Agilent BioTek Synergy Neo2 Hybrid Multi-Mode Microplate Reader
| Brand | Agilent Technologies |
|---|---|
| Origin | USA |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Origin | Imported |
| Model | BioTek Synergy Neo2 Hybrid |
| Detection Methods | Absorbance, Fluorescence Intensity (tunable bandwidth), Luminescence, Time-Resolved Fluorescence (TRF), Fluorescence Polarization (FP), TR-FRET, BRET, AlphaScreen/AlphaLISA |
| Detector Types | Dual Photomultiplier Tubes (PMTs), Monochromator-based and Filter-based Optical Paths, 337 nm Pulsed Laser for TRF, 100 mW Solid-State Laser for Alpha Detection |
| Automation | Fully Automated with BioStack Neo |
| Environmental Control | 4-Zone Temperature Control up to 70 °C, CO₂/O₂ Regulation, Anti-Condensation System, Linear & Orbital Shaking |
| Sample Formats | 6–1536-well Microplates, Take3/Take3 Trio Microvolume Plates, Petri Dishes, Cell Culture Dishes |
| Software | Gen5 v3.1+ with LIMS Integration, FDA 21 CFR Part 11 Compliance Support, Audit Trail, Electronic Signatures |
Overview
The Agilent BioTek Synergy Neo2 Hybrid Multi-Mode Microplate Reader is an engineered platform for high-performance, quantitative life science assays across academic, pharmaceutical, and biotechnology laboratories. Built upon a dual-optical-path architecture—integrating independent monochromator-based and filter-based detection systems—the instrument delivers uncompromised sensitivity, speed, and flexibility in absorbance, fluorescence intensity, luminescence, time-resolved fluorescence (TRF), fluorescence polarization (FP), TR-FRET, BRET, and AlphaScreen/AlphaLISA detection modes. Its hybrid design eliminates optical crosstalk and enables simultaneous dual-emission acquisition via two dedicated photomultiplier tubes (PMTs), making it especially suited for ratiometric assays requiring precise kinetic or concentration-dependent signal resolution. The system supports live-cell applications through tightly regulated environmental control—including programmable CO₂/O₂ gas mixing, condensation-suppressed 4-zone heating up to 70 °C, and configurable linear or orbital shaking—ensuring assay reproducibility under physiologically relevant conditions.
Key Features
- Dual-Optical Architecture: Independent monochromator and filter-based light paths eliminate cross-mode interference and maximize signal fidelity across all detection modalities.
- Tunable Bandwidth Monochromator: Adjustable excitation/emission bandwidths (1–50 nm) optimize signal-to-noise ratio—narrower settings enhance specificity in multiplexed assays; broader settings increase throughput and sensitivity.
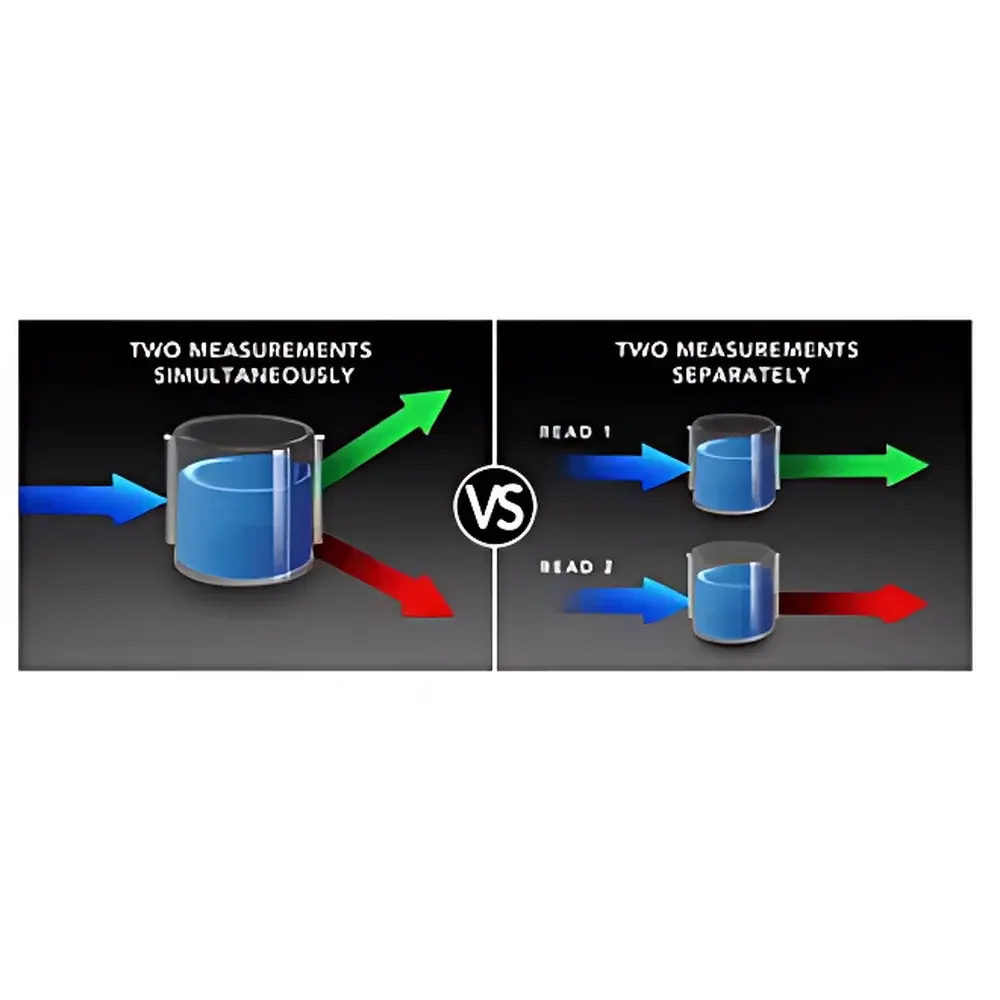
- Dual PMT Detection System: Enables true simultaneous dual-wavelength acquisition—critical for FP, FRET, TR-FRET, and BRET—reducing read time by up to 50% compared to sequential scanning.
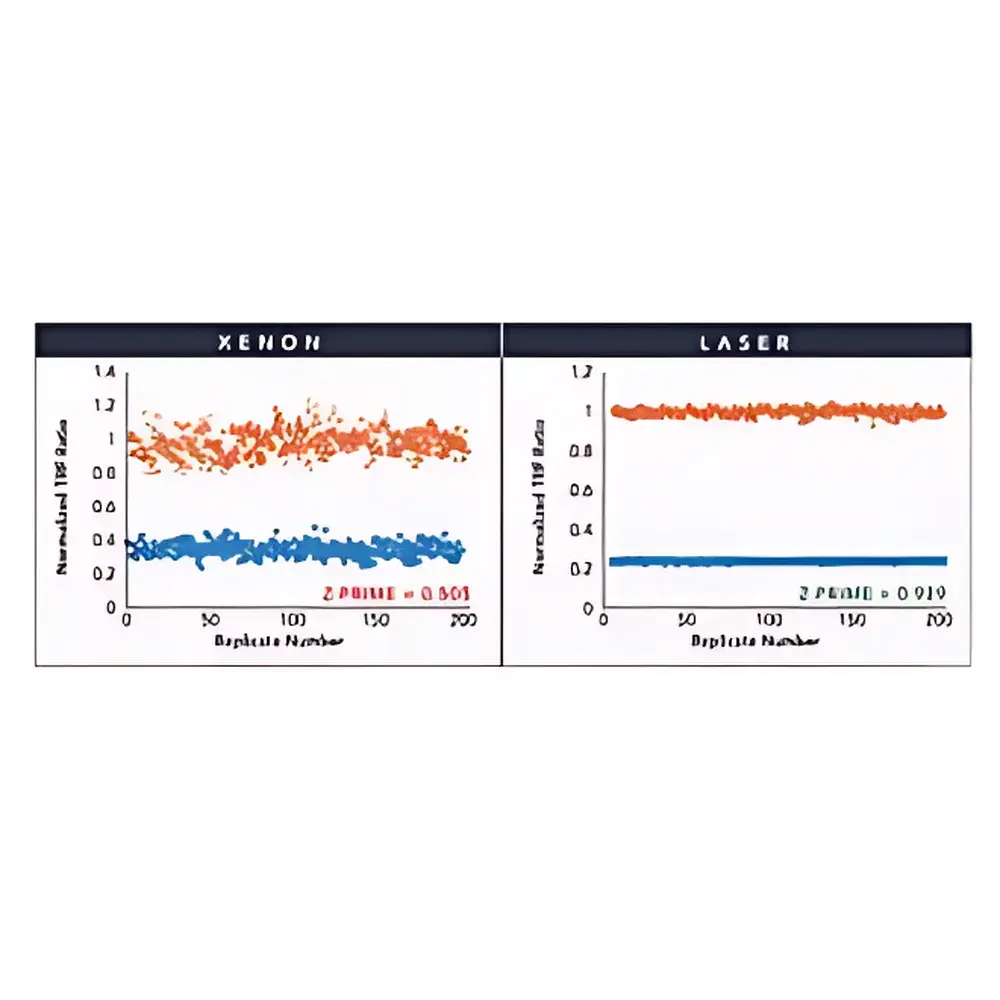
- Dedicated Laser Sources: Integrated 337 nm pulsed nitrogen laser for high-sensitivity TRF/TR-FRET and a 100 mW solid-state laser for AlphaScreen/AlphaLISA assays, delivering superior z′-factor performance and expanded dynamic range.
- Advanced Environmental Control: 4-zone thermal regulation (ambient to 70 °C), programmable CO₂ (0–20%) and O₂ (1–21%) control, anti-condensation optics, and bidirectional shaking ensure robust live-cell assay execution.
- BioStack Neo Automation Interface: Supports barcode-scanned, unattended processing of up to 50 microplates with dual-carrier staging, enabling walk-away operation compatible with GLP/GMP workflows.
Sample Compatibility & Compliance
The Synergy Neo2 accepts standard microplate formats from 6- to 1536-well, including opaque, clear, and black plates with various bottom geometries. It is fully compatible with Agilent’s Take3 and Take3 Trio microvolume plates for nucleic acid and protein quantification (2 µL per sample), as well as Petri dishes and cell culture vessels for imaging-integrated assays. Regulatory alignment includes support for 21 CFR Part 11-compliant data integrity practices via Gen5 software—featuring role-based access control, electronic signatures, full audit trail logging, and secure data export. Instrument validation documentation adheres to ASTM E2892–22 (Standard Guide for Validation of Microplate Readers) and ISO/IEC 17025 principles. All configurations are designated for research use only (RUO); not intended for diagnostic or clinical testing applications.
Software & Data Management
Gen5 v3.1+ software provides unified instrument control, protocol scripting, real-time kinetic monitoring, and advanced data reduction—including built-in algorithms for IC₅₀/EC₅₀ curve fitting, Z’-factor calculation, and multi-parameter normalization. The software supports direct integration with laboratory information management systems (LIMS) via HL7 and ASTM E1384-compliant interfaces. Data files are stored in vendor-neutral .xlsx and .csv formats, with optional encrypted SQLite database archiving. Audit trails record all user actions—including parameter changes, plate mapping edits, and result exports—with timestamped, immutable entries compliant with GLP and GMP documentation requirements.
Applications
The Synergy Neo2 serves as a central analytical node in diverse workflows: high-throughput screening (HTS) of compound libraries using Alpha or TR-FRET; kinetic enzyme activity profiling via absorbance or fluorescence; cellular toxicity and viability assessment using live/dead dyes under controlled CO₂/O₂; protein–protein interaction studies via BRET/FRET; and rapid nucleic acid/protein quantification using Take3 platforms. Its environmental control module enables longitudinal monitoring of proliferation, migration, and metabolic activity in primary and stem cell cultures. Academic labs leverage its modularity for teaching core assay principles; CMC and QC teams deploy it for stability-indicating assays and release testing where regulatory traceability is essential.
FAQ
Is the Synergy Neo2 validated for GxP environments?
Yes—when configured with Gen5’s 21 CFR Part 11 mode and operated under documented SOPs, the system meets ALCOA+ data integrity criteria and supports inspection-ready audit trails.
Can it perform both endpoint and kinetic measurements in the same run?
Yes—Gen5 allows mixed-read protocols combining static endpoint reads with multi-cycle kinetic loops, including temperature- or gas-triggered start conditions.
What is the minimum detectable concentration for TRF assays?
Detection limits depend on assay chemistry and plate type; typical Eu-chelate TRF sensitivity is ≤10 amol/well in low-background white plates.
Does the instrument support third-party automation integrations?
Yes—it communicates via RS-232, USB, and Ethernet using standard BioSpa, BenchCel, and Hamilton-compatible command sets.
Are calibration and performance verification tools included?
Agilent provides NIST-traceable optical calibration standards, pre-installed validation protocols (e.g., absorbance linearity, fluorescence uniformity), and IQ/OQ documentation templates.