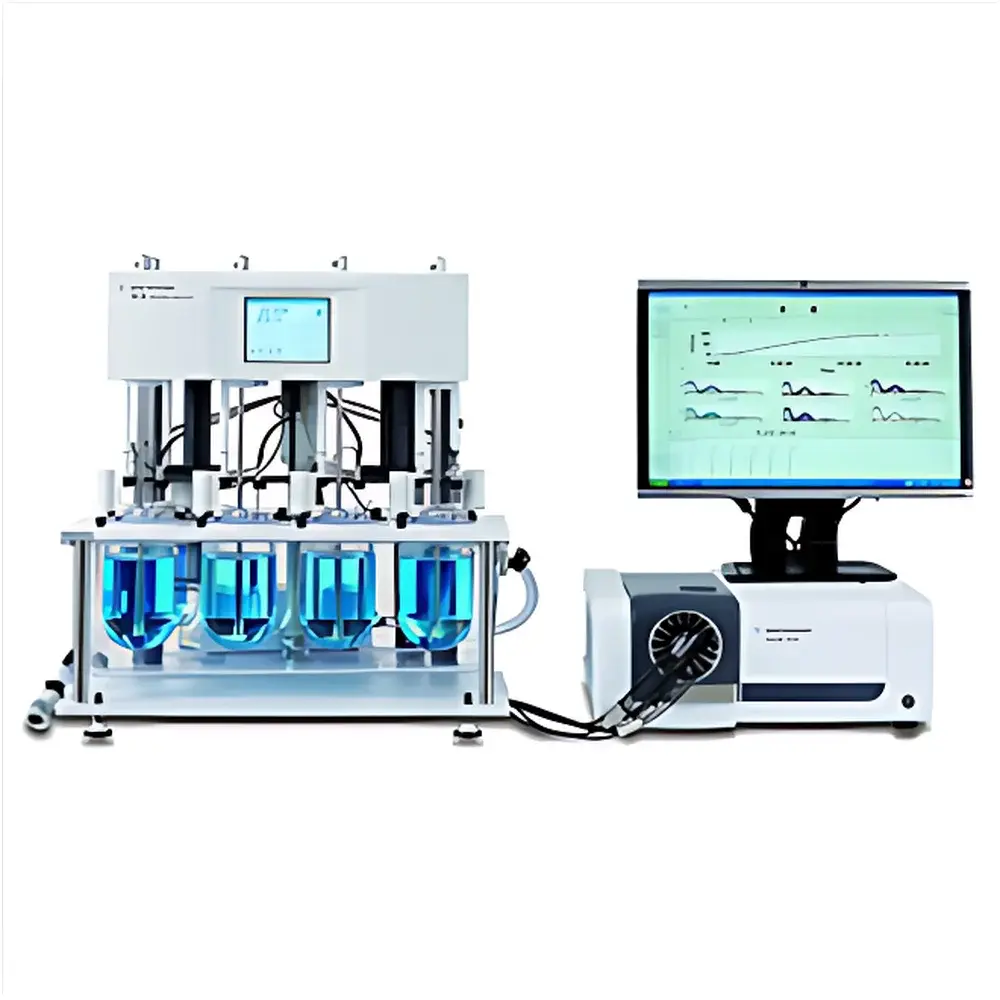
Agilent 708-DS Dissolution Tester
| Brand | Agilent Technologies |
|---|---|
| Origin | Imported |
| Manufacturer Type | Manufacturer |
| Instrument Type | Online Dissolution Tester |
| Vessel Capacity | 8-position |
| Temperature Range | Ambient +5 °C to 55 °C |
| Temperature Accuracy | ±0.1 °C |
| Rotation Speed Range | 10–250 rpm |
| Bath Volume | 18 L |
Overview
The Agilent 708-DS Dissolution Tester is a modular, GMP-aligned platform engineered for precision dissolution testing across pharmaceutical development, quality control, and regulatory submission workflows. It operates on the hydrodynamic principles defined in USP , Ph. Eur. 2.9.3, and JP 17, supporting all four official compendial methods—Basket (USP Apparatus 1), Paddle (USP Apparatus 2), Reciprocating Cylinder (USP Apparatus 6), and Flow-Through Cell (USP Apparatus 4)—as well as non-standard configurations including paddle-over-disk (USP Apparatus 5). Its design emphasizes mechanical stability, thermal uniformity, and fluid dynamic reproducibility, minimizing extrinsic variables such as vibration transmission, temperature gradient drift, and vessel misalignment. The system accommodates dissolution vessels from 100 mL to 2 L, enabling method scalability from early-stage formulation screening to commercial batch release.
Key Features
- Modular architecture with interchangeable dissolution accessories—including TruAlign-certified vessels, inert DLC-coated paddles, Apex (Peak) vessels, and low-volume mini-vessels—for method flexibility and material compatibility.
- Integrated bath-heating and circulation system positioned at the rear of the unit, reducing benchtop footprint and mechanical coupling to adjacent instrumentation; engineered to maintain ≤±0.1 °C temperature stability across all 8 vessels simultaneously.
- Touchscreen HMI with multilingual support (English, Spanish, Japanese, Simplified Chinese) and intuitive navigation for manual operation or parameter-driven protocol execution.
- Rugged mechanical drive system delivering precise rotational control from 10 to 250 rpm with ≤±0.5 rpm repeatability under load.
- 18-L thermostatically controlled water bath with rounded internal corners to minimize residue accumulation and accelerate cleaning validation cycles.
- Optional automated dosing, sampling, and UV-Vis integration via Agilent 850-DS Sampling Station and Cary 60 spectrophotometer—enabling end-to-end unattended dissolution analysis.
Sample Compatibility & Compliance
The 708-DS supports dissolution testing of diverse dosage forms: immediate- and extended-release oral solids (tablets, capsules), transdermal patches, topical semisolids, implantable matrices, and combination products incorporating drug-device interfaces. All hardware configurations comply with mechanical calibration requirements outlined in USP , EP 2.9.3, and JP 17. Vessel alignment accuracy is verified using Agilent’s TruAlign optical verification kit, ensuring centering tolerance within ±1.0 mm per USP specification. The system meets ISO/IEC 17025 prerequisites for analytical instrument qualification and supports full IQ/OQ/PQ documentation packages. When operated with Agilent Dissolution Workstation Software or Cary WinUV Dissolution Analysis Software, it satisfies data integrity requirements under FDA 21 CFR Part 11, EU Annex 11, and ICH GCP/GLP guidelines—including electronic signatures, audit trails, and role-based access control.
Software & Data Management
Agilent Dissolution Workstation Software enables centralized control of up to four 708-DS units from a single PC, synchronizing test parameters, sampling events, and data logging. The software supports method templates compliant with pharmacopeial time-point schedules (e.g., 5/10/15/30/45/60 min), automatic volume compensation for evaporation, and real-time monitoring of vessel temperature and rotation speed. Integrated export modules generate CSV, PDF, and XML outputs compatible with LIMS and statistical process control platforms. When coupled with Cary WinUV Dissolution Analysis Software, raw absorbance data from the Cary 60 UV-Vis spectrophotometer is automatically converted into concentration-time profiles, with built-in calculation of Q-value compliance, similarity factor (f2), and deconvolution modeling. All software modules are validated per GAMP 5 and include full change control documentation.
Applications
- Formulation development and optimization of release kinetics for modified-release dosage forms.
- Comparative dissolution profiling for bioequivalence studies (BCS Class I–IV).
- Stability-indicating dissolution method development per ICH Q5C and Q1A(R2).
- Quality control release testing of commercial batches against approved specifications.
- Investigational support for regulatory submissions (IND, NDA, ANDA, MAA) requiring dissolution data traceable to compendial standards.
- Method transfer between R&D, QC, and contract laboratories with documented equivalence protocols.
FAQ
Does the 708-DS meet USP Apparatus 4 (Flow-Through Cell) requirements?
Yes—the system supports USP Apparatus 4 via optional flow-through cell modules with programmable pump rates, temperature-controlled reservoirs, and inline filtration.
Can the 708-DS be qualified for 21 CFR Part 11 compliance?
Yes—when used with validated Agilent Dissolution Workstation or Cary WinUV software, the system implements electronic signatures, audit trails, and user access controls required for regulated environments.
Is temperature uniformity verified across all eight vessels?
Yes—thermal mapping per USP Annex is supported using Agilent-certified PT100 probes; typical inter-vessel deviation is ≤±0.05 °C at 37 °C.
What maintenance intervals are recommended for the bath circulation system?
Agilent recommends quarterly inspection of pump seals and annual calibration of temperature sensors using NIST-traceable references.
Can legacy dissolution methods be migrated to the 708-DS without revalidation?
Method transfer requires formal comparison testing per ICH Q5E; however, the 708-DS’s mechanical equivalency to USP reference apparatuses minimizes protocol adjustments.