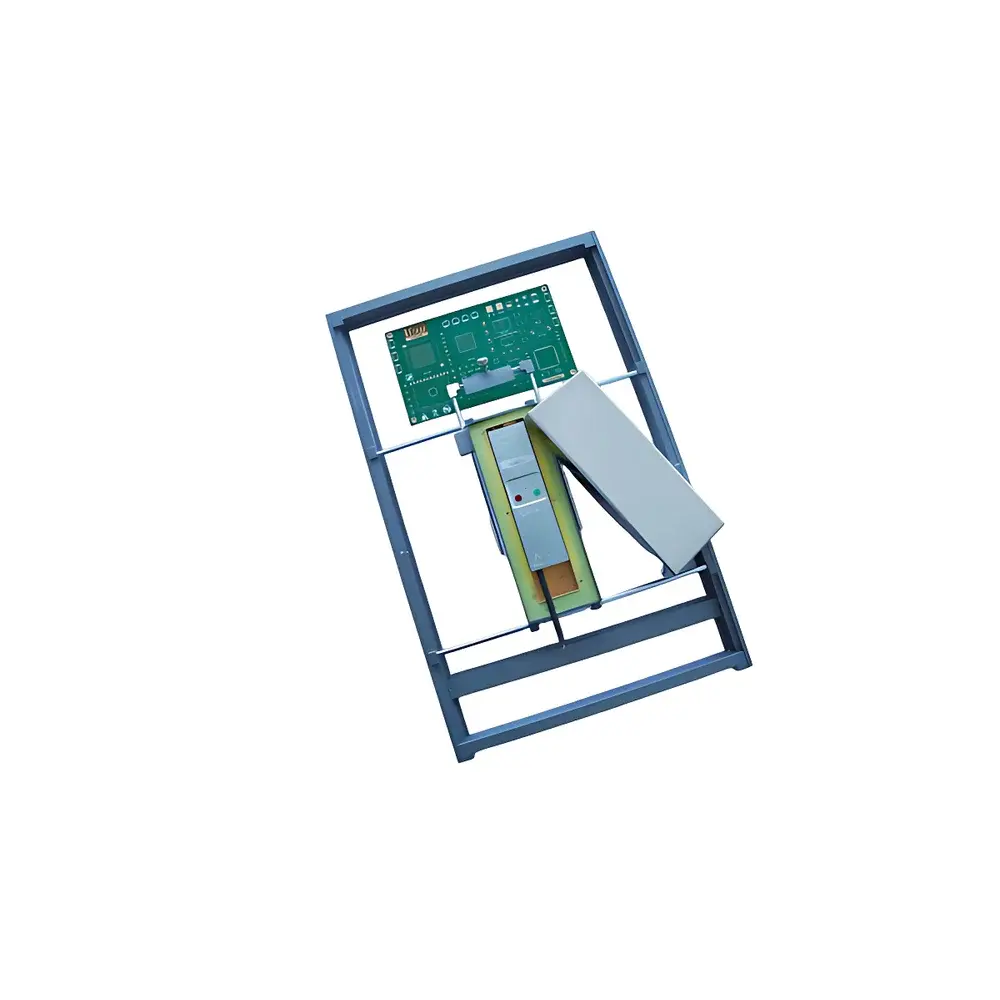
Allsheng Esan-Gene 164 Real-Time Fluorescent Quantitative PCR System
| Brand | Allsheng |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | Esan-Gene 164 Real-Time Fluorescent Quantitative PCR System |
| Quotation | Available Upon Request |
| Throughput | 16-well (0.2 mL tubes or 8-tube strips) |
| Sample Volume Range | 10–100 µL |
| Dynamic Range | 1–10¹⁰ copies |
| Excitation Source | High-Stability LED |
| Detection Channels | 4 (F1: 515–535 nm |
| F2 | 535–555 nm |
| F3 | 600–620 nm |
| F4 | 665–695 nm) |
| Fluorescence Detection Range | 400–770 nm |
| Detector Type | Photodiode (PD) |
| Block Temperature Range | 4–99 °C |
| Temperature Accuracy | ±0.15 °C |
| Temperature Uniformity | ±0.2 °C @ 95 °C |
| Max Heating Rate | 4.0 °C/s |
| Max Cooling Rate | 3.0 °C/s |
| Melting Curve Resolution | 0.1 °C |
| Heat Lid Temperature Range | 30–110 °C (default: block +10 °C) |
| Heat Lid Accuracy | ±2 °C |
| Power Supply | 220 V AC, 50 Hz |
| Max Power Consumption | 200 W |
| Dimensions (W×D×H) | 200 × 207 × 148 mm |
| Weight | <3.5 kg |
| Operating System | Embedded Windows-based UI |
| Display | 7-inch capacitive touchscreen |
| Connectivity | USB 2.0, Ethernet, optional direct thermal printer interface |
Overview
The Allsheng Esan-Gene 164 Real-Time Fluorescent Quantitative PCR System is a compact, self-contained thermal cycler engineered for high-fidelity nucleic acid amplification and real-time fluorescence detection. Based on the principle of sequence-specific DNA amplification coupled with synchronized optical monitoring of fluorescent reporter dyes (e.g., SYBR Green I, hydrolysis probes, and hybridization probes), the instrument enables quantitative determination of target nucleic acid copy numbers via threshold cycle (Ct) analysis. Its integrated thermoelectric heating/cooling architecture—comprising precision Peltier elements and active heat sink management—ensures rapid thermal ramping (up to 4.0 °C/s), tight temperature uniformity (±0.2 °C at 95 °C), and exceptional thermal accuracy (±0.15 °C). Designed explicitly for field-deployable molecular diagnostics, the Esan-Gene 164 meets the operational demands of biosafety level 2 (BSL-2) environments in public health laboratories, food safety inspection units, agricultural testing centers, and point-of-need veterinary diagnostics.
Key Features
- Standalone operation: Full workflow execution—including protocol setup, thermal cycling, fluorescence acquisition, Ct calculation, melt curve analysis, and result classification—is performed without external computers or software dependencies.
- Four-channel multiplex detection: Simultaneous excitation and emission filtering across four optically isolated channels (F1–F4) supports common dye sets (FAM/SYBR Green I, HEX/JOE/VIC, ROX, Cy5), enabling robust multi-target assays and internal passive reference normalization.
- LED-based optical engine: Solid-state light sources eliminate lamp aging, warm-up delays, and alignment drift—delivering stable excitation intensity over >50,000 cycles with no scheduled maintenance.
- High-resolution melt (HRM) capability: Thermal control precision of 0.1 °C enables reliable discrimination of single-nucleotide polymorphisms (SNPs) and allelic variants through post-amplification melting profile analysis.
- Embedded Windows OS with 7-inch capacitive touchscreen: Intuitive graphical interface supports bilingual (English/Chinese) language switching, drag-and-drop protocol editing, and on-device data visualization including amplification plots, melt curves, and scatter dot plots.
- Comprehensive data handling: Internal storage retains ≥10,000 run records; export formats include CSV, PDF, and proprietary .qpcr files compatible with third-party analysis tools.
Sample Compatibility & Compliance
The Esan-Gene 164 accepts standard 0.2 mL thin-wall transparent PCR tubes and 8-tube strips—compatible with widely adopted consumables from major global suppliers. Its 16-well block design conforms to ANSI/SLAS microplate footprint standards for mechanical interoperability. The system operates within ISO/IEC 17025-accredited laboratory frameworks when deployed under documented SOPs. While not pre-certified to FDA 21 CFR Part 11, its audit trail functionality—including timestamped user login logs, parameter change history, and electronic signature-ready report generation—supports GLP and GMP-aligned validation protocols. All thermal and optical performance metrics adhere to ASTM E1957-21 (Standard Practice for Validation of Real-Time PCR Instruments) guidelines for dynamic range verification, linearity assessment, and inter-run reproducibility.
Software & Data Management
The embedded firmware implements a deterministic real-time operating layer that guarantees consistent timing between thermal transitions and fluorescence acquisition windows. Data acquisition occurs at user-defined intervals (1–10 seconds per cycle), with raw fluorescence intensity values stored per channel and well. Post-run analysis includes automatic baseline correction, threshold setting (fixed, adaptive, or manual), efficiency-corrected quantification, and genotype clustering based on melt peak morphology. Exported datasets retain full metadata (instrument ID, operator, date/time, calibration status, thermal profile trace) to satisfy traceability requirements under ISO 15189 and CLIA regulations. Network connectivity permits secure remote monitoring via HTTPS-enabled web interface and centralized data aggregation using LIMS-compatible RESTful API endpoints.
Applications
- Public health surveillance: Rapid detection and quantification of SARS-CoV-2, influenza A/B, RSV, and other respiratory pathogens in mobile testing units.
- Foodborne pathogen screening: Enumeration of Salmonella spp., Listeria monocytogenes, and E. coli O157:H7 directly from enrichment cultures.
- Agricultural biotechnology: GMO event-specific detection (e.g., MON810, NK603), varietal authentication, and transgene copy number estimation in plant breeding programs.
- Veterinary diagnostics: Identification of avian influenza subtypes, porcine reproductive and respiratory syndrome virus (PRRSV), and foot-and-mouth disease virus (FMDV).
- Academic research: Gene expression profiling using reverse transcription qPCR (RT-qPCR), microRNA quantification, and CRISPR-based diagnostic assay development.
FAQ
Is the Esan-Gene 164 compliant with international regulatory standards for clinical diagnostics?
The instrument meets core technical requirements outlined in ISO 20387 (biobanking) and ISO 13485 (medical device quality management), though formal IVDR or CE-IVD certification requires end-user validation under site-specific intended use claims.
Can the system perform absolute quantification without a standard curve?
No—absolute quantification requires a serially diluted reference standard of known copy number; however, the system supports digital PCR cross-validation workflows when paired with appropriate sample partitioning accessories.
What is the recommended maintenance schedule for the optical module?
None—the LED excitation source and photodiode detector are solid-state components with no consumable parts; periodic verification of optical alignment and background fluorescence using blank wells is advised every 6 months.
Does the instrument support external barcode scanning for sample tracking?
Yes—USB-connected barcode scanners are natively recognized; scanned IDs auto-populate well labels during protocol setup, ensuring full chain-of-custody documentation.
How is thermal calibration verified during routine operation?
A built-in NIST-traceable thermistor array continuously monitors block temperature distribution; users may initiate automated calibration verification using certified reference materials (e.g., DNA melting standards) per ASTM E2551-20 Annex A1.