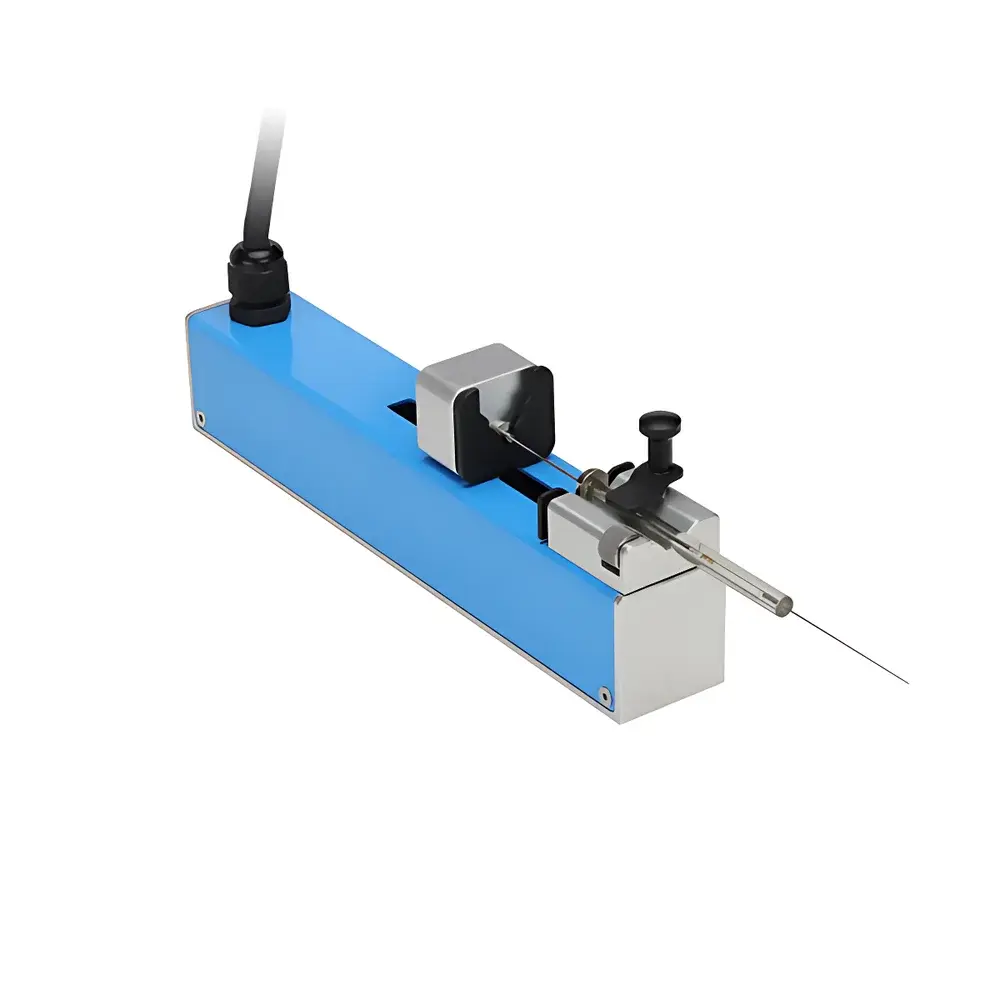
Acmer DSC-B01/W70B Precision Split-Body Syringe Pump
| Brand | Acmer |
|---|---|
| Model | DSC-B01/W70B |
| Type | Micro-syringe Pump |
| Flow Rate Range | 1 pL/min to 44.11 mL/min |
| Accuracy | ±0.5% |
| Repeatability | ±0.05% |
| Display | 5-inch HD Touchscreen |
| Control Interface | RS485 / USB (configurable address & baud rate) |
| External Trigger | TTL-compatible digital input (5–24 V or dry contact) |
| Status Outputs | Run/Stop, Direction signals |
| Syringe Compatibility | 1–10 mL plastic syringes and µL-range glass syringes |
| Force Adjustment | Programmable push force based on syringe barrel diameter and material |
Overview
The Acmer DSC-B01/W70B Precision Split-Body Syringe Pump is an engineered solution for high-fidelity fluid delivery in analytical, pharmaceutical, and microfluidic applications requiring nanoliter-to-milliliter volumetric control. Its split-body architecture separates the motor-driven actuator from the syringe mounting module—enabling flexible integration into custom rigs, glove boxes, or automated workstations while minimizing mechanical coupling and thermal drift. The pump operates on a precision lead-screw linear actuation principle, translating rotational motion into highly repeatable axial displacement of the syringe plunger. Designed for continuous, low-pulsation infusion and bidirectional operation (infuse/withdraw), it supports both time-critical dosing protocols and long-duration gradient elution workflows. Its flow range spans six orders of magnitude—from picoliter-per-minute ultra-low-flow regimes essential for single-cell electrophysiology or nanofluidic synthesis, up to 44.11 mL/min for rapid reagent priming or column equilibration—without hardware modification.
Key Features
- 5-inch HD capacitive touchscreen interface with intuitive icon-based navigation and real-time parameter visualization—including dynamic min/max flow limits calculated per selected syringe geometry.
- Built-in syringe library covering major global manufacturers (e.g., Hamilton, BD, SGE, Ito) with support for user-defined syringe profiles (barrel ID, dead volume, material compliance).
- On-device calibration routine enabling per-syringe volumetric correction; calibrated parameters are non-volatile and retained across power cycles.
- Comprehensive programmable protocol engine: independent setting of target volume, flow rate, infusion/withdrawal duration, pause intervals, cycle count, and force limit—all storable as named methods.
- Real-time flow rate adjustment during active operation without interrupting sequence execution—critical for adaptive titration or feedback-controlled delivery.
- Power-loss resilience: automatically saves runtime state (position, velocity, elapsed time, remaining volume) to flash memory; resumes from identical point upon power restoration.
- Dual-mode external control: accepts TTL-level voltage triggers (5–24 V) or isolated dry-contact signals for synchronized start/stop with chromatography systems, detectors, or PLCs.
- Configurable digital status outputs (TTL-level) indicating operational state (RUN/STOP) and direction (INFUSE/WITHDRAW), facilitating hardware-level interlock logic in regulated environments.
Sample Compatibility & Compliance
The DSC-B01/W70B accommodates standard Luer-lock syringes from 1 µL to 10 mL capacity—including polypropylene, glass, and fluoropolymer-barreled variants. Its programmable force algorithm dynamically constrains actuator torque to prevent syringe barrel deformation or seal extrusion, particularly critical for low-pressure elastomeric or thin-walled disposable units. While not certified to IEC 61010-1 or ISO 13485 out-of-box, the pump’s deterministic control architecture, non-volatile method storage, and timestamped audit trail (via optional PC software) support alignment with GLP and GMP documentation requirements. When integrated into validated systems, its RS485 Modbus RTU interface enables traceable command logging compatible with FDA 21 CFR Part 11-compliant data acquisition platforms.
Software & Data Management
Local configuration and monitoring occur exclusively via the embedded touchscreen—eliminating dependency on host computers during routine use. For system integration, optional Acmer PumpLink™ PC software (Windows/Linux) provides remote control, method import/export (.csv/.xml), and real-time streaming of position, flow, and alarm data at 10 Hz resolution. All user-modified parameters—including calibration coefficients, custom syringe definitions, and protocol sequences—are stored with timestamps and operator IDs in internal flash memory. Communication settings (Modbus slave ID, baud rate, parity) are configurable directly on-device via the USB or RS485 port—no firmware update required.
Applications
- HPLC/UHPLC mobile phase delivery and gradient mixing in research-scale separations
- Controlled reagent addition in kinetic studies, enzymatic assays, and crystallization screening
- Microfluidic device priming, cell culture media perfusion, and organ-on-chip nutrient delivery
- Calibration standard preparation for ICP-MS, AAS, and elemental analysis workflows
- Automated sample introduction in CE, capillary electrophoresis, and micro-CE systems
- Low-flow solvent delivery in cryo-EM grid preparation and serial femtosecond crystallography
FAQ
Does the pump support syringe calibration with traceable standards?
Yes—users may perform volumetric calibration using gravimetric or timed-collect methods; calibration factors are stored per syringe ID and applied in real time during operation.
Can multiple pumps be daisy-chained via RS485?
Yes—the Modbus RTU protocol supports up to 32 devices on a single bus with individually assignable slave IDs and configurable termination.
Is the force limit adjustable per protocol step?
No—force is set globally per syringe profile but may be overridden temporarily during manual jog mode; persistent step-specific force control requires external logic layer.
What is the minimum detectable volume increment?
The theoretical resolution is 0.001 µL (1 nL), limited by lead-screw pitch and encoder resolution; actual achievable precision depends on syringe compliance and fluid compressibility.
Does the pump meet electromagnetic compatibility (EMC) requirements for laboratory use?
It complies with CISPR 11 Group 1, Class B emission limits; conducted/radiated immunity testing per IEC 61326-1 is available upon request for system-level validation.