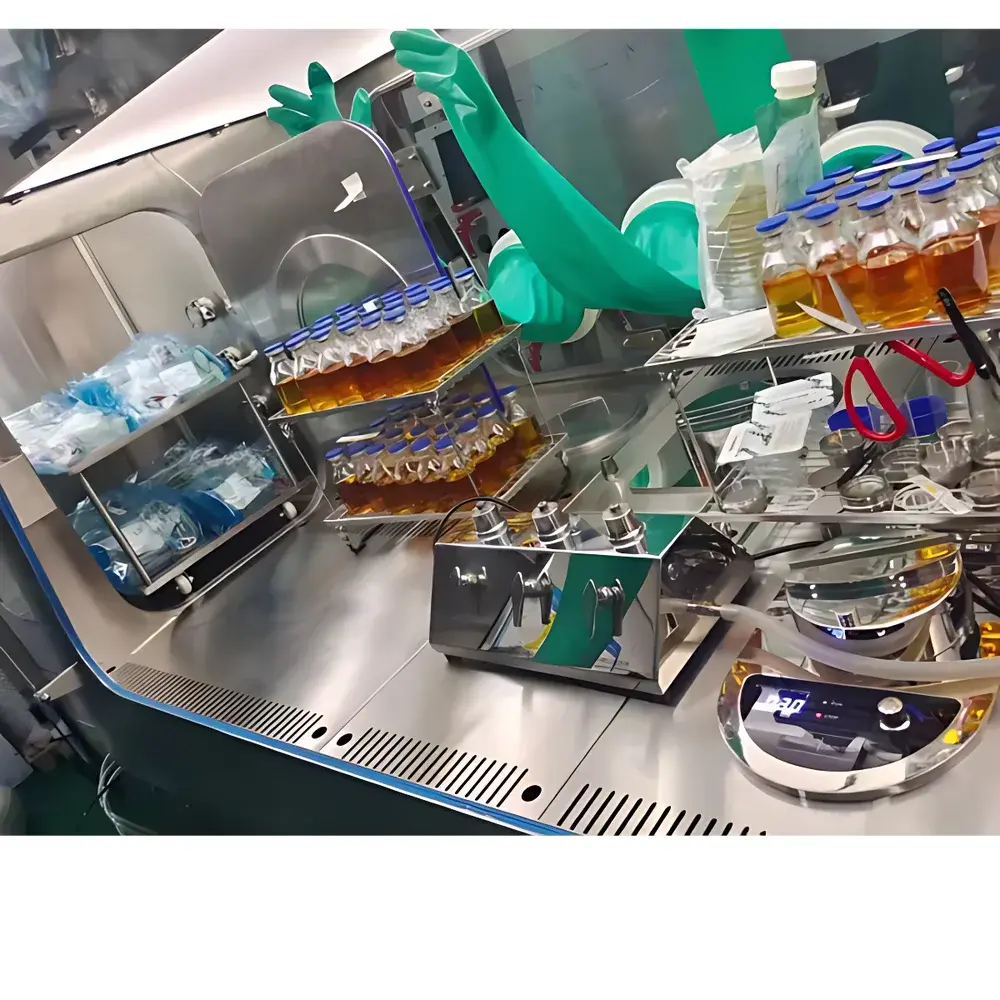
AA OPTO Electronic MIS1800C Aseptic Lyophilization & Sterility Testing Isolator
| Brand | AA OPTO Electronic |
|---|---|
| Origin | China |
| Manufacturer Type | Authorized Distributor |
| Model | MIS1800C |
| Pricing | Upon Request |
| Airflow Design | Positive-Pressure Vertical Unidirectional Laminar Flow |
| H₂O₂ Decontamination | Dry Vaporized Hydrogen Peroxide (VHP®-compatible flash-vaporization) |
| Leak Rate | ≤0.5 % vol/hr (per ISO 10648-2 Class 1) |
| Compliance | Meets requirements of Chinese Pharmacopoeia (ChP), EU GMP Annex 1 (2022), USP <1211>, and supports 21 CFR Part 11 electronic record/signature functionality |
| Integration | Supports real-time particle counters (0.5/5.0 µm), active air samplers, and environmental monitoring data logging via embedded HMI touchscreen |
Overview
The AA OPTO Electronic MIS1800C Aseptic Lyophilization & Sterility Testing Isolator is an ISO Class 5 (Grade A) compliant, hard-wall isolator engineered for high-integrity aseptic processing in regulated pharmaceutical and biotechnology environments. It operates on the principle of positive-pressure vertical unidirectional laminar airflow—validated to meet the airflow uniformity and velocity specifications outlined in the Chinese Pharmacopoeia (ChP), EU GMP Annex 1 (2022 revision), and ISO 14644-3. The isolator’s chamber maintains a consistent ≥0.45 m/s downward airflow across the entire work surface, minimizing turbulence and ensuring effective removal of airborne particulates and microorganisms. Its structural integrity is verified through quantitative leakage testing per ISO 10648-2 Class 1, achieving a maximum allowable leak rate of ≤0.5 % vol/hr—critical for sustaining long-term sterile integrity during lyophilization cycle interventions, sterility test manipulations, media fill simulations, and aseptic formulation transfers.
Key Features
- Integrated dry vaporized hydrogen peroxide (VHP®-equivalent) decontamination system utilizing flash-vaporization technology—enabling rapid phase transition from liquid H₂O₂ to saturated vapor without condensation, ensuring full penetration of HEPA/ULPA filters and achieving ≥6-log reduction of Geobacillus stearothermophilus biological indicators.
- Rigid stainless steel 316L chamber with electropolished internal surfaces and welded seam construction, minimizing crevices and facilitating cleaning validation and residue assessment per ICH Q5A and PDA TR #75.
- Touchscreen-based Human-Machine Interface (HMI) with audit-trail-enabled control logic compliant with GAMP 5 Category 4 software classification and FDA 21 CFR Part 11 requirements—including user role-based access, electronic signatures, and immutable event logging.
- Modular integration architecture supporting real-time connection to ISO 21501-4-compliant particle counters (0.5 µm and 5.0 µm channels), active microbial air samplers (e.g., MAS-100), and surface contact plates—all synchronized with process timestamps and exportable in CSV/PDF formats.
- Redundant pressure cascade design with differential pressure sensors (±0.1 Pa resolution) and automatic damper control to maintain stable overpressure gradients between isolator interior, anteroom, and surrounding cleanroom (typically +60 Pa / +30 Pa / +15 Pa).
Sample Compatibility & Compliance
The MIS1800C accommodates standard aseptic process interventions including vial loading/unloading for lyophilization cycles, membrane filtration sterility testing (per USP ), preparation of growth media and controls, and aseptic assembly of filtration trains. All materials in contact with product or critical surfaces comply with USP Class VI biocompatibility and ISO 10993-5 cytotoxicity standards. The isolator’s qualification documentation package supports IQ/OQ/PQ execution aligned with ASTM E2500-13 and EU Annex 15. It is designed to facilitate compliance with regulatory expectations for closed-system processing under FDA Guidance for Industry: Sterile Drug Products Produced by Aseptic Processing (2004), MHRA’s “Aseptic Processing – A Practical Guide” (2021), and WHO Technical Report Series No. 961, Annex 2.
Software & Data Management
The embedded control system runs on a deterministic real-time OS with dual SD-card redundancy for firmware and database storage. All operational parameters—including pressure differentials, H₂O₂ concentration (ppm), relative humidity, temperature, UV lamp status, and door interlock events—are recorded at 1-second intervals. Data archives are encrypted using AES-256 and support automated backup to networked NAS or FTPS servers. Reports—including decontamination cycle summaries, environmental monitoring trend charts, and alarm history logs—can be generated on-demand or scheduled, with configurable templates meeting ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available). Full electronic signature workflows include biometric or token-based authentication options.
Applications
- Sterility testing of final drug products and raw materials per USP , Ph. Eur. 2.6.1, and JP 4.07
- Aseptic intervention during freeze-drying cycles—including stopper seating, vial capping, and shelf temperature mapping probe insertion
- Media fill simulation studies under worst-case conditions (e.g., extended glove change, multiple interventions)
- Preparation and handling of low-bioburden excipients and sterile APIs prior to aseptic blending
- Isolated microbiological assay setup—including pour plates, spread plates, and membrane filtration assemblies
FAQ
Does the MIS1800C support automated cycle development for VHP decontamination?
Yes—the system includes a programmable VHP protocol editor allowing users to define multi-stage cycles (conditioning, injection, dwell, aeration) with adjustable setpoints for concentration, RH, and temperature. Optional cycle development services include mapping studies using chemical indicators and biological challenge testing per ISO 14644-3 and PDA TR #1.
Can the isolator be integrated into a facility’s MES or SCADA infrastructure?
Yes—via OPC UA (IEC 62541) or Modbus TCP interfaces, enabling bidirectional communication with manufacturing execution systems for batch record linkage, alarm forwarding, and centralized KPI dashboards.
What validation support is provided post-installation?
AA OPTO Electronic provides comprehensive qualification documentation (URS, FDS, SDS, FAT/SAT protocols), as well as on-site IQ/OQ execution support by qualified validation engineers holding ASQ CQE and ISPE GAMP certifications.