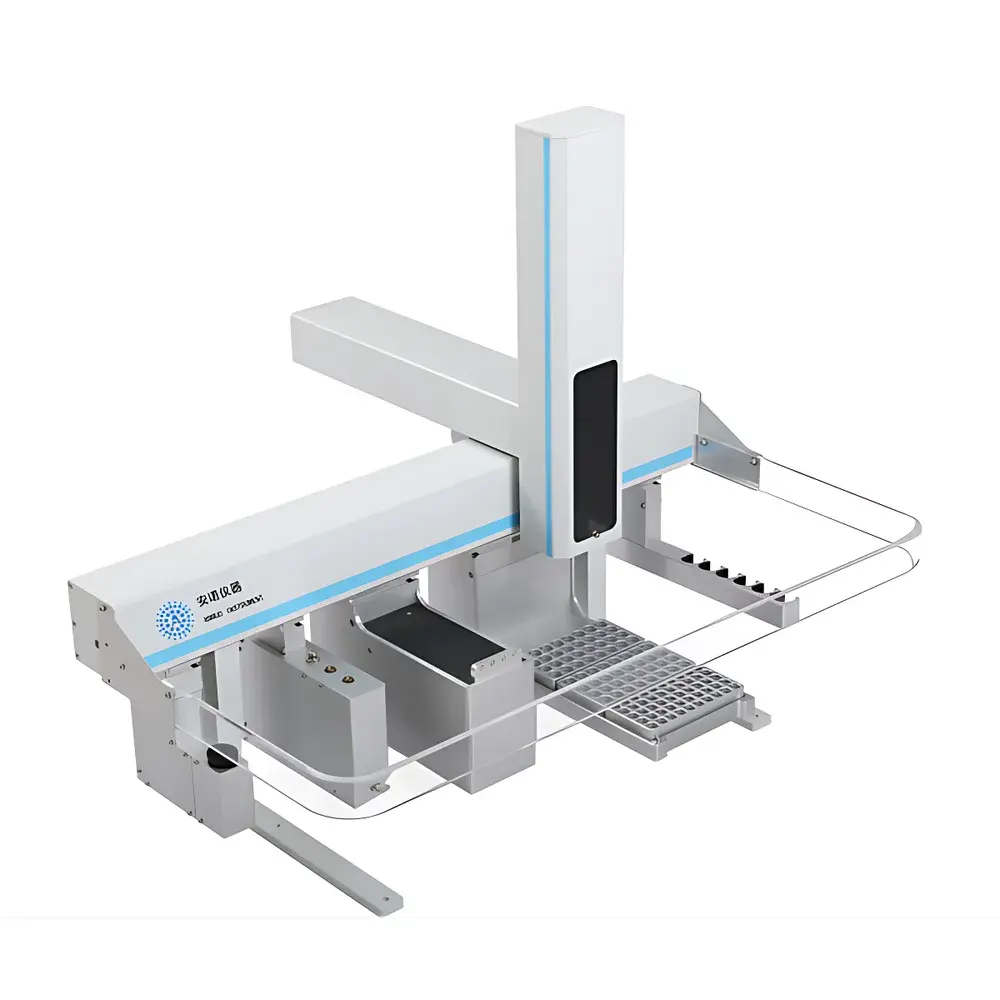
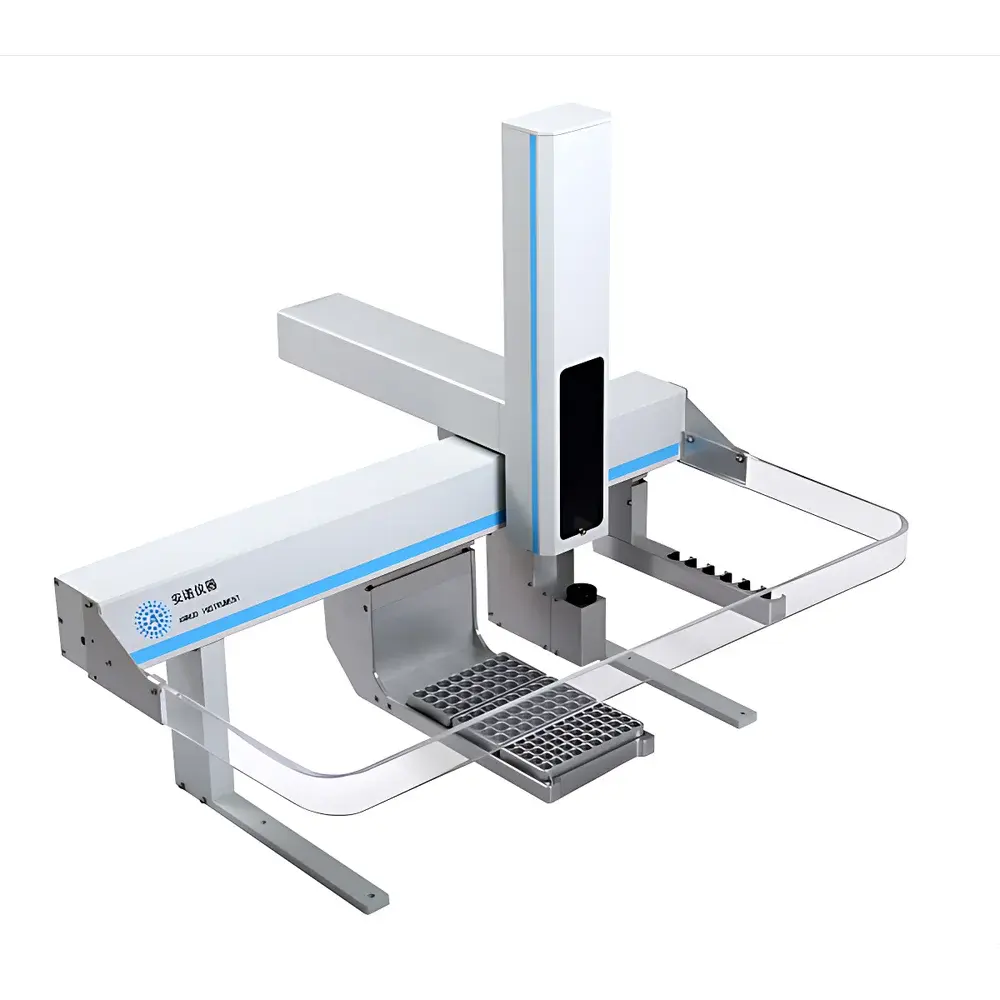
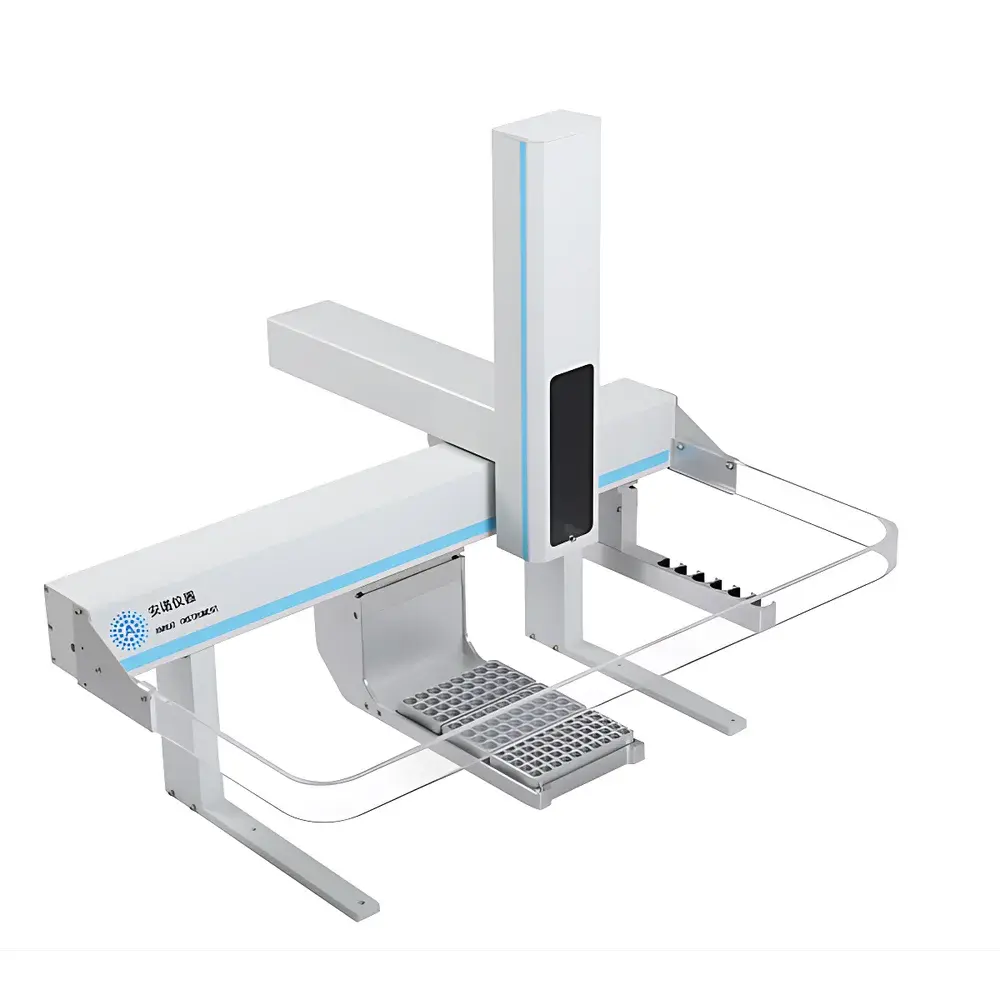
Annuo FAS-3823 Multi-Function Solid Phase Microextraction (SPME) Autosampler
| Brand | Annuo |
|---|---|
| Origin | Henan, China |
| Model | FAS-3823 |
| Sample Vial Capacity | 10 mL, 20 mL (standard) |
| Sample Positions | 70 positions (expandable to 140) |
| Extraction Temperature Range | Ambient to 150 °C (±0.5 °C control accuracy) |
| Stirring Speed | 0–3000 rpm |
Overview
The Annuo FAS-3823 is a fully integrated, multi-modal laboratory autosampler engineered for high-throughput, reproducible sample preparation and introduction in gas chromatography (GC) and GC–mass spectrometry (GC–MS) workflows. Unlike conventional single-function autosamplers, the FAS-3823 implements a unified hardware platform capable of executing liquid-phase injection, headspace sampling, solid-phase microextraction (SPME), and automated standard solution preparation—all under programmable, software-controlled conditions. Its operational principle relies on precise robotic positioning, thermally regulated sample conditioning, and modular actuation systems that support both fiber-based extraction (via motorized SPME probe deployment) and syringe-driven fluid handling. Designed for compliance-critical environments, the instrument adheres to fundamental design principles aligned with ISO/IEC 17025 method validation requirements and supports audit-ready operation when integrated with GLP/GMP-compliant chromatographic data systems.
Key Features
- Modular, CAN-bus-based architecture enabling independent firmware updates and functional expansion without system downtime
- 4.3-inch capacitive touchscreen interface with guided workflow navigation and real-time status feedback
- Rotary turret-style sampling mechanism ensuring sub-millimeter positional repeatability (< ±0.05 mm) across all vial types
- Dual-mode thermal control: independent heating zones for sample vials (ambient to 300 °C), SPME fiber desorption (up to 350 °C), and needle jacketing (up to 200 °C)
- Programmable agitation: magnetic stirring (0–750 rpm) and mechanical orbital shaking (0–3000 rpm) with configurable duration and delay intervals
- Multi-channel I/O interface supporting Modbus RTU and USB 2.0 protocols for seamless integration with third-party GC controllers and LIMS platforms
- Full PC-based remote control with bidirectional command execution, real-time parameter monitoring, and event logging compliant with FDA 21 CFR Part 11 requirements
- Redundant safety logic including input validation, motion boundary detection, and thermal runaway prevention with automatic shutdown
Sample Compatibility & Compliance
The FAS-3823 accommodates standard crimp-top and screw-cap vials (2 mL, 4 mL, 10 mL, and 20 mL formats) across multiple tray configurations—including 70-position SPME trays (expandable to 140), 180-position headspace trays, and 6×60-position liquid-injection racks. All thermal modules meet ASTM E2912-21 specifications for temperature uniformity and stability. The system supports method transfer per USP and ISO 17025 Annex A.3 guidelines, with traceable calibration records available for temperature sensors (NIST-traceable Pt100 elements), volume delivery (gravimetrically verified syringe calibration), and timing accuracy (±10 ms). Optional validation packages include IQ/OQ documentation templates aligned with pharmaceutical QC laboratories.
Software & Data Management
The bundled Annuo Control Suite provides full instrument configuration, sequence building, and real-time supervision via Windows-based desktop application. It supports method cloning, version-controlled protocol storage, and export of structured CSV/Excel logs containing timestamped events, temperature profiles, motion trajectories, and error codes. Audit trails record all user actions—including parameter edits, sequence starts/stops, and emergency stops—with immutable timestamps and operator ID tagging. Data export conforms to ASTM E1382-22 standards for analytical instrument metadata interchange. API access is provided through documented REST endpoints for custom LIMS or ELN integrations.
Applications
- Environmental analysis: SPME-GC–MS quantification of volatile organic compounds (VOCs) in water, soil leachates, and air samples
- Food & flavor profiling: headspace analysis of esters, aldehydes, and terpenes in beverages and dairy products
- Pharmaceutical residual solvent testing: automated USP compliant headspace-GC assays for Class 2 and 3 solvents
- Clinical toxicology: high-precision calibration curve generation using robotic dilution series (1:2 to 1:100)
- Polymer additive migration studies: thermal desorption of plasticizers from packaging matrices into food simulants
- Forensic toxicology: unattended batch processing of blood, urine, and hair extracts prior to GC–NPD/FID analysis
FAQ
Does the FAS-3823 support dual-column GC configurations?
Yes—the system features six independently addressable injection ports and can be programmed to deliver sequential or parallel injections to two GC instruments using time-scheduled valve switching.
Can SPME fiber aging be performed automatically?
Yes—two dedicated aging stations are included, supporting both manual initiation and scheduled thermal conditioning (0–350 °C, up to 120 min) with programmable ramp rates and hold times.
Is the instrument compatible with third-party SPME fibers?
Yes—it accepts industry-standard 1 cm and 2 cm fiber assemblies from Supelco, Restek, and other ISO 11843-compliant manufacturers via standardized holder mounts.
What level of traceability does the system provide for regulatory submissions?
All temperature, timing, and motion parameters are logged with millisecond resolution and digitally signed; raw logs meet ALCOA+ criteria and are exportable in PDF/A-2b format for submission to health authorities.
How is carryover mitigated during high-concentration sample runs?
The system performs up to 99 automated needle wash cycles per injection using configurable solvent sequences, dwell times, and aspiration volumes, validated to achieve < 0.05% carryover for chlorinated hydrocarbons at 1000 ppm levels.