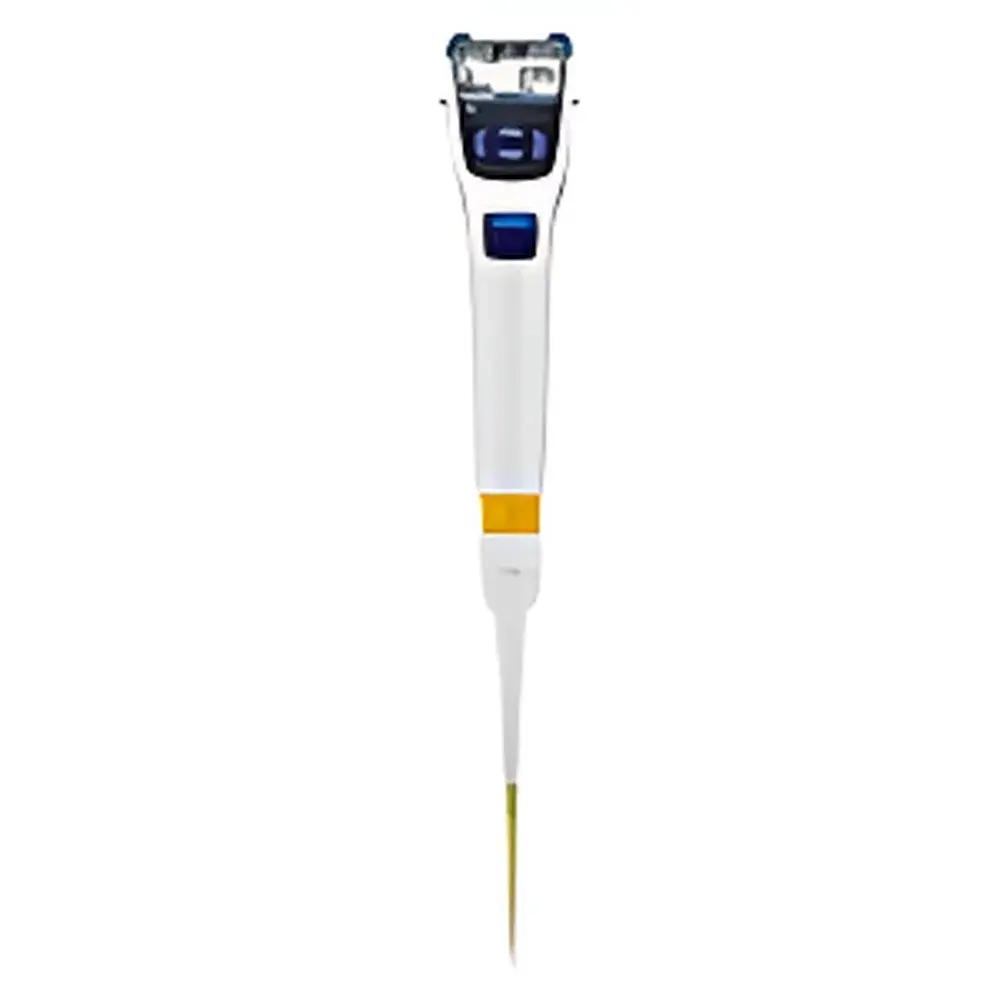
A&D MPA-200 Electronic Single-Channel Pipette
| Brand | A&D |
|---|---|
| Origin | Japan |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Origin | Imported |
| Model | MPA-200 |
| Instrument Type | Single-Channel Pipette |
| Automation Level | Motorized |
| Channels | 1 |
| Volume Range | 10–200 µL |
| Maximum Capacity | 200 µL |
| Accuracy | ±2.5% at 10 µL, ±0.6% at 200 µL |
| Precision (CV) | ≤1.0% at 10 µL, ≤0.15% at 200 µL |
| Power Source | Rechargeable Lithium-ion Battery |
| Charging Interface | USB-C |
| Calibration | User-accessible gravimetric and volumetric calibration routines |
Overview
The A&D MPA-200 is a high-precision, motorized single-channel pipette engineered for reproducible liquid handling in regulated laboratory environments—including clinical diagnostics, pharmaceutical QC, academic research, and GLP-compliant assay development. Operating on a brushless DC motor-driven piston mechanism, the MPA-200 delivers consistent aspiration and dispense performance across its full 10–200 µL range by eliminating manual force variability. Its design adheres to ISO 8655-2:2022 requirements for piston-operated volumetric apparatus, ensuring traceability to national metrology institutes when calibrated per manufacturer-recommended protocols. Unlike mechanical or semi-automated alternatives, the MPA-200 integrates closed-loop position sensing and adaptive torque control to maintain plunger displacement accuracy under varying ambient temperatures (15–30 °C) and tip resistance conditions—critical for viscous or volatile samples.
Key Features
- Four programmable pipetting modes: Standard (forward) pipetting, Repeat Dispensing (multi-dispense), Mix-Dispense (aspirate-mix-dispense cycles), and Reverse Pipetting—optimized for low-volume, high-viscosity, or foaming liquids.
- Ergonomic soft-touch keypad with tactile feedback reduces finger fatigue and mitigates risk of repetitive strain injury (RSI) during extended workflows; actuation force is <0.3 N per key press.
- One-click tip attachment system employs spring-loaded conical coupling to ensure gas-tight seal without manual compression—validated against ISO 8655-6:2022 tip retention force specifications (≥25 N).
- Shock-absorbing polymer housing and reinforced LCD bezel provide drop resistance up to 1 m onto hard surfaces (per IEC 60068-2-32), protecting internal mechanics and display integrity.
- Integrated rechargeable lithium-ion battery supports ≥1,000 pipetting cycles per charge (at 100 µL, standard mode); USB-C port enables full recharge in ≤90 minutes with power delivery up to 5 V/1.5 A.
- User-accessible calibration suite includes both gravimetric (mass-based) and volumetric (density-compensated) adjustment routines—compatible with ASTM E1154-20 and ISO/IEC 17025:2017 documentation requirements.
Sample Compatibility & Compliance
The MPA-200 accommodates universal low-retention and filtered tips (10–200 µL nominal capacity), including OEM-certified A&D PTFE-coated tips and third-party ISO 8655-compliant variants. It maintains volumetric fidelity across aqueous buffers (e.g., PBS, Tris-HCl), organic solvents (e.g., ethanol, DMSO), and protein-rich matrices (e.g., serum, cell lysates) when used with appropriate tip selection and pre-wetting protocols. The device complies with electromagnetic compatibility standards IEC 61326-1:2013 and safety standard IEC 61010-1:2010. For regulated environments, audit trails generated during user calibration are timestamped and stored locally—supporting alignment with FDA 21 CFR Part 11 (electronic records/signatures) when paired with validated LIMS integration.
Software & Data Management
While the MPA-200 operates as a standalone instrument, its calibration logs and usage history (including date/time stamps, volume settings, and mode selections) are exportable via USB-C connection to host PCs running Windows/macOS. Exported CSV files include unique instrument ID, operator ID field (user-definable), and ISO 8655-conforming uncertainty calculations. No proprietary software installation is required; raw data conforms to FAIR principles (Findable, Accessible, Interoperable, Reusable) and integrates natively with ELN platforms supporting .csv ingestion. Firmware updates are delivered through A&D’s secure web portal (accessible globally without regional restrictions) and verified via SHA-256 checksum prior to installation.
Applications
- Quantitative PCR (qPCR) setup requiring precise 10–50 µL master mix dispensing with ≤0.15% CV at upper range.
- Cell culture passaging where reverse pipetting minimizes shearing of sensitive suspension lines (e.g., iPSCs, primary neurons).
- High-throughput ELISA plate preparation with repeat dispensing to 96- or 384-well formats.
- Reference material dilution series in metrology labs accredited to ISO/IEC 17025.
- Method validation studies per USP and EP 2.6.17, where documented calibration frequency and traceability are mandatory.
FAQ
Does the MPA-200 support GLP/GMP documentation requirements?
Yes—calibration events generate immutable, timestamped records with operator ID fields and uncertainty values calculated per ISO 8655-5:2022 Annex B. These logs meet ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) data integrity criteria.
Can the pipette be recalibrated in-house without external service?
Yes—users may perform both gravimetric and volumetric recalibration using certified weights and reference fluids (e.g., distilled water at 20 °C, density = 0.998203 g/mL), following procedures outlined in the included ISO 8655-aligned calibration manual.
Is tip ejection force adjustable?
No—the ejection mechanism delivers a fixed, optimized force of 3.2 N (±0.3 N) to ensure reliable tip release without damaging racks or causing aerosol generation.
What is the recommended recalibration interval?
A&D recommends verification every 3 months for routine use, or after 10,000 actuations—whichever occurs first—as aligned with ISO/IEC 17025:2017 clause 7.7.1 for measurement equipment management.
Are firmware updates backward-compatible with older calibration data formats?
Yes—all firmware versions preserve legacy CSV schema structure and field definitions to ensure continuity of long-term trend analysis and regulatory submissions.