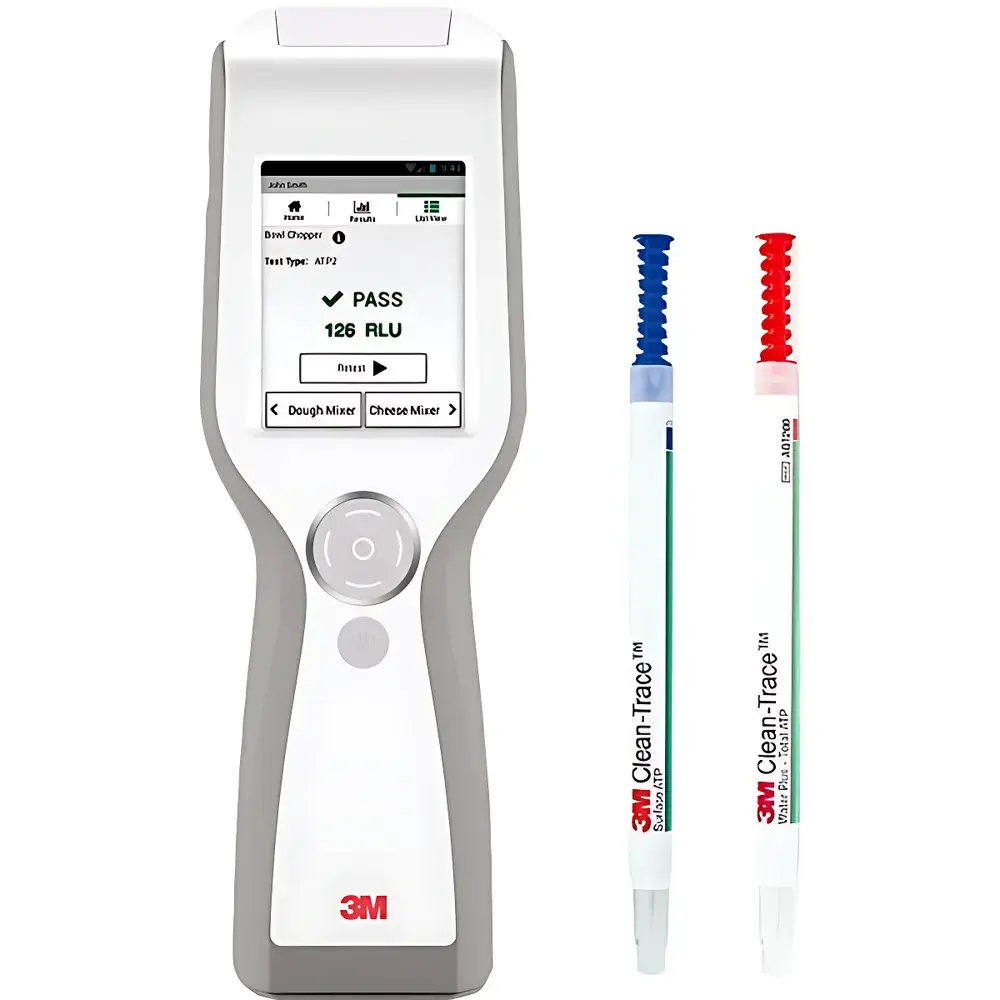
3M Clean-Trace LM1 ATP Bioluminescence Detector
| Brand | 3M |
|---|---|
| Origin | USA |
| Model | LM1 |
| Detection Principle | ATP-dependent luciferase bioluminescence assay |
| Sample Types | Surface swabs (UXL100), free ATP in water (AQF100), total ATP in water (AQT200) |
| Software | Clean-Trace Hygiene Management Software with wireless connectivity, automated reporting, and audit-ready data logging |
| Compliance Context | Designed for use in environments requiring adherence to HACCP, ISO 22000, FDA Food Safety Modernization Act (FSMA), and GMP-aligned hygiene verification protocols |
Overview
The 3M Clean-Trace LM1 ATP Bioluminescence Detector is a portable, field-deployable luminometer engineered for rapid, quantitative assessment of surface and liquid hygiene status in food production, pharmaceutical manufacturing, healthcare facilities, and environmental monitoring settings. It operates on the well-established principle of ATP-dependent bioluminescence: adenosine triphosphate (ATP), a universal intracellular energy molecule present in all viable microorganisms—including bacteria, yeasts, and molds—is extracted from sampled surfaces or aqueous matrices and reacted with recombinant firefly luciferase and its substrate D-luciferin. This enzymatic reaction emits photons proportional to ATP concentration, which the LM1’s calibrated photomultiplier tube (PMT) detects with high sensitivity and linearity across a dynamic range spanning 0–99,999 relative light units (RLUs). Unlike culture-based methods requiring 24–72 hours, the LM1 delivers objective, instrument-based hygiene verification results within seconds—enabling real-time decision-making at critical control points (CCPs) in HACCP plans and facilitating immediate corrective actions.
Key Features
- Compact, ergonomic handheld design with integrated rechargeable battery—optimized for single-handed operation in confined or elevated spaces.
- Dedicated optical path and temperature-stabilized PMT sensor ensuring consistent RLU measurement reproducibility (<5% CV between replicates under controlled conditions).
- Pre-calibrated detection system compliant with ASTM E2631–22 “Standard Practice for Determining the Relative Light Unit Response of ATP Bioluminescence Assays”.
- Intuitive touchscreen interface with multilingual support (English, Spanish, French, German, Chinese) and context-sensitive prompts for step-by-step assay execution.
- On-device data storage for up to 10,000 test records, including timestamp, operator ID, location tag, and RLU value—retained even during power loss.
- Integrated Bluetooth 5.0 and optional Wi-Fi module enabling secure, encrypted wireless synchronization with Clean-Trace Hygiene Management Software without physical cable dependency.
Sample Compatibility & Compliance
The LM1 supports three validated sampling formats: Clean-Trace UXL100 surface swabs for non-porous and textured food contact surfaces; AQF100 liquid swabs targeting free ATP in process water, rinse water, or CIP return lines; and AQT200 swabs quantifying total ATP (free + cellular-bound) in wastewater, cooling tower water, or post-sanitization effluent. All swab chemistries are manufactured under ISO 13485-certified conditions and undergo lot-specific bioluminescence response validation. The system architecture supports 21 CFR Part 11-compliant electronic signatures and full audit trails when deployed with Clean-Trace software in regulated environments. Data outputs meet requirements for internal hygiene verification per ISO 22000:2018 Clause 8.2.3 and external third-party audits under BRCGS Food Safety Issue 9 and SQF Edition 9.
Software & Data Management
Clean-Trace Hygiene Management Software serves as the central analytical and compliance hub. Its re-engineered dashboard provides role-based access to real-time RLU heatmaps, trend analysis over time (daily/weekly/monthly), statistical process control (SPC) charts, and automated threshold alerts (e.g., “Caution” at 100 RLUs, “Action Required” at 300 RLUs per 3M’s validated thresholds). Reports are exportable in PDF, CSV, and XML formats—with embedded metadata required for GLP/GMP documentation. Wireless synchronization eliminates manual transcription errors and ensures traceability from instrument to enterprise quality management system (QMS). Version-controlled software updates are delivered via secure HTTPS channels and include change logs aligned with ICH Q9 risk management principles.
Applications
The LM1 is routinely deployed across hygienically sensitive operational zones: pre-operational verification of equipment after cleaning-in-place (CIP) cycles in dairy processing plants; post-maintenance validation of HVAC ductwork in sterile pharmaceutical cleanrooms; routine monitoring of conveyor belts, slicers, and packaging stations in ready-to-eat (RTE) meat facilities; and rapid screening of potable water distribution systems in municipal utilities. Its portability and ruggedized IP54-rated enclosure make it suitable for field use in USDA-inspected slaughterhouses, FDA-regulated contract manufacturers, and WHO-prequalified vaccine production sites where mobility and regulatory defensibility are equally critical.
FAQ
What does an RLU reading represent, and how is it interpreted?
RLU (Relative Light Unit) is a vendor-specific, instrument-normalized measure of photon emission intensity. It is not an absolute ATP concentration but correlates linearly with ATP mass (femtomoles) within the validated assay range. Interpretation requires site-specific action limits established through correlation studies with microbiological plating data.
Can the LM1 differentiate between microbial ATP and non-microbial ATP sources?
No—the assay detects total ATP. However, Clean-Trace swab formulations include proprietary inhibitors that suppress background interference from plant tissue, food residues, and cleaning chemicals, improving specificity for microbial contamination.
Is calibration required before each use?
The LM1 performs automatic self-calibration upon startup using an internal reference LED. No user-performed calibration is needed; annual factory recalibration is recommended per ISO/IEC 17025 guidelines.
How is data integrity ensured during wireless transmission?
All wireless transfers employ TLS 1.2 encryption and require authenticated session initiation. Each transmitted record includes digital hash verification to prevent tampering or packet corruption.
Does the system support integration with LIMS or MES platforms?
Yes—via Clean-Trace software’s configurable RESTful API and HL7-compatible data export modules, enabling bidirectional synchronization with laboratory information management systems (LIMS) and manufacturing execution systems (MES) without custom middleware.