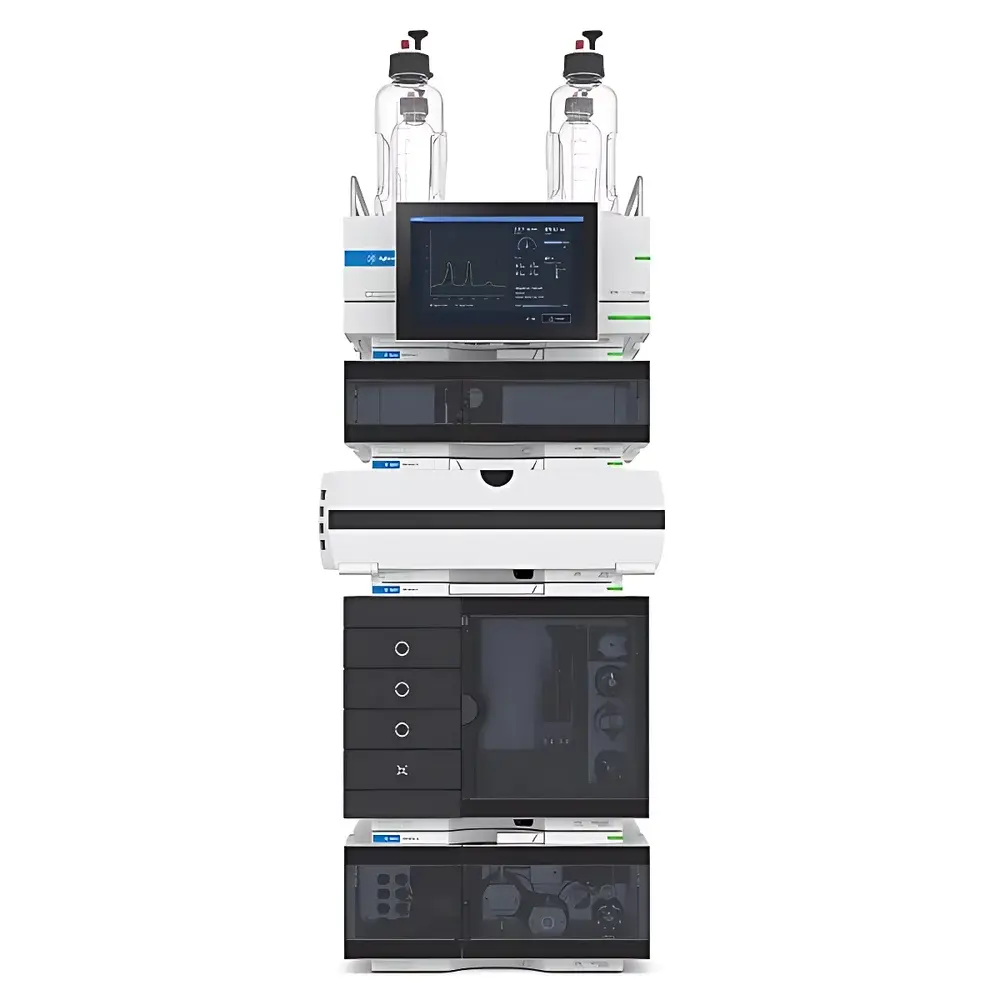
Agilent 1260 Infinity III Amino Acid Analysis System
| Brand | Agilent Technologies |
|---|---|
| Origin | Germany |
| Manufacturer | Agilent Technologies |
| Import Status | Imported |
| Model | 1260 Infinity III Amino Acid Analysis System |
| Instrument Type | Ultra-High Performance Liquid Chromatograph (UHPLC) |
| Application Scope | General-purpose amino acid analysis |
| Flow Rate Range | Up to 10 mL/min (G7111B pump module) |
| Maximum Operating Pressure | 600 bar |
| Injection Volume Range | 0.1–100 µL |
| Column Oven Temperature Range | 5 °C above ambient to 80 °C |
| Solvent Reservoir Capacity | 4 channels |
| Power Supply | 100–240 VAC |
| Dimensions (W × D × H) | 396 × 468 × 560 mm |
| Column ID Reader | Optional |
| Smart System Emulation | Not supported |
| ACT Certification | My Green Lab ACT certified for environmental accountability, consistency, and transparency |
Overview
The Agilent 1260 Infinity III Amino Acid Analysis System is a purpose-engineered UHPLC platform designed specifically for high-throughput, quantitative, and qualitative amino acid profiling in regulated and research environments. It integrates the robustness of Agilent’s InfinityLab liquid chromatography architecture with optimized column chemistry, automated pre-column derivatization, and multi-modal detection—enabling reproducible separation and detection of all 20 proteinogenic amino acids plus common non-proteinogenic analogs (e.g., ornithine, citrulline, γ-aminobutyric acid). The system operates on reversed-phase chromatography principles, leveraging post-derivatization or pre-column o-phthalaldehyde (OPA), fluorenylmethyloxycarbonyl chloride (FMOC), or ninhydrin chemistries—fully compatible with ISO 13022, AOAC 994.12, and USP guidelines for amino acid quantitation in pharmaceutical excipients, biologics, nutritional supplements, and infant formula matrices.
Key Features
- Automated pre-column derivatization using the Agilent 1260 Infinity III Sample Tray Autosampler (G7129B), eliminating manual reagent handling and ensuring inter-run precision (RSD < 1.5% for peak area across 24-h sequence)
- Agilent InfinityLab Poroshell 120 HPH column (2.7 µm superficially porous particles, 120 Å pore size), delivering high-resolution separation at sub-400 bar backpressure—enabling extended column lifetime and reduced solvent consumption
- Dual-detection capability: Simultaneous UV/VIS diode-array detection (DAD) at 200–400 nm for peak identification and purity assessment, coupled with programmable fluorescence detection (FLD) for enhanced selectivity in complex biological matrices
- Thermostatically controlled column compartment (5–80 °C, ±0.1 °C stability) and autosampler cooling (4–40 °C) ensure retention time reproducibility and analyte stability during long sequences
- Modular pump architecture (G7111B quaternary pump) supporting gradient elution from 0.1% to 99.9% organic modifier, with pulse-free flow delivery across 0.05–10 mL/min range
- My Green Lab ACT certification validates the system’s lifecycle environmental impact—covering energy use, material sourcing, packaging recyclability, and end-of-life recovery pathways
Sample Compatibility & Compliance
The system supports hydrolyzed protein digests (acidic or enzymatic), cell culture supernatants, plasma/serum extracts, food hydrolysates, and fermentation broths. Sample introduction is compatible with standard 2-mL vials and 96-well microplates. All derivatization protocols are aligned with GLP/GMP documentation requirements, and the system supports 21 CFR Part 11-compliant audit trails when operated with OpenLab CDS software. Method validation parameters—including linearity (r² ≥ 0.999 over 0.1–100 µM), LOD (≤ 5 fmol for glycine under FMOC-FLD), and carryover (< 0.05%)—meet ICH Q2(R2) expectations for QC release testing.
Software & Data Management
Controlled via Agilent OpenLab CDS ChemStation Edition or OpenLab ECM, the system enables full method development, sequence scheduling, real-time monitoring, and automated report generation. Built-in compliance tools include electronic signatures, user role-based access control, instrument qualification status dashboards, and configurable electronic audit trails. Raw data files (.D format) are stored with embedded metadata (temperature, pressure, flow, injection volume, derivatization timing), ensuring traceability for FDA inspections and ISO/IEC 17025 accreditation audits.
Applications
- Quantitative amino acid profiling in monoclonal antibody formulations and biosimilars per ICH Q5C stability guidelines
- Verification of nitrogen content and essential amino acid ratios in infant nutrition products (Codex Alimentarius STAN 72-1981)
- Residual host cell protein characterization in upstream bioprocess development
- Authenticity testing of hydrolyzed collagen and whey protein concentrates in food fraud mitigation workflows
- Metabolomic screening of amino acid dysregulation in clinical research cohorts (e.g., phenylketonuria, maple syrup urine disease)
FAQ
Is this system compliant with pharmacopeial monographs such as USP <467> and EP 2.2.57?
Yes—the hardware configuration, column chemistry, and derivatization workflow are fully aligned with these monographs when implemented using Agilent-certified methods and reference standards.
Can the system be integrated into an existing OpenLab CDS network environment?
Yes—it supports seamless integration with OpenLab CDS v2.5 or later, including centralized instrument management, shared method libraries, and cross-platform data review workflows.
What column dimensions and particle sizes are recommended for routine QC applications?
The standard configuration uses a 150 × 4.6 mm, 2.7 µm Poroshell 120 HPH column; alternative configurations (e.g., 100 × 3.0 mm for LC-MS coupling) are validated and documented in Agilent Application Notes #5991-8704EN.
Does the system support post-run column cleaning and equilibration protocols?
Yes—gradient methods can include programmable wash steps up to 95% organic, followed by automatic re-equilibration to initial conditions, minimizing carryover and extending column longevity.
Is remote monitoring and diagnostics available?
Yes—Agilent Remote Manager (ARM) provides secure, browser-based access to real-time pressure/flow/temperature telemetry, alarm notifications, and predictive maintenance alerts via TLS 1.2 encrypted connection.