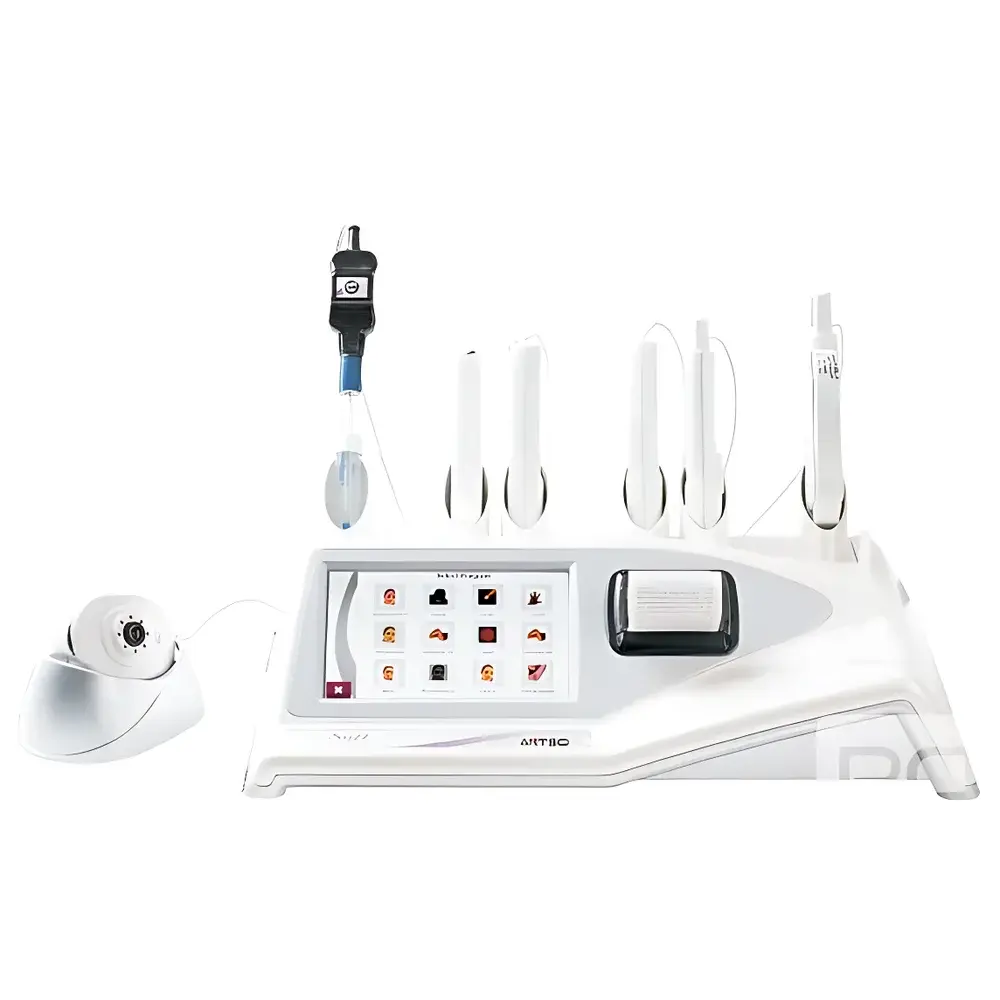
ANTSCI Skin-SP Multi-Parameter Cutaneous Analysis System
| [Brand | ANTSCI |
|---|---|
| Origin | Italy |
| Model | Skin-SP |
| Probe Count | 6 external + 1 integrated sebum sensor |
| Moisture Range | 0–100 u.c., ±5% |
| Sebum Range | 0–100 u.c., ±5% |
| pH Range | 2–12, ±1% |
| Melanin Range | 0–100, ±5% |
| Elasticity Range | 0–50, ±5% |
| Temperature Range | 20–40 °C, ±0.1 °C |
| Measurement Time | 4–10 s |
| Display | 7″ color LCD touch screen |
| Printer | thermal, 34-column report output |
| Power | 12 V DC / 3.5 A, max. 60 W |
| Dimensions | 450 × 260 × 120 mm |
| Weight | 2.8 kg |
| Compliance | CE] |
Overview
The ANTSCI Skin-SP Multi-Parameter Cutaneous Analysis System is a CE-certified, portable clinical-grade instrument engineered for objective, non-invasive assessment of six key biophysical skin parameters: hydration (capacitance-based), sebum secretion (reflectometric), melanin index (dual-wavelength reflectance photometry at 660 nm and 875 nm), elasticity (mechanical pressure/deformation response), surface pH (dual-junction electrode), and local skin temperature (infrared thermometry). Designed for dermatological research, cosmetic efficacy evaluation, and professional aesthetic clinics, the Skin-SP implements standardized measurement protocols aligned with ISO 24442 (in vitro determination of sunscreen UVA protection), ISO 24444 (in vivo SPF testing methodology), and ASTM E2958 (standard guide for instrumental assessment of skin barrier function). Its modular probe architecture enables repeatable, site-specific acquisition under controlled ambient conditions (15–30 °C, 20–80% RH), supporting longitudinal monitoring and statistical comparison across cohorts.
Key Features
- Integrated 7-inch high-resolution color LCD touch interface with on-screen step-by-step procedural guidance — no external PC required
- Dedicated thermal printer generating standardized 34-column analytical reports including reference norms, measured values, deviation indices, and timestamped metadata
- Universal probe holder accommodating six interchangeable, ergonomically designed ABS-bodied probes (moisture, sebum, melanin, elasticity, temperature, pH) plus one built-in sebum sensor
- Illuminated capacitive touch stylus minimizing fingerprint residue and enhancing precision input in clinical environments
- USB-powered digital dermoscopic camera (1/4″ CMOS, USB 3.0 output) with adjustable focus, polarized/vertical/side illumination modes, and fixed white LED array (8×)
- Compact footprint (450 × 260 × 120 mm) and lightweight chassis (2.8 kg) enabling benchtop deployment or mobile cart integration
- Compliance with EU Directive 2014/30/EU (EMC) and 2014/53/EU (Radio Equipment), certified per EN 60601-1 (medical electrical equipment safety)
Sample Compatibility & Compliance
The Skin-SP supports quantitative analysis across anatomical regions including facial zones (forehead, cheek, nasolabial fold), dorsal hand, volar forearm, scalp, and post-treatment lesion sites. Each probe is calibrated for epidermal stratum corneum interaction — moisture and sebum probes require direct contact; melanin and elasticity probes apply controlled mechanical coupling; pH electrodes necessitate brief (<5 s) contact with cleansed, dry skin. All measurements adhere to Good Clinical Practice (GCP) documentation standards, with audit-trail-capable data export (CSV, PDF) compatible with FDA 21 CFR Part 11–compliant LIMS integration. Device firmware and calibration certificates are traceable to NIST-traceable reference standards. Storage and transport conditions (0–50 °C, 10–90% RH non-condensing) ensure metrological stability between deployments.
Software & Data Management
The embedded operating system provides real-time visualization of all six parameters simultaneously or individually, with configurable pass/fail thresholds relative to age- and gender-matched normative databases (preloaded per ISO/TR 24447). Data is stored locally in encrypted binary format and exportable via USB port in CSV (for SPSS, R, or Python analysis) or printer-ready PDF. The system logs operator ID, test location, environmental conditions, probe serial number, and calibration status for each session — satisfying GLP audit requirements. Firmware updates are delivered via signed .bin files; no cloud dependency or third-party telemetry is implemented. Camera images are timestamped and geotagged (if enabled via external GPS module), supporting longitudinal image overlay and change-detection algorithms.
Applications
- Cosmetic formulation development: quantifying moisturizer-induced hydration kinetics, anti-sebum agent efficacy, and pigment-modulating ingredient performance over 28-day cycles
- Dermatological clinical trials: objective endpoint assessment in atopic dermatitis (TEWL correlation), rosacea (erythema/melanin differentiation), and photoaging studies (elasticity decline rate)
- Aesthetic clinic workflows: pre- and post-procedure benchmarking for chemical peels, microneedling, RF lifting, and laser resurfacing
- Sun exposure monitoring: UV-induced melanin response tracking using the dedicated solar simulation protocol
- Regulatory dossier preparation: generation of ISO-compliant measurement reports for CPNP notifications and Health Canada submissions
- Educational laboratories: hands-on training in biophysical skin property interrelationships and instrument validation methodology
FAQ
Is the Skin-SP compliant with FDA or Health Canada regulatory requirements for clinical use?
The device carries CE marking under MDD 93/42/EEC as a Class I medical device. While not FDA-cleared, its measurement methodologies align with FDA-recognized consensus standards (e.g., ISO 24444 for SPF testing); users must validate protocols per their institutional IRB or regulatory pathway.
Can measurement data be exported to electronic health record (EHR) systems?
Yes — CSV exports include structured fields (patient ID, timestamp, parameter values, units, uncertainty estimates) compatible with HL7 FHIR ingestion pipelines when configured via middleware.
What is the recalibration interval for the pH electrode probe?
ANTSCI recommends recalibration before each daily use using two-point buffer verification (pH 4.01 and 7.00); full probe recalibration is required every 90 days or after 200 measurements, documented in the internal service log.
Does the system support multi-language UI beyond English?
The firmware includes localized UI overlays for English, German, French, Spanish, and Simplified Chinese — selectable via Settings > Language without firmware update.
How is probe cross-contamination prevented during high-throughput testing?
Each probe features disposable polyethylene caps (included in consumables kit ANTSCI-CAP-6); the system enforces mandatory cap detection via optical sensor prior to measurement initiation.