ANPRO SKJ-IIA Multi-Function Food Safety Rapid Detector
| Brand | ANPRO |
|---|---|
| Origin | Sichuan, China |
| Manufacturer Type | Manufacturer |
| Model | SKJ-IIA |
| Price | ¥80,000 |
| Function Type | Multi-analyte (200+ targets) |
| Sample Applicability | Universal for food, agricultural, aquatic, livestock, and feed samples |
| Wavelengths | 410 nm, 538 nm, 590 nm |
| Detection Time | <15 min per sample |
| Repeatability | ≤0.2% CV |
| Detection Architecture | 3 independent assay zones, 20-channel parallel optical readout |
Overview
The ANPRO SKJ-IIA Multi-Function Food Safety Rapid Detector is an integrated, field-deployable analytical platform engineered for regulatory-grade screening of chemical contaminants, biological hazards, and quality indicators in food supply chains. It operates on three orthogonal detection principles—colloidal gold immunochromatography (lateral flow), spectrophotometry (absorbance at fixed wavelengths), and bioluminescent ATP quantification—enabling concurrent qualitative and quantitative analysis without laboratory infrastructure. Designed for compliance with ISO/IEC 17025 pre-screening workflows and aligned with FDA Bacteriological Analytical Manual (BAM) and EU Commission Regulation (EC) No 882/2004 guidance, the instrument supports decentralized monitoring at ports of entry, processing facilities, mobile inspection units, and rural extension centers. Its architecture eliminates reliance on external computing hardware, integrating a full Android-based control system with embedded GPS geotagging, real-time data synchronization, and audit-trail-capable storage—critical for GLP-aligned field operations and traceability-driven food safety management systems.
Key Features
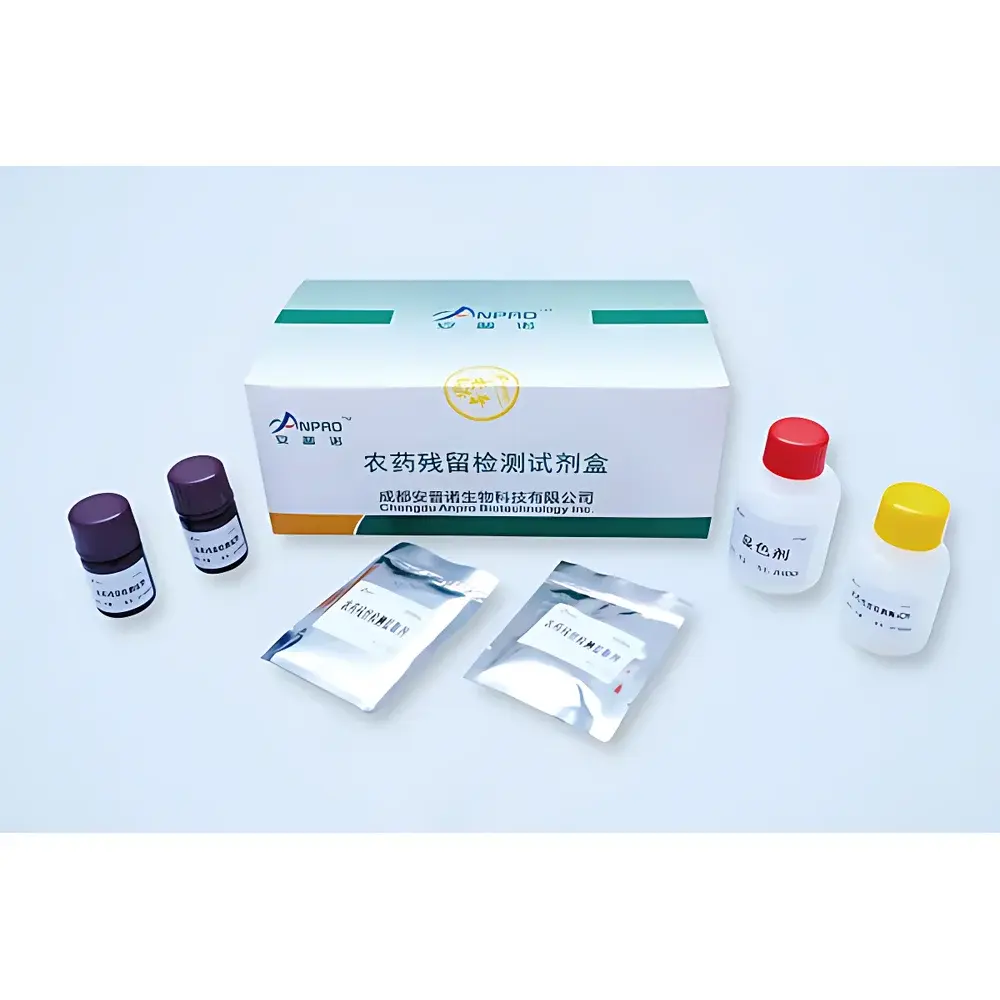
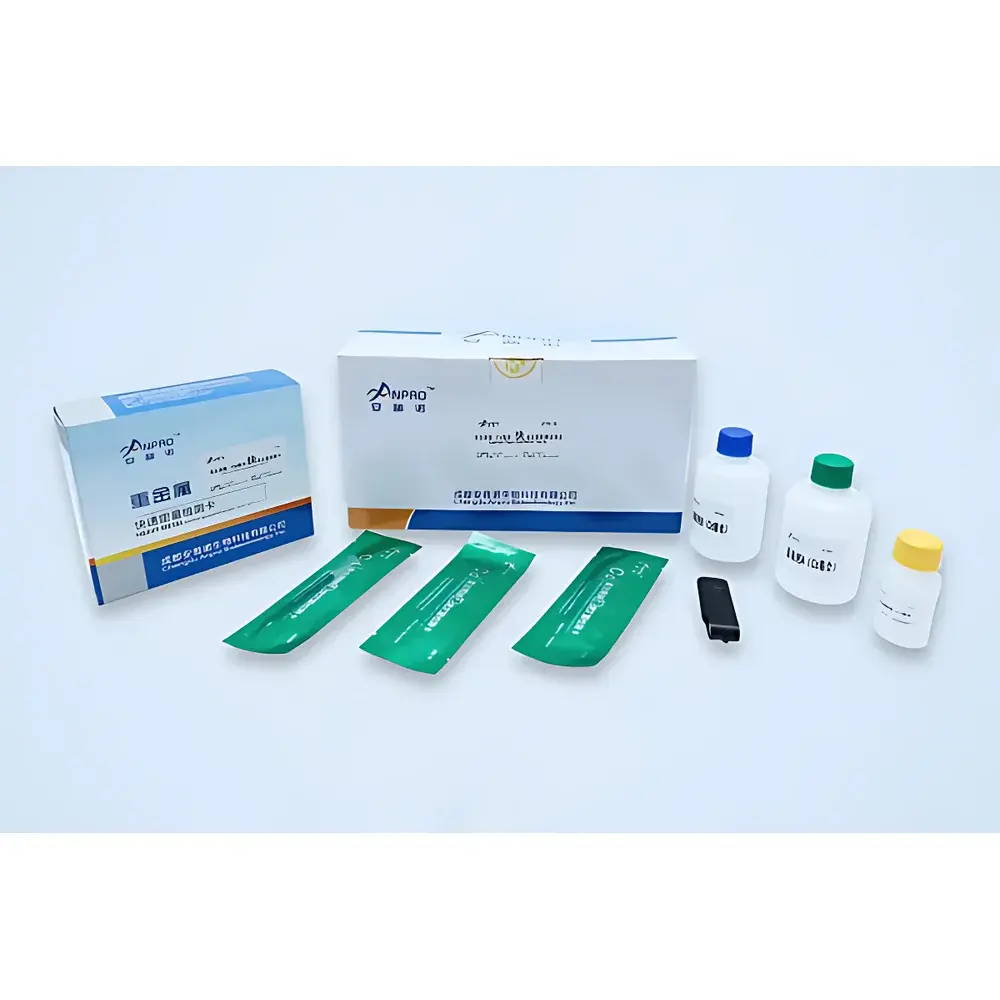
- Triple-mode detection engine: Simultaneous colloidal gold strip scanning (for mycotoxins, antibiotics, hormones), fixed-wavelength photometry (for colorimetric assays including nitrites, formaldehyde, SO₂), and ATP bioluminescence (for surface hygiene validation)
- Self-contained operation: Integrated 10-inch capacitive touchscreen, 4 GB RAM, built-in thermal printer, dual-mode connectivity (Wi-Fi + Ethernet), optional GPS module, and rechargeable Li-ion battery supporting >5 hours continuous use
- Modular assay architecture: Three physically isolated detection zones accommodate simultaneous runs of different assay types; 20-channel optical reader ensures high-throughput batch processing with channel-level calibration traceability
- Regulatory-ready data handling: Automatic timestamping, operator ID logging, assay lot tracking, and result metadata (e.g., LOD, reference range, unit) stored per test; export formats include CSV, PDF, and XML compliant with national food safety information platforms
- Low-power design: Total system power consumption ≤15 W; compatible with 220 V AC, vehicle DC adapters, and portable power stations for off-grid deployment
Sample Compatibility & Compliance
The SKJ-IIA accepts homogenized extracts from diverse matrices—including fresh produce, cereals, dairy, meat, seafood, spices, edible oils, and processed foods—without requiring solvent purification or column cleanup. Sample preparation kits are included in the transport case, enabling standardized extraction protocols per AOAC Official Method guidelines. All optical measurements adhere to Beer–Lambert law assumptions under controlled pathlength conditions, while colloidal gold interpretation follows WHO-recommended visual/semi-quantitative scoring thresholds validated against ELISA reference methods. The system meets technical requirements for rapid screening under China’s GB 5009 series standards and supports alignment with international frameworks including Codex Alimentarius risk-based monitoring and EU Rapid Alert System for Food and Feed (RASFF) reporting templates.
Software & Data Management
Running Android 9.0 with custom firmware, the SKJ-IIA features a role-based user interface supporting multi-level access control (operator, supervisor, administrator). Data integrity is ensured via embedded cryptographic hashing of each record and time-stamped digital signatures compliant with FDA 21 CFR Part 11 Annex 11 principles. Up to 20,000 test records are retained locally with automatic backup to cloud-based dashboards via configurable API endpoints. Integration modules support HL7 v2.x and FHIR R4 message exchange for interoperability with national food safety surveillance networks, provincial inspection databases, and enterprise quality management systems (QMS). Audit logs capture all configuration changes, calibration events, and result modifications with immutable timestamps.
Applications
- Routine surveillance of pesticide residues (organophosphates, carbamates) in fruits and vegetables using acetylcholinesterase inhibition assays
- Quantitative detection of adulterants including melamine in liquid dairy, methanol in distilled spirits, and borax in rice noodles
- Screening for mycotoxins (aflatoxin B₁, deoxynivalenol, zearalenone) in grains and oilseeds via lateral flow immunoassays
- Verification of preservative levels (sorbic acid, benzoic acid) and spoilage markers (peroxide value, acid value) in edible oils and sauces
- Hygiene verification of food contact surfaces using ATP bioluminescence with RLU-to-CFU correlation curves established per ISO 22000 Annex H
- Field validation of veterinary drug residues (clenbuterol, salbutamol, ractopamine) in muscle tissue and liver samples
FAQ
Does the SKJ-IIA require external calibration standards for routine operation?
No—each assay module includes factory-calibrated reference strips and wavelength-specific optical filters. User-performed verification uses supplied control solutions prior to daily use, documented within the electronic logbook.
Can test results be exported in formats compatible with LIMS or national food safety portals?
Yes—CSV, PDF, and XML exports include structured metadata fields (sample ID, location coordinates, assay type, LOD, result status) and support automated ingestion into regional food safety information systems.
Is the device certified for use in official inspection programs?
The SKJ-IIA complies with technical specifications outlined in China’s JJG (Food) 001–2021 metrological verification regulation for rapid food safety detectors and is listed in the National Market Supervision Administration’s Recommended Equipment Catalog for Grassroots Inspection Units.
What maintenance is required for long-term operational reliability?
Optical components undergo quarterly self-diagnostic routines; battery health is monitored via embedded BMS; firmware updates are delivered over-the-air and include version-controlled change logs traceable to ISO/IEC 17025 clause 5.9.4.
How is measurement uncertainty addressed across different assay modalities?
Uncertainty budgets are preloaded per assay type based on inter-laboratory validation studies (n ≥ 30) and reflect contributions from pipetting variability, matrix interference, and photometric noise—reported alongside each quantitative result in the output file.