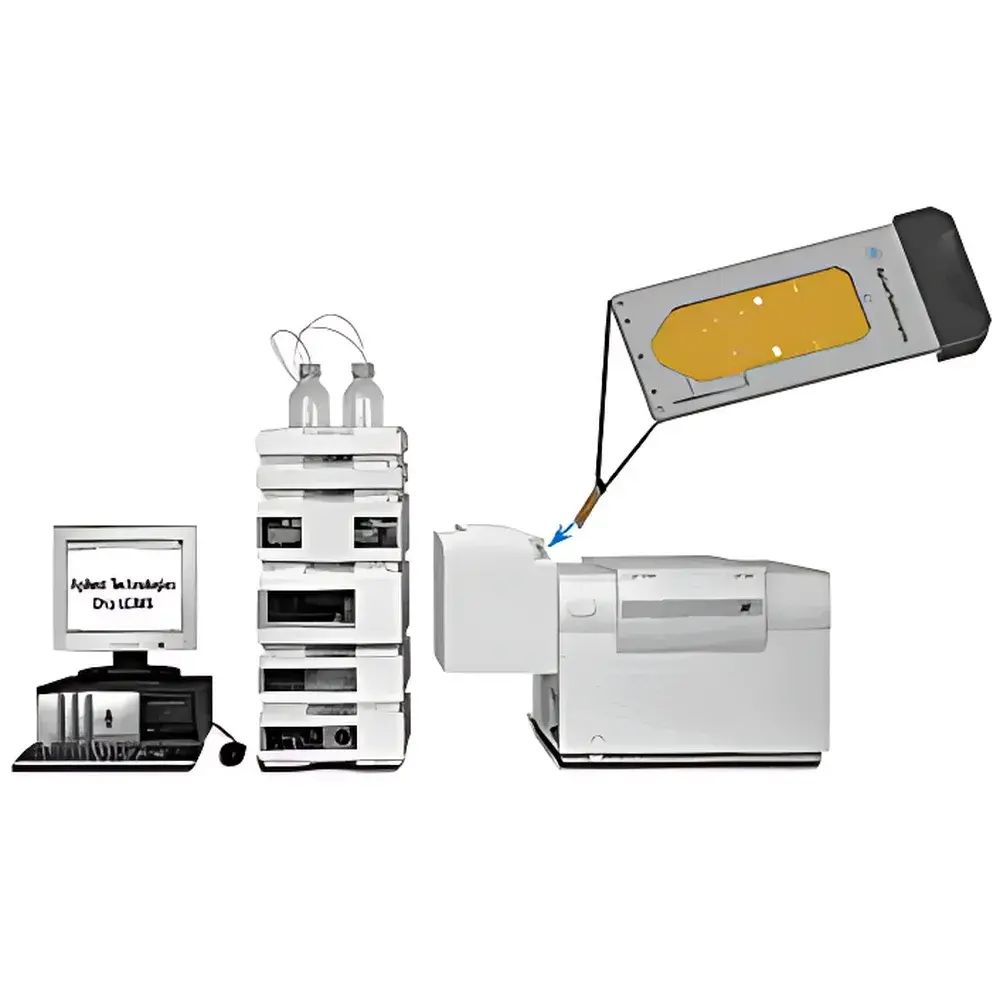
Agilent 1200 Series LC-Chip/MS System
| Brand | Agilent Technologies |
|---|---|
| Origin | Singapore |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Origin Category | Imported Instrument |
| Model | 1200 |
| Quotation | Upon Request |
| Instrument Type | Triple Quadrupole |
Overview
The Agilent 1200 Series LC-Chip/MS System is a fully integrated liquid chromatography–mass spectrometry platform engineered for high-sensitivity, high-reproducibility analysis of small molecules and biomolecules in complex matrices. Unlike conventional LC-MS interfaces, this system employs Agilent’s proprietary chip-based nanoflow electrospray ionization (nanoESI) technology—where the separation column, enrichment column, and electrospray emitter are monolithically fabricated on a single silicon or polymer microfluidic chip. This architecture minimizes dead volume, suppresses analyte dispersion, and delivers exceptional robustness for low-flow (50–300 nL/min) and high-efficiency separations. Coupled with triple quadrupole mass spectrometry detection, the system operates under multiple reaction monitoring (MRM), precursor ion scanning, neutral loss scanning, and selected reaction monitoring (SRM) modes—enabling quantitative precision at sub-pg/mL levels and structural elucidation via controlled fragmentation. It is purpose-built for regulated environments requiring compliance with ICH guidelines, USP , and FDA 21 CFR Part 11–compliant data integrity frameworks.
Key Features
- Integrated LC-Chip interface with dual-column configuration (enrichment + analytical) for automated sample cleanup and on-chip gradient elution
- Triple quadrupole mass analyzer with unit-mass resolution, <0.7 Da full-width half-maximum (FWHM), and <2 ms dwell time per transition
- Electrospray ion source optimized for nanoflow operation, delivering >80% ion transmission efficiency across 50–2000 m/z range
- Dynamic range exceeding 4 orders of magnitude in quantitative mode (linear response from 1 pg to 100 ng on-column)
- Real-time polarity switching capability (positive/negative mode within 20 ms) without signal loss or calibration drift
- Rugged, modular design supporting 24/7 unattended operation with automated calibration and performance verification protocols
- Compliance-ready firmware with audit trail logging, electronic signatures, and role-based access control
Sample Compatibility & Compliance
The system accommodates a broad spectrum of sample types—including plasma, serum, urine, tissue homogenates, cell lysates, fermentation broths, and environmental extracts—without requiring extensive offline derivatization or solid-phase extraction. Its chip-based interface tolerates moderate levels of salts, detergents, and organic modifiers common in biological workflows. All hardware and software components meet ISO 9001-certified manufacturing standards. The platform supports GLP/GMP-aligned workflows through built-in instrument qualification (IQ/OQ/PQ) templates, traceable calibration records, and raw data immutability features aligned with Annex 11 and ALCOA+ principles.
Software & Data Management
Controlled by Agilent MassHunter Workstation Software (Version B.08.00 or later), the system provides end-to-end method development, acquisition, processing, and reporting. Key modules include Quantitative Analysis, Qualitative Analysis, and Metabolite ID. Data files adhere to open mzML format for third-party interoperability. Audit trails capture every user action—including parameter changes, integration edits, and report generation—with timestamps, operator IDs, and reason codes. Electronic signatures comply with 21 CFR Part 11 requirements when configured with validated certificate authority integration and password policies.
Applications
- Pharmaceutical bioanalysis: PK/PD studies, metabolite profiling, and impurity quantification per ICH M10 guidance
- Clinical toxicology: Multi-analyte screening of drugs of abuse, pharmaceuticals, and endogenous biomarkers in human matrices
- Environmental monitoring: Detection of pesticides, PFAS, and pharmaceutical residues in water and soil extracts
- Food safety: Residue testing for veterinary drugs, mycotoxins, and allergenic peptides in animal-derived products
- Proteomics support: Targeted peptide quantification (e.g., SRM for biomarker verification) and post-translational modification analysis
FAQ
Is the Agilent 1200 Series LC-Chip/MS compatible with existing Agilent 6400-series triple quadrupole instruments?
Yes—the LC-Chip interface is mechanically and electronically compatible with Agilent 6410, 6460, and 6470 triple quadrupole mass spectrometers via standard API source mounting and MassHunter driver integration.
What chip formats are supported?
Standard configurations include HPLC-Chip/MS (300 µm × 5 mm C18 enrichment / 75 µm × 43 mm C18 analytical) and Nano-Chip/MS (160 µm × 5 mm C18 / 75 µm × 150 mm C18), with optional porous graphitic carbon (PGC) and HILIC variants.
Does the system support method transfer from conventional HPLC methods?
Yes—Agilent provides method translation tools in MassHunter that automatically convert flow rates, gradients, and injection volumes from standard LC to chip-based nanoflow conditions while preserving peak capacity and selectivity.
How is system performance verified?
Built-in performance checks include tuning compound response (e.g., reserpine), mass accuracy verification (<±0.1 Da), sensitivity assessment (S/N ≥ 500:1 for 100 fg on-column), and retention time stability (<0.2% RSD over 24 h).