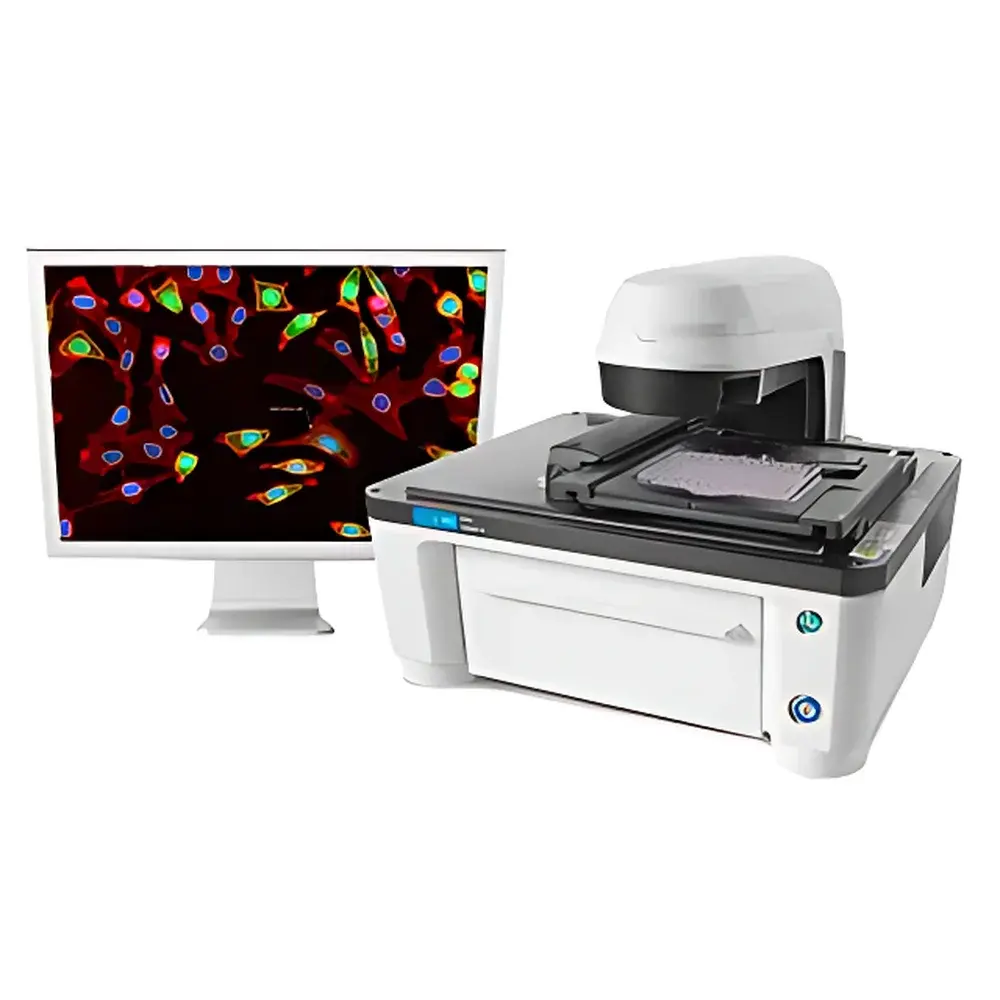
Agilent Lionheart LX Intelligent Cell Imaging and Analysis System
| Brand | Agilent Technologies |
|---|---|
| Origin | USA |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Origin Category | Imported |
| Model | Lionheart LX |
| Pricing | Available Upon Request |
Overview
The Agilent Lionheart LX Intelligent Cell Imaging and Analysis System is a compact, fully automated inverted microscope platform engineered for quantitative live-cell and fixed-tissue imaging in regulated and discovery-oriented life science laboratories. Built upon a robust optical architecture integrating high-precision motorized components and multi-modal illumination, the system performs label-free and fluorescence-based assays without requiring manual eyepiece observation—eliminating observer-induced variability and visual fatigue while supporting reproducible, operator-independent data acquisition. Its core measurement principle relies on digital widefield microscopy coupled with z-axis motorized stage control, enabling optical sectioning (Z-stacking), intensity-based projection algorithms (e.g., maximum-intensity, sum-intensity), and time-resolved image capture across multiple focal planes. Designed for integration into GLP-compliant workflows and routine QC environments, the Lionheart LX delivers consistent spatial resolution, photometric linearity, and temporal stability essential for longitudinal cell health assessment and morphometric phenotyping.
Key Features
- Fully automated inverted microscope with six-position motorized objective turret, supporting simultaneous four-channel fluorescence imaging (e.g., DAPI/FITC/TRITC/Cy5) and seamless switching between brightfield, color brightfield, and epifluorescence modes
- High-accuracy motorized XY stage with sub-micron positioning repeatability and integrated autofocus (using contrast-based or laser-assisted algorithms) for rapid, drift-compensated image acquisition
- Intelligent exposure control with dynamic gain and integration time adjustment per channel and field—optimized to preserve signal-to-noise ratio across heterogeneous samples
- Z-stack acquisition capability (up to 100 slices per position) with programmable step size (0.1–20 µm), enabling volumetric reconstruction and 3D morphometric analysis
- Montage imaging with automatic tile stitching and edge-blending correction for whole-well or full-slide coverage at user-defined magnifications
- Time-lapse video recording (up to 4K resolution) with configurable frame intervals, temperature/humidity/CO2 monitoring compatibility (when paired with environmental chamber accessories)
Sample Compatibility & Compliance
The Lionheart LX accommodates standard microplate formats (6–1536-well), glass-bottom dishes, chamber slides, and histological tissue sections mounted on conventional microscope slides. It supports both adherent and suspension cell models, including primary cultures, iPSC-derived lineages, and co-culture systems. All imaging modalities comply with ISO 13485 design control requirements for IVD-related research use, and software operation aligns with FDA 21 CFR Part 11 principles through Gen5’s audit-trail-enabled user authentication, electronic signature support, and immutable data logging. The system meets ASTM E2912-22 guidelines for digital microscopy validation and is routinely deployed in labs operating under GLP and GMP-aligned quality management systems.
Software & Data Management
Controlled exclusively by Agilent’s Gen5 Microplate Reader and Imaging Software (v3.12+), the Lionheart LX provides an integrated environment for protocol-driven acquisition, batch processing, and quantitative analysis. Gen5 includes over 100 pre-validated application protocols—including confluence measurement, apoptosis (Annexin V/PI), mitochondrial membrane potential (TMRM/JC-1), γH2AX foci quantification, comet assay scoring, and hematoxylin & eosin (H&E) tissue segmentation. Image analysis leverages machine-learning-assisted object recognition (via trainable classifiers), multi-threshold segmentation, and pixel-intensity-based feature extraction (e.g., nuclear area, cytoplasmic texture, fluorescence intensity distribution). Raw image datasets (TIFF, PNG, OME-TIFF) and processed results are exportable in CSV, Excel, or structured JSON formats, supporting traceability and downstream statistical analysis in R, Python, or commercial biostatistics platforms.
Applications
- Endpoint and kinetic live-cell assays: cytotoxicity (LDH, ATP), proliferation (BrdU, EdU), viability (Calcein AM/Propidium Iodide), and functional readouts (Ca2+ flux, ROS generation)
- Subcellular phenotyping: autophagosome formation (LC3 puncta), mitotic index, nuclear fragmentation, and organelle morphology (mitochondria, lysosomes, ER)
- Tissue-level quantification: H&E-stained section area fraction analysis, tumor-infiltrating lymphocyte (TIL) density mapping, and fibrosis scoring
- Genotoxicity assessment: single-cell gel electrophoresis (comet assay) tail moment calculation and γH2AX focus enumeration in irradiated or chemically treated cells
- Cell therapy process monitoring: non-invasive confluence tracking during expansion, differentiation staging via morphological clustering, and post-thaw viability profiling
FAQ
Does the Lionheart LX support environmental control for long-term live-cell imaging?
Yes—when integrated with third-party incubation chambers (e.g., OkoLab, PeCon), the system maintains precise CO2, temperature, and humidity regulation during extended time-lapse experiments.
Can Gen5 software perform batch analysis across multiple plates or time points?
Yes—Gen5 supports plate-level metadata tagging, cross-plate normalization, and time-series alignment using internal reference standards or background subtraction algorithms.
Is the Lionheart LX compatible with high-content screening (HCS) workflows?
While optimized for mid-throughput applications (up to 100 plates/day), it supports HCS-ready features including multi-field-of-view acquisition per well, multi-parameter phenotypic profiling, and LIMS integration via Gen5’s API and secure FTP modules.
What fluorescence filter sets are included by default?
Standard configurations include DAPI, GFP, TRITC, and Cy5; additional filter cubes and LED excitation modules are available as optional accessories to expand spectral coverage.
How is instrument calibration verified and maintained?
Agilent provides NIST-traceable calibration kits for intensity uniformity, stage positioning accuracy, and Z-axis linearity—validatable per ISO/IEC 17025 procedures and documented in the system’s IQ/OQ/PQ qualification package.