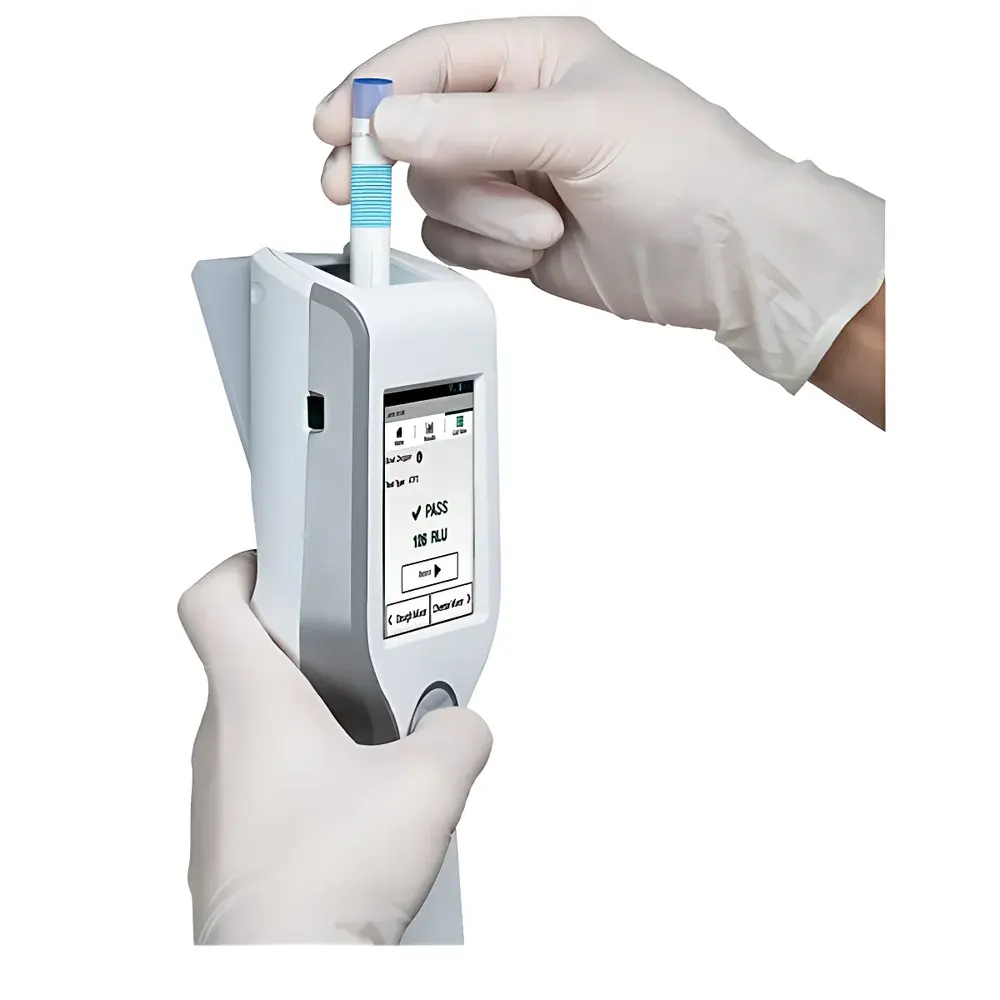
3M Clean-Trace™ ATP Bioluminescence Detector
| Brand | 3M |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | Clean-Trace™ |
| Pricing | Upon Request |
Overview
The 3M Clean-Trace™ ATP Bioluminescence Detector is a handheld, field-deployable luminometer engineered for rapid, quantitative assessment of surface and liquid hygiene in food production, pharmaceutical manufacturing, healthcare, and institutional environments. It operates on the principle of bioluminescent enzymatic reaction: adenosine triphosphate (ATP) extracted from residual organic matter reacts with recombinant firefly luciferase and its substrate luciferin, producing light proportional to ATP concentration. This photonic signal is measured in relative light units (RLUs) and converted into actionable microbial cleanliness indicators—enabling real-time verification of cleaning efficacy without microbiological culturing. Designed for routine environmental monitoring programs, the system supports preventive control strategies aligned with HACCP, ISO 22000, SQF Code Edition 9, and BRCGS Food Safety Issue 9. Its compact architecture, low power consumption, and ruggedized housing meet IP54 ingress protection standards for use in wet, chilled, or high-traffic processing zones.
Key Features
- Single-handed, ergonomic operation with intuitive tactile interface and backlit LCD display for low-light environments
- Optimized optical path with thermally stabilized photomultiplier tube (PMT) ensuring <±5% RLU repeatability across 0–99,999 RLU range
- Integrated Bluetooth 5.0 and Ethernet connectivity for secure, audit-ready wireless data transmission to local networks or cloud-based platforms
- Battery life of ≥8 hours per charge (rechargeable Li-ion), supporting >1,000 consecutive measurements without recalibration
- Auto-zeroing and internal calibration verification before each measurement cycle to maintain traceability per ISO/IEC 17025 requirements
- On-device pass/fail threshold configuration with customizable alert logic (e.g., color-coded LED feedback, audible tone)
Sample Compatibility & Compliance
The Clean-Trace™ system is validated for use with three proprietary swab formats: Clean-Trace™ Surface Sampling Swabs (ATP UXL100), Enhanced Water Sampling Swabs – Total ATP (AQT200), and Free ATP Water Sampling Swabs (AQF100). Each swab incorporates optimized lysis chemistry and buffer stabilization to maximize ATP recovery from diverse substrates—including stainless steel, polypropylene, conveyor belts, gaskets, and membrane filters—while minimizing interference from sanitizers (e.g., quaternary ammonium compounds, chlorine dioxide). All reagents are manufactured under ISO 13485-certified processes and comply with FDA 21 CFR Part 11 for electronic records and signatures when used with validated software configurations. The platform meets EN 13697:2015 for chemical disinfectant efficacy testing and supports GLP/GMP documentation workflows through time-stamped, user-authenticated result logging.
Software & Data Management
The Clean-Trace™ Hygiene Monitoring Management Software (v4.x) provides a validated, role-based desktop and web application for comprehensive data governance. It features an ISO 27001-aligned security framework with AES-256 encryption, multi-factor authentication, and full 21 CFR Part 11 compliance—including electronic signatures, audit trails with immutable timestamps, and configurable retention policies. The dashboard enables dynamic trend analysis via SPC charts (X-bar/R, CUSUM), root cause correlation mapping across zones/time shifts, and automated PDF report generation compliant with FSMA Preventive Controls requirements. Data export supports CSV, XML, and direct integration with LIMS or MES systems via RESTful API. Language support includes 23 localized UIs, including English, Spanish, French, German, Mandarin, Japanese, and Arabic—facilitating global deployment consistency.
Applications
- Routine post-cleaning verification of food contact surfaces (conveyors, mixers, fillers, slicers) prior to startup
- Validation of CIP/SIP cycles in dairy, beverage, and biopharma facilities
- Monitoring of water quality in rinse lines, boiler feedwater, and clean-in-place return loops
- Environmental monitoring in ready-to-eat (RTE) production zones and sterile compounding areas
- Training and competency assessment for sanitation teams using objective, quantitative metrics
- Supporting third-party audits (e.g., FDA inspections, GFSI-benchmarked certifications) with retrievable, timestamped digital records
FAQ
Does the Clean-Trace™ system require daily calibration?
No—each measurement cycle includes automatic optical zeroing and internal reference verification; formal calibration is required only annually or after instrument service, per 3M’s Certificate of Conformance.
Can RLU values be directly correlated to CFU counts?
No—ATP detection measures total bioburden-associated energy molecules, not viable microorganisms; it serves as a rapid proxy for cleaning effectiveness, not a replacement for microbiological enumeration methods.
Is the software compatible with existing enterprise IT infrastructure?
Yes—the application supports Windows Server 2016+, SQL Server 2019+, and integrates with Active Directory for centralized user management and group policy enforcement.
What is the minimum detectable ATP concentration?
The system reliably detects ≥1 femtomole (10⁻¹⁵ mol) of ATP per swab, equivalent to approximately 1,000 bacterial cells under standardized recovery conditions.
How does temperature variation affect measurement stability?
An integrated thermal sensor compensates for ambient fluctuations between 5°C and 40°C; RLU output remains within ±3% deviation across this operational range.