee Range | USD 700–1,400 per incident (excl. parts) |
| Technician Experience | 10+ years |
| Instrument Categories Supported | Analytical Instruments (GC, LC, GC-MS, AAS, ICP-OES, UV-Vis, FTIR, Sample Prep Systems) |
| Response Time | Within 24 hours |
| Service Scope | Preventive Maintenance, Corrective Repair, Relocation, Calibration Support, Spare Parts Supply, Lifecycle Management |
| Compliance Framework | Aligned with ISO/IEC 17025:2017 laboratory competence requirements for service delivery |
| Brand | Chemlab Pro |
|---|---|
| Origin | Shanghai, China |
| Distributor Type | Authorized Regional Distributor |
| Product Origin | Domestic (China) |
| Model | LA-ICP-TOFMS Bioimaging Service |
| Quotation | Upon Request |
| Service Experience | 3–5 Years |
| Sample Types | Animal & Plant Tissue Sections, Fresh/Dried Plant Specimens |
| Turnaround Time | 5–10 Business Days |
| Sample Preparation Requirements | Cryosections or resin-embedded sections (OCT or epoxy resin acceptable) |
| minimum thickness | 10 µm |
| Sample Mounting | Fresh/dried plant samples must be flat-mounted on conductive glass or indium-tin oxide (ITO)-coated slides |
| max. dimensions | 9 cm × 9 cm × 0.8 cm |
| Certified Reference Materials | NIST SRM 1486 Bone Ash, GBW 07605 Spinach Leaves, GBW 07607 Citrus Leaves |
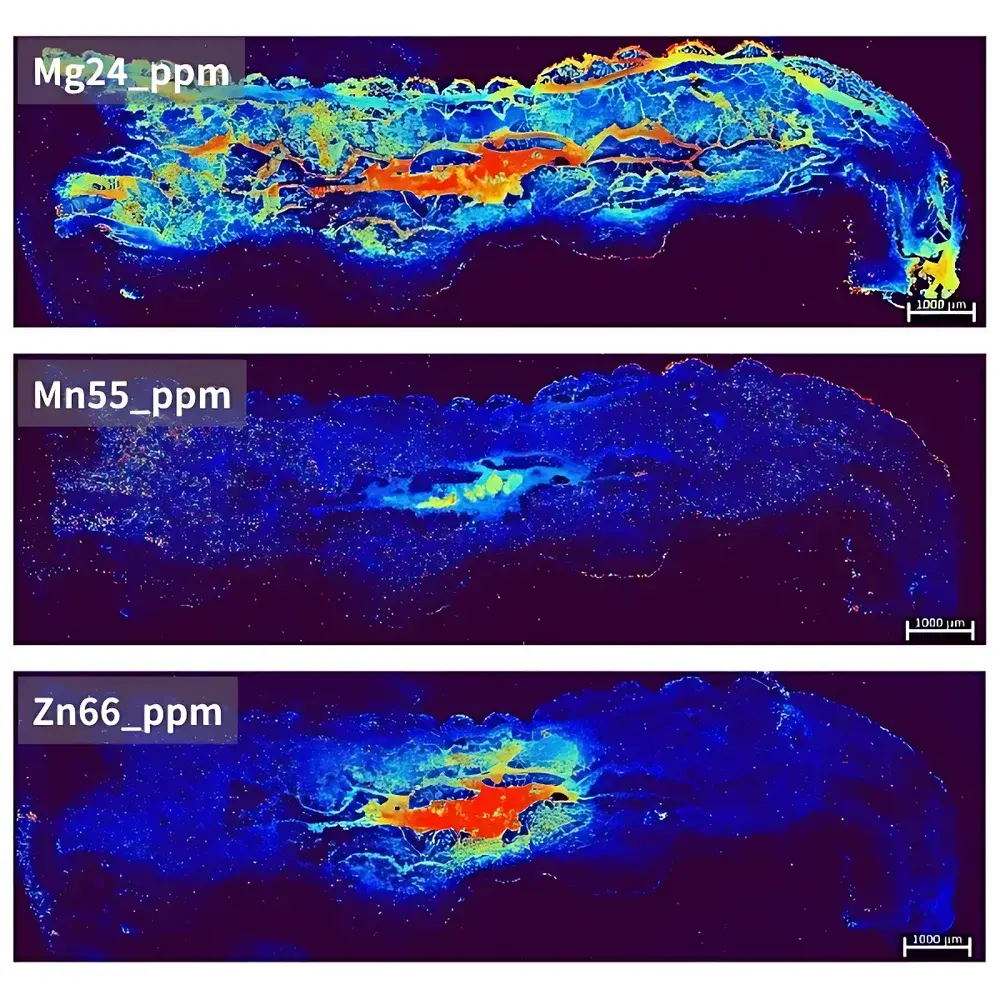
| Target Application | *Ophiocordyceps sinensis* (caterpillar fungus) elemental mapping of insect body |
| Spatial Resolution | 20 µm |
| Pixel Count | 187,500 |
| Imaging Area | 15 mm × 5 mm |
| Brand | Chemlab Pro |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Domestic |
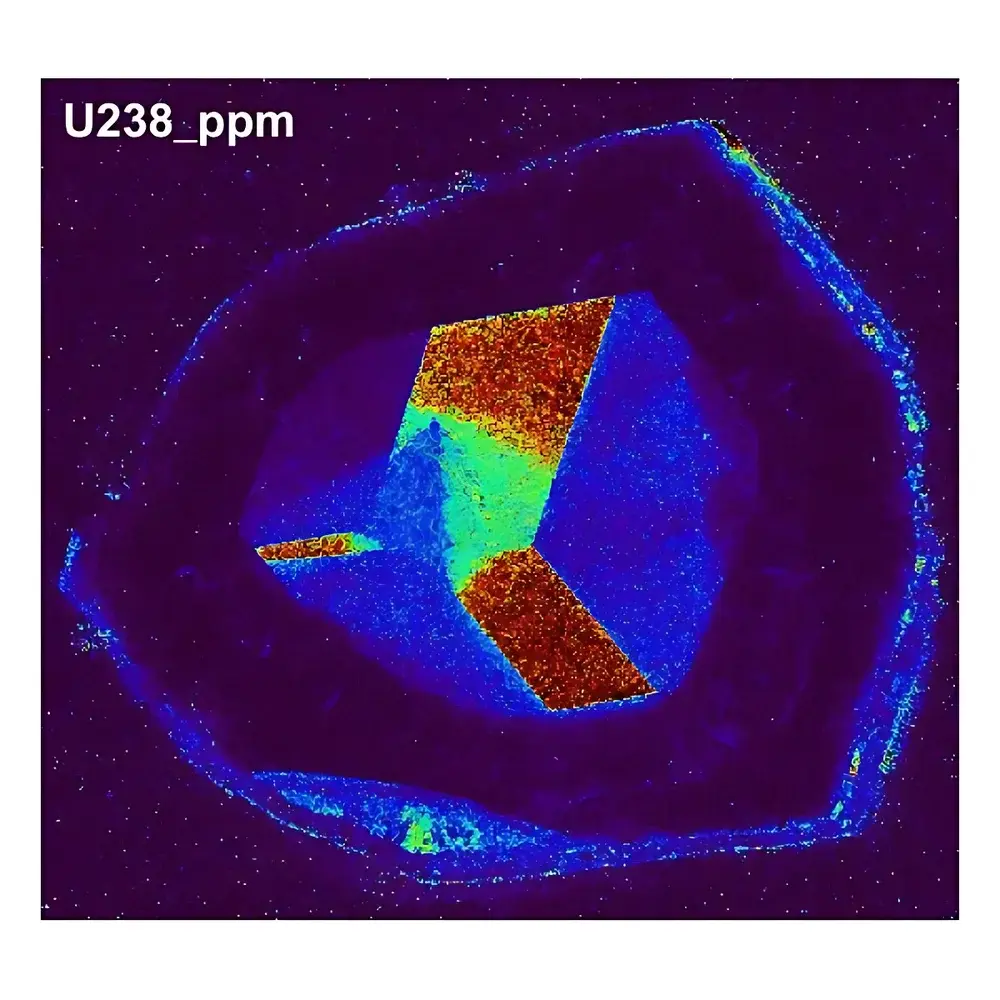
| Model | LA-ICP-TOFMS Elemental Imaging (Geological Application) |
| Quotation | Upon Request |
| Service Experience | 3–5 Years |
| Sample Dimensions | ≤9 cm × 9 cm × 2.5 cm |
| Surface Requirement | Flat polished surface preferred |
| Matrix-Matched Standards Available | For silicates and carbonates only |
| Quantitative Capability | Quantitative imaging only with matrix-matched reference materials |
| Spatial Resolution | 50 µm |
| Pixel Count | 137,000 |
| Imaging Area | 15.7 mm × 12.6 mm |
| Acceptance Criteria | Clear, artifact-free elemental distribution maps |
| Brand | Laballiance |
|---|---|
| Model | Laballiance |
| Origin | USA |
| Software Type | Chromatography Data Acquisition & Processing System |
| Compliance | Compatible with Microsoft Office Suite (Word, Excel, Access) |
| Deployment | Standalone Windows-based CDS workstation software |
| Regulatory Context | Designed for GLP-compliant laboratories |
| Brand | LabHawk |
|---|---|
| Origin | China |
| Manufacturer Type | Authorized Distributor |
| Model | LabHawk |
| Software Type | GxP-Compliant Excel Add-in for 21 CFR Part 11 |
| Regulatory Scope | FDA 21 CFR Part 11, Annex 11, ALCOA+ Principles, GMP/GLP Data Integrity Requirements |
| Deployment | On-premises or virtualized Windows environment (64-bit) |
| Supported Excel Versions | Microsoft Excel 2016–2021, Microsoft 365 (Desktop App) |
| User Authentication | LDAP/Active Directory integrated |
| Audit Trail | Immutable, time-stamped, user-attributed, exportable to CSV/PDF |
| Electronic Signature | PKI-based, role-enforced, with reason-for-change field (5–200 characters) |
| Version Control | Automatic versioning on save |
| Print Control | Centralized print logging |
| Search Capability | Full-text and metadata indexing across unlimited .xlsx/.xls files (local or network file shares) |
| Origin | Shanghai |
|---|---|
| Manufacturer Type | Distributor |
| Origin Category | Domestic |
| Model | E&I |
| Quotation | Upon Request |
| Service Fee Range | USD 7,200–14,400 (based on project scope) |
| Project Experience | 100+ completed installations |
| Brand | Agilent Technologies |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
| Model | Brand- and Model-Agnostic |
| Service Fee | USD 1,500–3,000 (custom quoted) |
| Relocation Experience | 10+ years |
| Response Time | Within 24 hours |
| Service Scope | Full-service laboratory instrument relocation |
| Coverage | Nationwide (U.S.) |
| Brand | Palon |
|---|---|
| Origin | Tianjin, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | Instrument Moving Service |
| Quotation Range | USD 1,400 – 11,200 (service fee: USD 1,400 – 2,800) |
| Relocation Experience | 10 years |
| Response Time | ≤ 48 hours |
| Sample Compatibility | Life Sciences Laboratories |
| Service Coverage | Tianjin-based operations and nationwide project coordination |
| Brand | LabTech |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Product Origin | Domestic (China) |
| Model | LabTech-1 |
| Pricing | Available Upon Request |
| Brand | LabTech |
|---|---|
| Model | LabTech-2 |
| Construction Materials | Polypropylene (PP), All-Steel, or Solid Wood |
| Dimensions (with exhaust) | 900 × 450 × 1800 mm |
| Origin | Beijing, China |
| Manufacturer | LabTech (China) Co., Ltd. |
| Compliance | Designed for general laboratory chemical storage per ISO 3696 and GB/T 27476 standards |
| Brand | LabTech |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | LabTech-9 |
| Pricing | Available Upon Request |
| Gas Distribution Architecture | Centralized, Zoned (Toxic/Flammable/Inert Separation) |
| Ventilation | 24/7 Intermittent + Emergency Exhaust |
| Pressure Regulation | Medium-Pressure Primary Supply with Secondary Pressure Reduction |
| Safety Systems | Low-Pressure Alarm, Gas Leak Detection (Toxic/Flammable Gases), Interlocked Emergency Exhaust Activation |
| Brand | LabTech |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | Domestic (China) |
| Model | LabTech-10 |
| Pricing | Available Upon Request |
| Pipe Material | PVDF/CPVC |
| Main Loop Velocity | 1.5–3.0 m/s |
| Branch Loop Velocity | 1.0–2.0 m/s |
| Recirculation Ratio | 30–50% of Design Flow Rate |
| Compliance Focus | Low Extractables, Smooth Internal Surface, Air-Tight Integrity |
| Brand | LabTech |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | Domestic (China) |
| Model | LabTech-3 |
| Pricing | Available Upon Request |
| Brand | LabTech |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Manufacturer |
| Origin Category | Domestic |
| Model | LabTech-4 |
| Pricing | Upon Request |
| Brand | LabTech |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Manufacturer |
| Country of Origin | Domestic (China) |
| Model | LabTech-5 |
| Pricing | Upon Request |
| Brand | LabTech |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Manufacturer |
| Product Origin | Domestic (China) |
| Model | LabTech-6 |
| Pricing | Upon Request |
| Brand | LabTech |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Product Origin | Domestic (China) |
| Model | LabTech-7 |
| Pricing | Available Upon Request |
| Brand | LabTech |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Pricing | Upon Consultation |
| Origin | Beijing |
|---|---|
| Manufacturer Type | Distributor |
| Origin Category | Domestic (China) |
| Model | Image Analysis V3.0 |
| Price Range | USD 0–70 (Training-Only Engagement) |
| Training Experience | 15+ Years |
| Instrument Type | Software-Based Image Analysis Platform |
| Brand | Lihero |
|---|---|
| Origin | Hunan, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | Pollution Source Online Monitoring Center Management System |
| Pricing | Available Upon Request |
| Brand | Metrohm |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Manufacturer |
| Product Category | Domestic |
| Model | Cloud Academy |
| Pricing | Upon Request |
| Brand | Metrohm |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Domestic |
| Service Type | Instrument Certification |
| Scope | Installation Qualification (IQ) and Operational Qualification (OQ) |
| Documentation | Fully traceable digital IQ/OQ protocols compliant with GxP and 21 CFR Part 11 |
| Delivery Model | On-site and remote software validation supported |
| Lifecycle Coverage | DQ, IQ, OQ, PV, PQ, periodic requalification, and decommissioning support |
| Brand | Metrohm |
|---|---|
| Origin | Switzerland |
| Manufacturer | Yes |
| Import Status | Imported |
| Model | eTCC |
| Service Fee Range | USD 14,000–28,000/year (approx.) |
| Field Experience | 10+ years |
| Instrument Category | Analytical Instruments |
| Response Time | ≤6 hours |
| Scope of Service | Comprehensive maintenance, calibration, verification, and consumables support for all Metrohm analytical instruments |
| Service Hub | Beijing, China |
| Brand | Metrohm |
|---|---|
| Origin | Switzerland |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Laboratory Instrument Maintenance & Support Service |
| Model | MSA |
| Service Fee Range | USD 1,200–2,400/year (approx.) |
| Field Experience | >10 years |
| Response Time | 4–8 hours |
| Scope of Service | Preventive maintenance, performance verification, corrective repair, operator training, application support, calibration verification, and spare parts logistics |
| Coverage | Nationwide (China) |
| Brand | METTLER TOLEDO |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Domestic (China) |
| Model | ACC |
| Service Type | Instrument Calibration |
| Application Scope | Balances and Scales |
| Experience | 1–10 Years |
| Response Time | 24–48 Hours |
| Calibration Scope | Full-range performance evaluation including measurement uncertainty quantification |
| Regulatory Alignment | Supports ISO/IEC 17025, USP <41>, <1251>, ASTM E898, and FDA 21 CFR Part 11 audit-trail requirements for documented calibration evidence |
| Brand | METTLER TOLEDO |
|---|---|
| Origin | Switzerland |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | Digital Training Course |
| Topic | Calibration vs. Adjustment for Tank Weighing Systems |
| Instructor Experience | >10 Years |
| Equipment Type | Laboratory & Industrial Weighing Systems |
| Compliance Context | ASTM E74, ISO/IEC 17025, USP <41>, FDA 21 CFR Part 11 (Data Integrity in Training Records) |