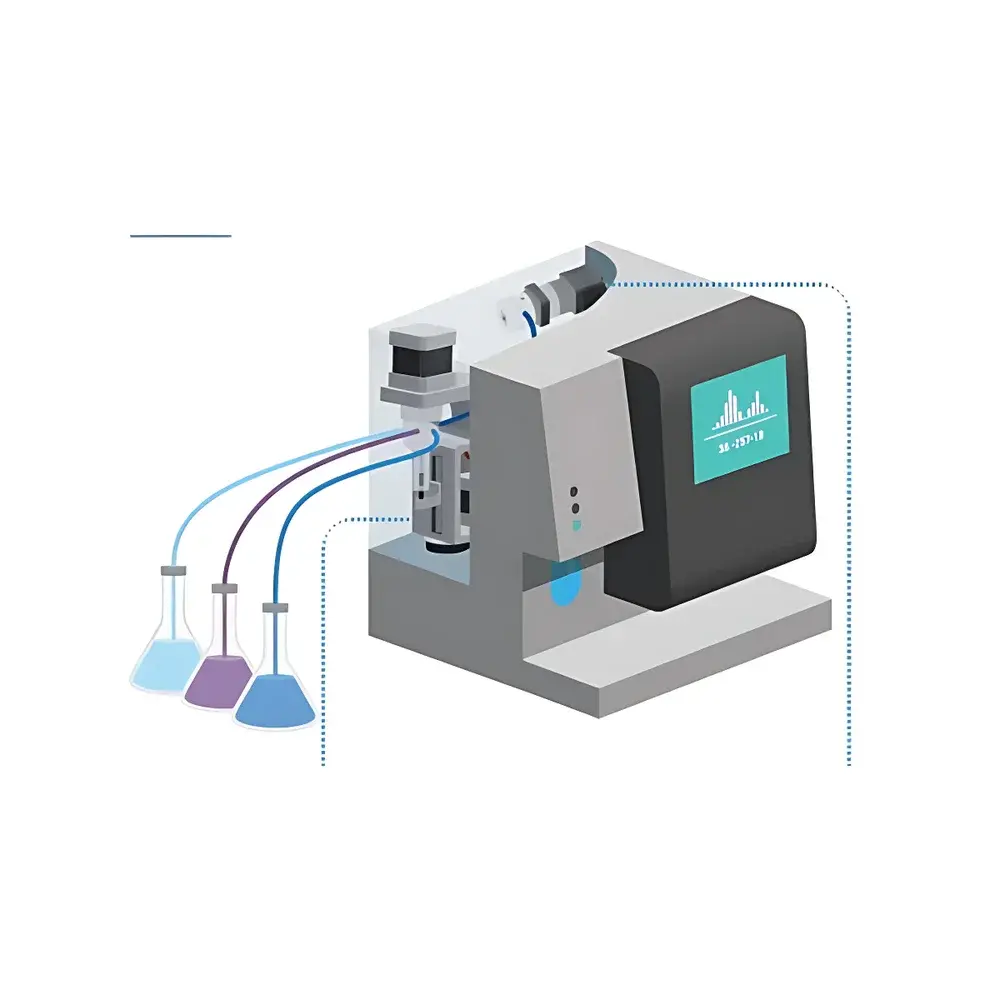
Advanced Microfluidics LSPone Integrated Microfluidic Syringe Pump
| Brand | Advanced Microfluidics |
|---|---|
| Origin | Switzerland |
| Model | LSPone |
| Instrument Type | Precision Microfluidic Syringe Pump |
| Fluid Interface | 1/4-28 UNF |
| Valve Options | 6/8/10/12-port rotary selector valve |
| Valve Materials | PCTFE, PTFE, or UHMW-PE |
| Syringe Capacity | 25–1000 µL |
| Internal Volume | <4 µL |
| Residual Volume | ≤1.5 µL |
| Software | LSPoneQuick (Windows/macOS) |
| Compliance | Designed for GLP/GMP-aligned workflows, supports audit-trail-capable operation via software logging |
Overview
The Advanced Microfluidics LSPone Integrated Microfluidic Syringe Pump is an engineered solution for high-fidelity, multi-fluid handling in academic and translational research environments. Based on a co-integrated architecture—combining a precision stepper-motor-driven syringe pump with a zero-dead-volume microfluidic rotary selector valve—the LSPone operates on the principle of positive displacement with deterministic fluid path selection. Its core design eliminates traditional dead volumes inherent in tubing-based manifolds or solenoid-switched systems, thereby minimizing carryover, reducing reagent consumption, and enabling reproducible nanoliter-scale delivery across heterogeneous fluids. The system is calibrated for volumetric accuracy across its full 25–1000 µL syringe range and maintains stable flow rates from sub-nL/min to >100 µL/min, depending on syringe geometry and fluid viscosity. Manufactured in Switzerland under strict ISO 9001-certified processes, the LSPone meets the dimensional, chemical, and operational constraints typical of lab-on-a-chip, single-cell analysis, and automated assay development platforms.
Key Features
- Zero-dead-volume integrated rotary selector valve with selectable port count (6/8/10/12) and chemically inert wetted materials (PCTFE, PTFE, or UHMW-PE)
- Ultra-low internal volume (<4 µL) and residual volume (≤1.5 µL), validated per ASTM D7219 for residual quantification in microfluidic systems
- Borosilicate glass syringes with PTFE- or UHMW-PE-coated plungers, compatible with organic solvents, aqueous buffers, and biological matrices including serum and cell lysates
- Patented air-ejection valve architecture enabling bubble-free priming without manual purging or syringe pre-filling
- Standardized 1/4-28 UNF female fluid ports for direct integration with microfluidic chips, capillaries, or analytical interfaces
- Compact footprint (190 × 140 × 120 mm) suitable for benchtop use inside biosafety cabinets or instrument enclosures
- Firmware-upgradable control electronics supporting TTL, RS-232, and USB virtual COM protocols for OEM integration
Sample Compatibility & Compliance
The LSPone accommodates a broad spectrum of sample types—including viscous polymer solutions (up to 100 mPa·s), particulate suspensions (e.g., microbeads, exosomes), and shear-sensitive biologics—without clogging or degradation, thanks to its large-diameter microchannels (≥250 µm) and absence of sharp constrictions. All wetted surfaces comply with USP Class VI and ISO 10993-5 cytotoxicity standards. The system supports traceable operation under GLP and GMP frameworks: LSPoneQuick software logs timestamped events (valve actuation, syringe position, flow rate changes) with optional digital signature and user authentication, aligning with FDA 21 CFR Part 11 requirements for electronic records. It is routinely deployed in laboratories adhering to ISO/IEC 17025 for method validation and ASTM E2917 for liquid handling system verification.
Software & Data Management
LSPoneQuick is a native cross-platform application (Windows 10/11, macOS 12+) providing intuitive graphical sequencing, real-time flow visualization, and macro-based automation. Users define multi-step protocols—including sequential aspiration/dispense, dynamic dilution gradients, timed valve switching, and synchronized dual-pump coordination—via drag-and-drop workflow builder. All sequences are exportable as XML for version control and audit readiness. Raw positional data (motor step counts) and derived volumetric outputs are logged at ≥10 Hz resolution in CSV format, enabling post-hoc correlation with optical, electrochemical, or mass spectrometric detection signals. An open API (RESTful HTTP + Python SDK) permits integration into LabVantage, Benchling, or custom LIMS environments, supporting FAIR data principles.
Applications
- Lab-on-a-chip device characterization and calibration, especially for pressure-driven and electrokinetic microfluidic platforms
- Automated preparation of serial dilutions for ELISA, qPCR, and immunoassay standard curves with CV <1.2% at 100 nL steps
- Controlled reagent injection in microscale reaction monitoring (e.g., stopped-flow kinetics, enzyme turnover assays)
- Multi-analyte sampling for coupled microfluidic–mass spectrometry workflows
- Cell culture media exchange and gradient generation in organ-on-chip models
- OEM integration into point-of-care diagnostic instruments requiring precise, low-volume fluid metering under battery-constrained conditions
FAQ
What syringe sizes are supported, and how does size selection affect flow precision?
The LSPone accepts syringes from 25 µL to 1000 µL. Smaller syringes yield higher volumetric resolution (e.g., 25 µL syringe achieves ~80 pL/step), while larger syringes provide extended run time and lower relative error at high flow rates.
Can the LSPone be used with non-standard or custom valves?
Yes—Advanced Microfluidics offers OEM valve redesign services, including port layout modification, material substitution (e.g., fused silica for UV transparency), and motor integration for specialized timing profiles.
Is remote operation possible over Ethernet or Wi-Fi?
While the standard interface is USB, optional Ethernet-to-serial gateways enable networked control; Wi-Fi operation is not natively supported due to latency and determinism requirements in microfluidic timing-critical applications.
How is calibration traceability maintained?
Each unit ships with a factory calibration certificate referencing NIST-traceable gravimetric measurements. End-users may perform in-house recalibration using certified microgravimetric balances and ISO 8655-compliant procedures.
Does the system support bidirectional flow (e.g., recirculation loops)?
Yes—the integrated rotary valve enables fully programmable forward/reverse flow paths, including closed-loop perfusion configurations when paired with external pressure sensors or flow meters.