ANPRO SKJ-IA Food Safety Rapid Quantitative Detection System (Heavy Metals, Mycotoxins & Pesticide Residues)
| Brand | ANPRO |
|---|---|
| Origin | Sichuan, China |
| Manufacturer Type | Manufacturer |
| Product Origin | Domestic (China) |
| Model | SKJ-IA |
| Pricing | Upon Request |
| Function Type | Multi-analyte Quantitative Detection Platform |
| Sample Applicability | Universal for Grains, Feed, Fruits, Vegetables, Tea, Dairy Products |
| Optical Detection Wavelength | 410 nm |
| Assay Time per Sample | 15–30 minutes |
| Coefficient of Variation (CV) | ≤0.2% |
| Detection Channels | 6 independent optical channels |
Overview
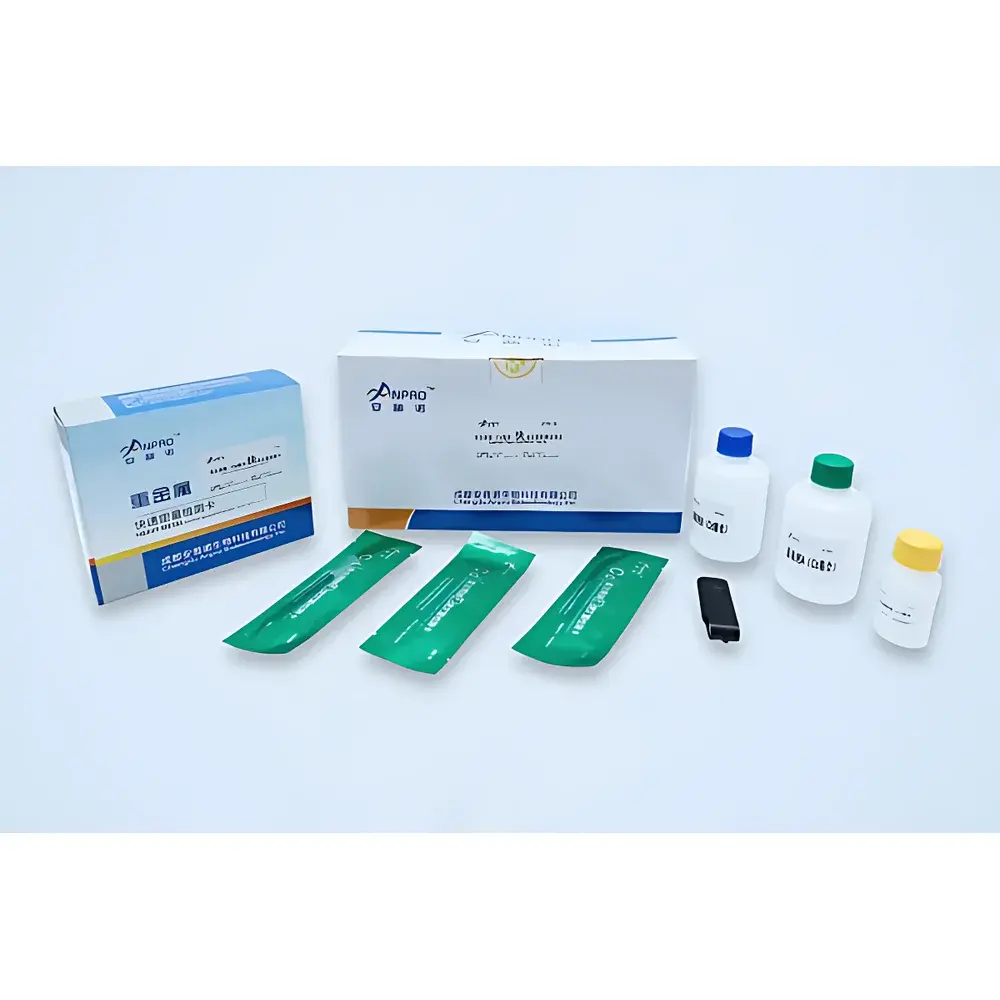
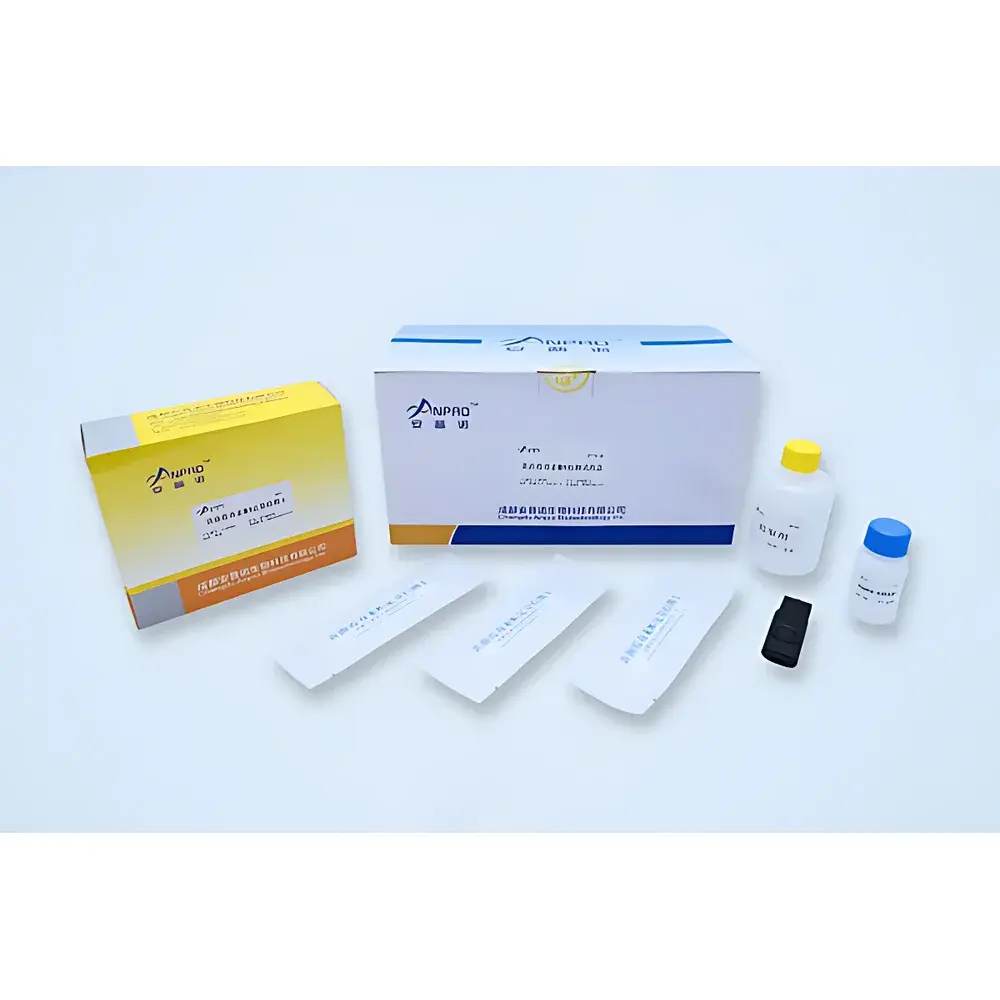
The ANPRO SKJ-IA Food Safety Rapid Quantitative Detection System is an integrated, field-deployable immunoassay platform engineered for simultaneous quantitative analysis of three critical food safety hazard classes: heavy metals (Cd, Pb), mycotoxins (aflatoxin B₁, deoxynivalenol, zearalenone, ochratoxin A, fumonisin B₁, T-2 toxin), and pesticide residues (organophosphates, carbamates, pyrethroids, and specific compounds including chlorpyrifos, carbendazim, and acetamiprid). Unlike conventional qualitative lateral flow assays, this system employs stabilized colloidal gold conjugates with enhanced batch-to-batch reproducibility and signal linearity, enabling true quantitative measurement via reflectance photometry at 410 nm. The instrument operates on the principle of competitive immunochromatographic assay—where analyte concentration inversely correlates with test line intensity—and applies pre-calibrated, instrument-specific calibration curves stored in firmware to convert optical density readings into concentration values (ppb or ppm). Designed for compliance with national grain quality monitoring protocols—including those issued by the National Grain and Oils Information Center (China)—the SKJ-IA supports regulatory-grade decision-making in grain procurement, feed manufacturing, and border inspection contexts.
Key Features
- Six-channel independent optical detection module ensures parallel processing of up to six samples without cross-interference, significantly improving throughput in batch screening workflows.
- Pre-loaded, non-user-adjustable calibration curves eliminate operator exposure to reference standards and reduce inter-operator variability—supporting safe, standardized operation by non-laboratory personnel.
- Integrated GPS-enabled geotagging and timestamping of each assay result, facilitating traceability and alignment with HACCP-based monitoring systems.
- Onboard thermal printer provides immediate hard-copy documentation compliant with internal audit requirements and field reporting needs.
- Robust portable design: all core components—including reader, reagent strips, extraction kits, and accessories—fit within a single shock-resistant carry case; no external power supply or PC required for basic operation.
- Embedded memory capacity ≥20,000 test records with metadata (sample ID, location, operator ID, date/time, assay type, raw signal, calculated concentration).
Sample Compatibility & Compliance
The SKJ-IA platform accepts homogenized extracts from diverse matrices including paddy rice, wheat flour, corn meal, soybean meal, animal feed, green leafy vegetables, apples, tea leaves, and pasteurized milk. Extraction protocols follow AOAC Official Method 2012.01 (for mycotoxins) and GB 5009.12–2023 / GB 5009.15–2023 (for Cd/Pb in grains), ensuring analytical consistency with national regulatory frameworks. All assay kits have undergone technical evaluation and performance verification by the National Grain Quality Supervision and Inspection Center (State Administration of Grain and Strategic Reserves), confirming accuracy, precision, and limit-of-quantitation (LOQ) alignment with statutory thresholds. While not FDA 21 CFR Part 11–certified out-of-the-box, the system’s audit trail functionality (full data logging, immutable timestamps, user authentication logs) supports GMP/GLP-aligned validation when deployed within controlled quality management systems.
Software & Data Management
Data export is supported via USB 2.0 and Bluetooth 5.0 interfaces. Raw and processed results—including chromatographic images, signal intensities, concentration values, and QC flags—are exportable in CSV and XML formats compatible with LIMS platforms (e.g., LabWare, STARLIMS) and national grain safety surveillance networks such as China’s National Grain Monitoring and Early Warning System. Firmware updates are delivered via encrypted OTA packages, maintaining version control and cybersecurity integrity. Custom API integration is available for enterprise-level deployment, enabling real-time dashboard visualization, automated alert generation upon threshold exceedance, and longitudinal trend analysis across regional sampling sites.
Applications
- Grain receiving inspection at elevators and silos—rapid triage of incoming lots prior to storage or milling.
- Feed mill quality assurance—monitoring raw material contamination before formulation and post-production batch release.
- Field-based surveillance by agricultural extension services and food safety regulators during harvest season.
- Emergency response testing during contamination incidents (e.g., aflatoxin outbreaks in maize, lead adulteration in spices).
- Contract laboratory support for certified reference material verification and method transfer studies.
FAQ
Does the SKJ-IA require daily calibration with external standards?
No. The system uses factory-programmed, lot-specific calibration curves embedded in firmware. Each test strip batch is supplied with a QR-coded calibration card scanned during initialization to load corresponding curve parameters.
Can the instrument distinguish between structurally similar mycotoxins (e.g., zearalenone vs. α-zearalenol)?
No. The assay relies on antibody specificity validated for target analytes only. Cross-reactivity data for each kit is provided in the Certificate of Analysis and conforms to ISO/IEC 17025–compliant validation reports.
Is remote data upload possible without cellular connectivity?
Yes. Data can be transferred offline via USB to a central server or uploaded later when Wi-Fi or LTE becomes available—the local database retains full integrity until synchronization.
What is the shelf life of the test strips under field conditions?
When stored at 2–30 °C in sealed aluminum pouches with desiccant, strips maintain performance for ≥18 months. Stability is verified per ICH Q5C guidelines and documented in each kit’s CoA.
Are consumables compatible with other colloidal gold readers?
No. Strip format, membrane composition, and conjugate chemistry are proprietary to the SKJ-IA optical architecture and firmware algorithms; interoperability is not supported.