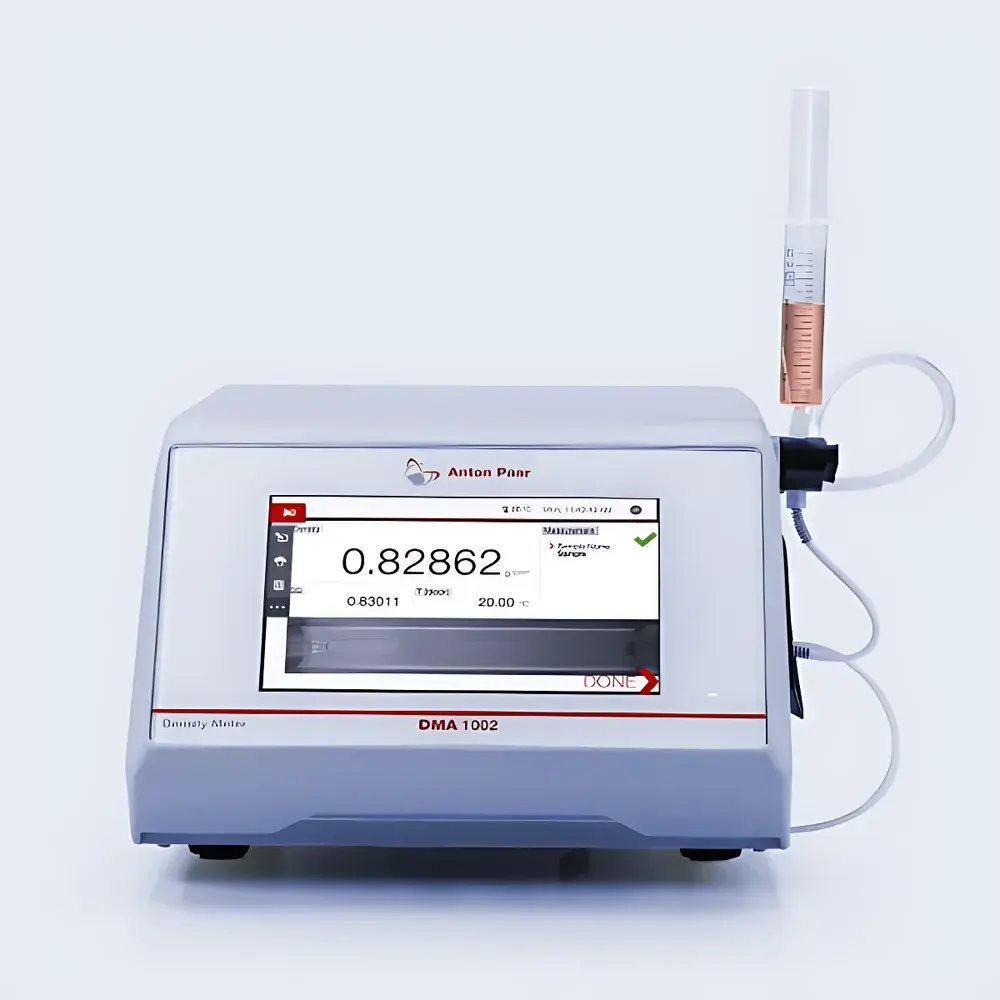
Anton Paar DMA 1002 Digital Oscillating U-Tube Density Meter
| Brand | Anton Paar |
|---|---|
| Origin | Austria |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Origin Category | Imported Instrument |
| Model | DMA 1002 |
| Instrument Type | Oscillating U-tube Densimeter |
| Form Factor | Benchtop Laboratory Instrument |
| Sample Type | Liquid |
| Density Range | 0–3 g/cm³ |
| Density Accuracy | ±0.0001 g/cm³ |
| Density Repeatability | ±0.00005 g/cm³ |
| Density Resolution | 0.00001 g/cm³ |
| Temperature Range | 15–60 °C |
| Temperature Stability | ±0.02 °C |
| Minimum Sample Volume | 1 mL |
| Display | 7″ TFT WVGA Touchscreen (800 × 480 px, PCAP) |
Overview
The Anton Paar DMA 1002 is a high-precision benchtop digital density meter based on the oscillating U-tube principle—a well-established, internationally standardized method for liquid density determination (ASTM D4052, ASTM D5002, ISO 12185, USP , Ph. Eur. 2.2.39). In this technique, a precisely machined U-shaped glass tube—filled with the sample—is electromagnetically excited to oscillate at its natural resonant frequency. Since the oscillation period is directly related to the mass of the sample and inversely proportional to its density, the instrument calculates density with exceptional reproducibility and minimal operator dependency. Engineered for routine laboratory environments, the DMA 1002 delivers four-decimal-place resolution (0.00001 g/cm³) and meets stringent regulatory requirements including FDA 21 CFR Part 11 compliance when integrated with AP Connect software. Its compact footprint, robust borosilicate glass measurement cell, and PTFE/ETFE-wetted materials ensure long-term stability and chemical compatibility across diverse industrial sectors—from pharmaceuticals and food & beverage to petrochemicals and fine chemicals.
Key Features
- Four-digit density resolution (0.00001 g/cm³) and repeatability of ±0.00005 g/cm³ (standard deviation) under controlled conditions
- Integrated U-View imaging system: real-time, zoomable visual monitoring of the U-tube during filling and measurement to detect microbubbles or incomplete filling
- FillingCheck function: automated bubble detection within seconds—critical for high-accuracy applications where entrapped air compromises result integrity
- Intuitive 7″ capacitive touchscreen interface (800 × 480 px) with glove-compatible operation and multilingual support
- Automatic environmental compensation: built-in barometric pressure and humidity sensors adjust for altitude and ambient fluctuations
- Flexible sample introduction: manual syringe filling, Xsample 200 automated peristaltic pump, or gravity-fed funnel—each optimized for safety, speed, and contamination control
- Onboard temperature control (15–60 °C) with ±0.02 °C stability and programmable ramping for temperature-dependent density profiling
Sample Compatibility & Compliance
The DMA 1002 accommodates liquids ranging from low-viscosity solvents (e.g., ethanol, water, hydrocarbons) to moderately viscous formulations (up to ~1000 mPa·s with optional viscosity correction algorithms). Its 1 mL minimum sample volume reduces reagent consumption and waste—particularly advantageous in QC labs handling high-value or hazardous materials. All wetted parts—including the U-tube, capillary, and valve seals—are constructed from chemically inert borosilicate glass, PTFE, and ETFE, ensuring resistance to acids, bases, and organic solvents. The instrument complies with ISO/IEC 17025 traceability requirements when used with certified reference materials and supports full audit trails, electronic signatures, and data integrity protocols required under GLP, GMP, and FDA 21 CFR Part 11 frameworks. Built-in conversion tables (140+), concentration libraries (400+ validated methods), and API gravity calculation align with ASTM D1250, D1298, and IP 200 standards.
Software & Data Management
The DMA 1002 operates natively via its embedded Linux-based firmware but achieves full regulatory compliance and enterprise integration through Anton Paar’s AP Connect laboratory execution system. AP Connect provides centralized data storage, role-based user management, secure electronic signatures, and immutable audit trails—including timestamps for all measurements, calibrations, and parameter changes. Measurement results (up to 5000 entries) are stored locally with metadata (operator ID, method name, temperature, pressure, lot number), and can be exported in CSV, PDF, or XML formats compliant with LIMS and ERP systems. Optional ISO 17025-compliant calibration certificates and printer modules enable paper-based reporting without compromising digital traceability. Firmware updates are delivered via USB or Ethernet, ensuring continuous alignment with evolving metrological best practices.
Applications
- Pharmaceutical QC: excipient density verification, solvent purity assessment, and formulation density consistency checks per USP
- Food & Beverage: alcohol content (ethanol/water), sugar concentration (Brix), and extract density in juices, wines, and syrups
- Petrochemicals: API gravity, distillate density grading, and blend verification per ASTM D1298 and D4052
- Chemical Manufacturing: acid/base concentration monitoring (H₂SO₄, NaOH), polymer solution density, and reaction mixture tracking
- Academic Research: thermophysical property studies, solvent mixture characterization, and density-derived partial molar volume calculations
FAQ
What is the minimum sample volume required for accurate measurement?
The DMA 1002 requires only 1 mL of sample—optimized for laboratories prioritizing material conservation and rapid throughput.
Does the instrument support automated sample handling?
Yes—via optional Xsample 200 peristaltic pump module, enabling unattended sequential analysis of up to 20 samples with programmable rinse cycles.
Can the DMA 1002 measure viscous or non-Newtonian liquids?
It is primarily designed for Newtonian liquids; however, viscosity correction algorithms and dedicated high-viscosity measurement modes (with appropriate calibration) extend usability to fluids up to ~1000 mPa·s.
How does the instrument ensure compliance with FDA 21 CFR Part 11?
When paired with AP Connect software, it enforces electronic signature workflows, audit trail generation, and data immutability—fulfilling ALCOA+ principles for regulated environments.
Is external calibration necessary before first use?
No—the instrument ships pre-calibrated using NIST-traceable standards; users perform simple one-point verification with air and water prior to routine operation.