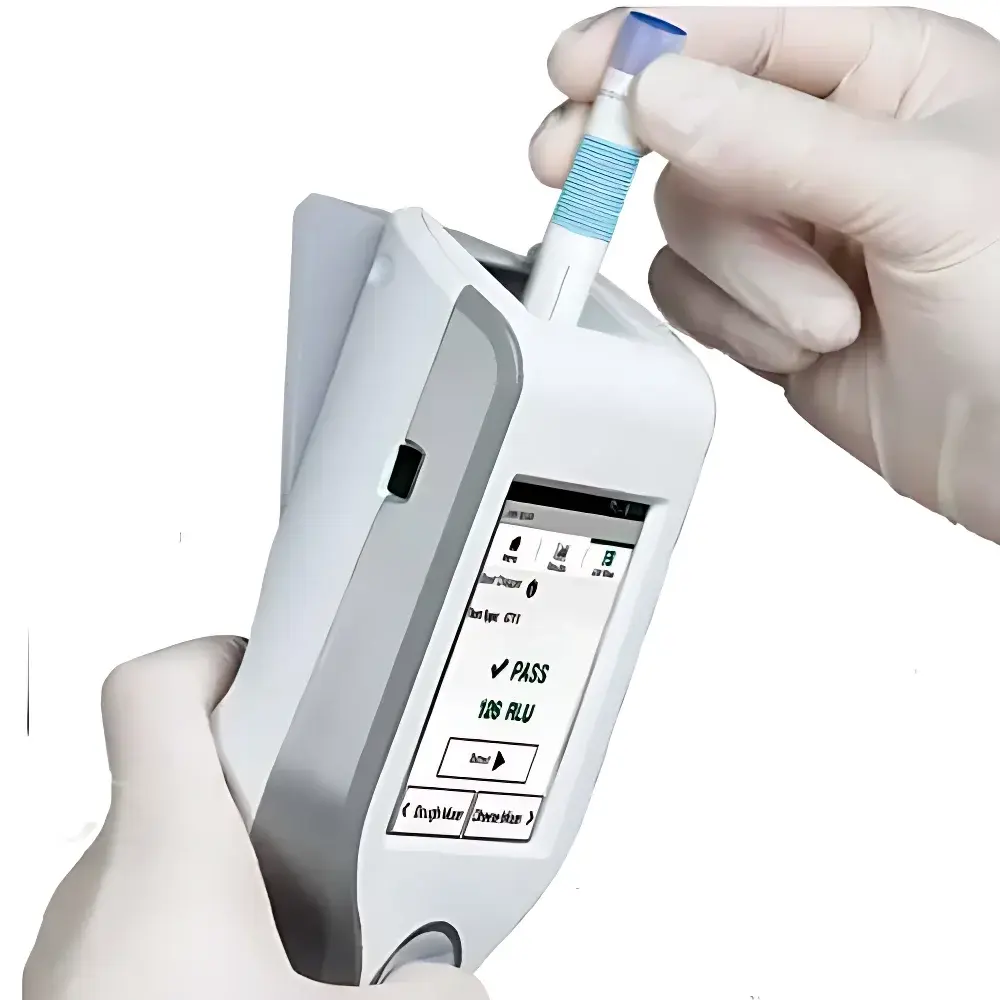
3M Clean-Trace ATP LM1 Handheld Bioluminescence Luminometer
| Brand | 3M |
|---|---|
| Origin | USA |
| Model | Clean-Trace ATP LM1 |
| Detection Principle | ATP-dependent bioluminescence assay |
| Sample Types | Surface swabs (UXL100), water samples (AQT200, AQF100) |
| Data Connectivity | Wireless (Bluetooth/Wi-Fi enabled) |
| Software Platform | 3M Clean-Trace Hygiene Management Software |
| Compliance Support | Designed for use in environments requiring adherence to HACCP, ISO 22000, FDA Food Code, and GMP-aligned hygiene verification protocols |
| Power | Rechargeable battery with AC adapter |
| Portability | Handheld, ergonomic design for single-hand operation |
Overview
The 3M Clean-Trace ATP LM1 Handheld Bioluminescence Luminometer is an engineered solution for rapid, quantitative hygiene verification across food production, retail, healthcare, and hospitality environments. It operates on the well-established principle of ATP-dependent bioluminescence: adenosine triphosphate (ATP), a universal intracellular energy molecule present in all viable microorganisms—including bacteria, yeasts, and molds—is extracted from environmental surfaces or liquid samples using proprietary 3M Clean-Trace sampling devices. Upon reaction with recombinant firefly luciferase and its substrate D-luciferin, ATP catalyzes light emission at ~560 nm. The LM1 precisely measures this photon output (reported in Relative Light Units—RLUs) using a calibrated photomultiplier tube (PMT) detector. RLU values correlate directly with total biological load, enabling objective, real-time assessment of cleaning efficacy and residual contamination risk—without culturing or incubation.
Key Features
- Intuitive handheld form factor with one-handed operation and tactile button layout optimized for gloved use in production settings.
- Integrated rechargeable lithium-ion battery supporting >8 hours of continuous operation; includes AC charging dock and USB-C data sync capability.
- High-sensitivity PMT-based optical detection system calibrated per ISO 22000–aligned verification protocols.
- On-device pass/fail threshold indicators with configurable alert levels aligned to facility-specific hygiene standards (e.g., <100 RLU for critical zones).
- Preloaded firmware supporting automatic firmware updates via secure over-the-air (OTA) delivery through the Clean-Trace software ecosystem.
- Ruggedized housing rated IP54 for dust and splash resistance, validated for operation in refrigerated, humid, or temperature-controlled food processing areas.
Sample Compatibility & Compliance
The LM1 is validated exclusively with 3M’s CE-marked, FDA-registered Clean-Trace sampling consumables: UXL100 surface swabs for non-porous and textured surfaces; AQT200 total ATP water swabs; and AQF100 free ATP water swabs. These devices feature controlled lysis chemistry and standardized extraction volumes to ensure reproducible ATP recovery (>92% mean recovery rate per internal 3M validation studies). The system supports audit-ready documentation required under FDA 21 CFR Part 11 (when used with Clean-Trace software’s electronic signature and audit trail functions), HACCP prerequisite program verification, and ISO 22000 Clause 8.2 hygiene monitoring requirements. All consumables are manufactured under ISO 13485-certified processes and supplied with lot-specific CoA documentation.
Software & Data Management
The 3M Clean-Trace Hygiene Management Software (v5.2+) serves as the central analytical and compliance hub. Its re-engineered dashboard enables dynamic visualization of RLU trends across shifts, zones, equipment IDs, and operator groups. Automated report generation supports PDF/Excel export with embedded metadata (timestamp, GPS coordinates if enabled, user ID, calibration status). Wireless synchronization (via Bluetooth 5.0 or 2.4 GHz Wi-Fi) eliminates manual data entry errors and reduces reporting latency to <60 seconds post-measurement. Audit trails log every action—including result edits, threshold changes, and user logins—with immutable timestamps compliant with GLP/GMP record retention policies. Role-based access control (RBAC) ensures data integrity across multi-site deployments.
Applications
- Post-clean verification of food contact surfaces (conveyors, mixers, slicers, packaging lines) prior to startup.
- Validation of sanitation procedures during SSOP (Sanitation Standard Operating Procedure) audits.
- Root-cause analysis of microbial excursions via spatial RLU mapping across production zones.
- Training verification: objective measurement of staff swabbing technique consistency and coverage accuracy.
- Water system hygiene monitoring in CIP rinse cycles, ice machines, and potable water dispensers.
- Supplier qualification support through standardized, instrument-based hygiene scoring against contractual KPIs.
FAQ
What is the detection limit of the LM1 in femtomoles of ATP?
The LM1 achieves a functional detection limit of ≤10 fmol ATP per sample when used with UXL100 swabs under standard extraction conditions, consistent with AOAC PTM-01-2017 performance criteria.
Does the LM1 require daily calibration?
No. The device performs self-diagnostic checks at power-on and uses factory-calibrated optical components traceable to NIST standards; annual verification with 3M-provided reference standards is recommended.
Can the LM1 store results offline?
Yes. Internal memory retains ≥10,000 test records with full metadata; synchronization occurs automatically upon wireless reconnection.
Is the Clean-Trace software compatible with LIMS integration?
Yes. RESTful API and HL7-compliant export modules enable bidirectional data exchange with major laboratory information management systems.
Are replacement swabs supplied sterile?
UXL100 and AQT200 swabs are non-sterile but individually sealed and validated for ambient stability; AQF100 swabs are gamma-irradiated and supplied sterile for aseptic water testing applications.