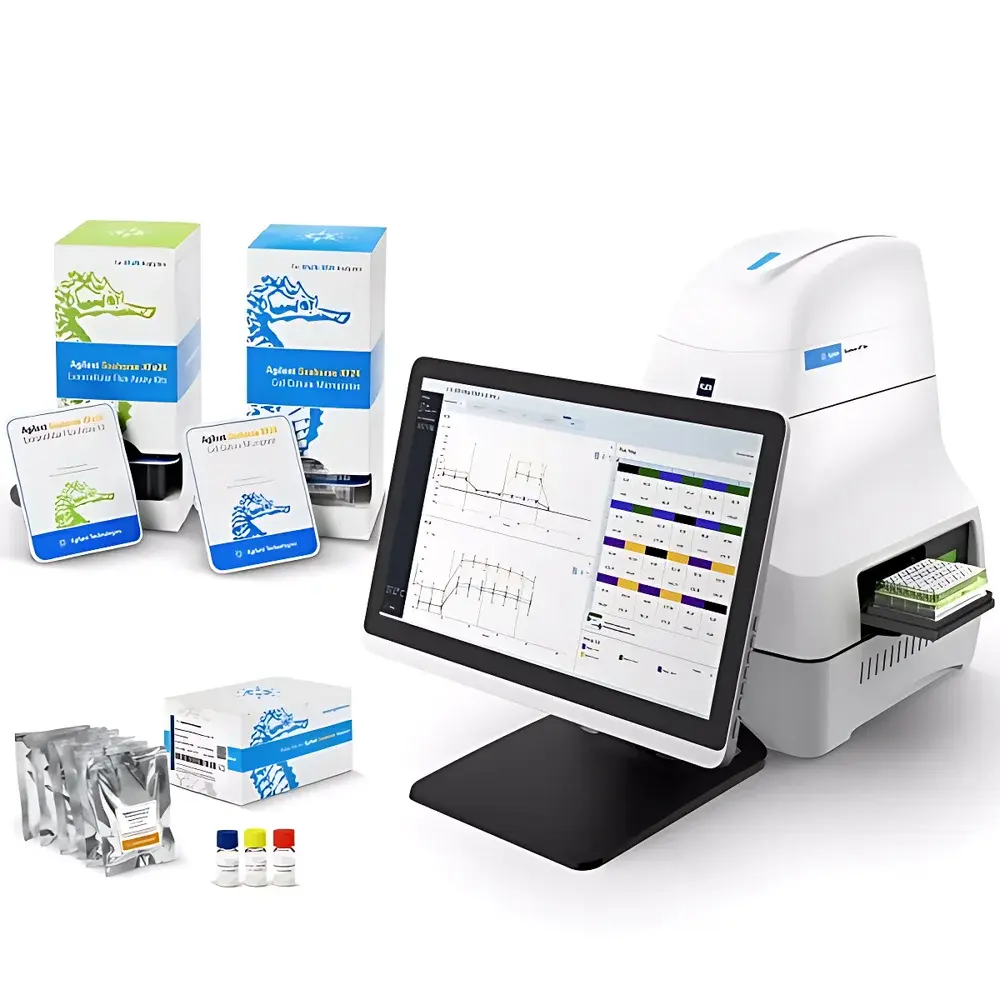
Agilent Seahorse XF Flex Real-Time Cell Metabolic Analyzer
| Brand | Agilent Technologies |
|---|---|
| Origin | USA |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Imported Instrument |
| Model | Seahorse XF Flex |
| Pricing | Available Upon Request |
Overview
The Agilent Seahorse XF Flex Real-Time Cell Metabolic Analyzer is an engineered platform for label-free, functional assessment of cellular bioenergetics in live cells and complex 3D models. It employs patented solid-state sensor technology to simultaneously quantify extracellular oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) — direct proxies for mitochondrial respiration and glycolytic flux, respectively — within standard 24-well microplates. Unlike endpoint assays or genetically encoded reporters, the XF Flex system measures real-time metabolic dynamics under physiologically relevant conditions by forming transient microchambers above each well via a vertically actuated probe cartridge. This architecture enables high-sensitivity detection of minute changes in dissolved O2 and pH, with temporal resolution down to 3-minute measurement cycles. The instrument supports automated, sequential injection of up to four compounds per well, enabling kinetic pharmacological profiling of metabolic responses to substrates, inhibitors, or modulators without perturbing cell integrity.
Key Features
- Flexible 24-well format compatible with both monolayer (2D) cultures and physiologically relevant 3D models including organoids, tissue explants, and multicellular spheroids
- Integrated four-port per-well reagent injection system with programmable mixing, ensuring uniform compound distribution and reproducible stimulus kinetics
- Enhanced dynamic range enabled by proprietary sensor cartridges optimized for high-respiratory models (e.g., primary hepatocytes, cardiomyocytes)
- Stable thermal control across a broad ambient operating range (4–30 °C), maintaining precise assay temperatures from 16–42 °C within the microplate chamber
- Validated intra- and inter-plate data consistency through factory-calibrated optical sensors and standardized probe alignment protocols
- Automated calculation of derived parameters including ATP production rate, proton efflux rate (PER), coupling efficiency, and spare respiratory capacity
Sample Compatibility & Compliance
The XF Flex accommodates diverse biological samples requiring minimal preprocessing: adherent or suspension cells (8,000–500,000 cells/well), intact tissue fragments, cryopreserved organoids, and scaffold-based 3D constructs. Sample volumes per well are maintained at 600–1000 µL to ensure optimal diffusion kinetics within the microchamber. All consumables — including assay plates, sensor cartridges, and calibration kits — are manufactured under ISO 13485-certified quality systems and validated for lot-to-lot performance consistency. The platform supports GLP-compliant workflows through audit-trail-enabled software logging, electronic signatures, and full traceability of instrument settings, calibration history, and raw sensor output files. Data generated meet requirements for regulatory submissions aligned with FDA 21 CFR Part 11 and ICH M10 guidelines for bioanalytical method validation.
Software & Data Management
The XF Flex operates via the Agilent Seahorse Wave Pro software suite running on a dedicated Windows 11 Enterprise controller workstation equipped with a 16 GB RAM, touchscreen interface, and integrated TCP/IP/USB/WiFi connectivity. Wave Pro provides guided experimental design, real-time instrument monitoring, and automated QC flagging based on predefined thresholds for signal drift, baseline stability, and injection artifact detection. Raw time-series data (.xfp files) are natively compatible with Agilent Seahorse Analytics — a secure, browser-based analytical engine supporting batch normalization, kinetic curve fitting, pathway-specific metric derivation (e.g., glycolytic capacity, maximal respiration), and statistical comparison across treatment groups. All data files retain embedded metadata including plate map configuration, reagent lot numbers, environmental logs, and user-defined annotations, facilitating FAIR (Findable, Accessible, Interoperable, Reusable) data stewardship.
Applications
- Immunometabolism: Quantify metabolic reprogramming during T-cell activation, macrophage polarization, and memory B-cell differentiation — linking OCR/ECAR shifts to functional outcomes such as cytokine secretion or cytotoxic activity
- Oncology Research: Characterize Warburg effect dynamics, mitochondrial uncoupling, and metabolic plasticity in patient-derived tumor organoids under therapeutic pressure
- Toxicology & Mitochondrial Health: Assess compound-induced bioenergetic dysfunction using tiered stress tests (e.g., oligomycin/FCCP/rotenone/antimycin A), calculating key indices like basal respiration, ATP-linked respiration, and non-mitochondrial oxygen consumption
- Neurodegenerative Disease Modeling: Monitor progressive bioenergetic decline in iPSC-derived neurons and glia exposed to amyloid-beta or alpha-synuclein aggregates
- Metabolic Disease Studies: Evaluate insulin resistance phenotypes in primary adipocytes or skeletal myotubes following chronic fatty acid exposure
FAQ
What sample types are supported beyond standard 2D cell cultures?
The XF Flex has been validated for use with primary tissue slices, decellularized scaffolds seeded with stromal cells, air-liquid interface (ALI) epithelial cultures, and cryopreserved human organoids — all without enzymatic dissociation.
How does the XF Flex ensure measurement reproducibility across laboratories?
Through factory-installed optical calibration, standardized probe cartridge geometry, and firmware-controlled microchamber formation algorithms that eliminate manual alignment variability.
Can data from older Seahorse instruments be reanalyzed in Seahorse Analytics?
Yes — Seahorse Analytics supports legacy .xls and .txt exports from XF96/XF24 platforms, enabling longitudinal comparison when migrating to XF Flex workflows.
Is the system compliant with electronic lab notebook (ELN) integration standards?
The XF Flex exports structured JSON and CSV metadata packages compatible with major ELNs via RESTful API, supporting automated ingestion of assay parameters, QC metrics, and final computed values.
What maintenance is required to sustain sensor accuracy over time?
Annual recalibration using Agilent-provided reference cartridges is recommended; no user-serviceable optical components exist — all sensors are single-use and replaced with each assay plate.