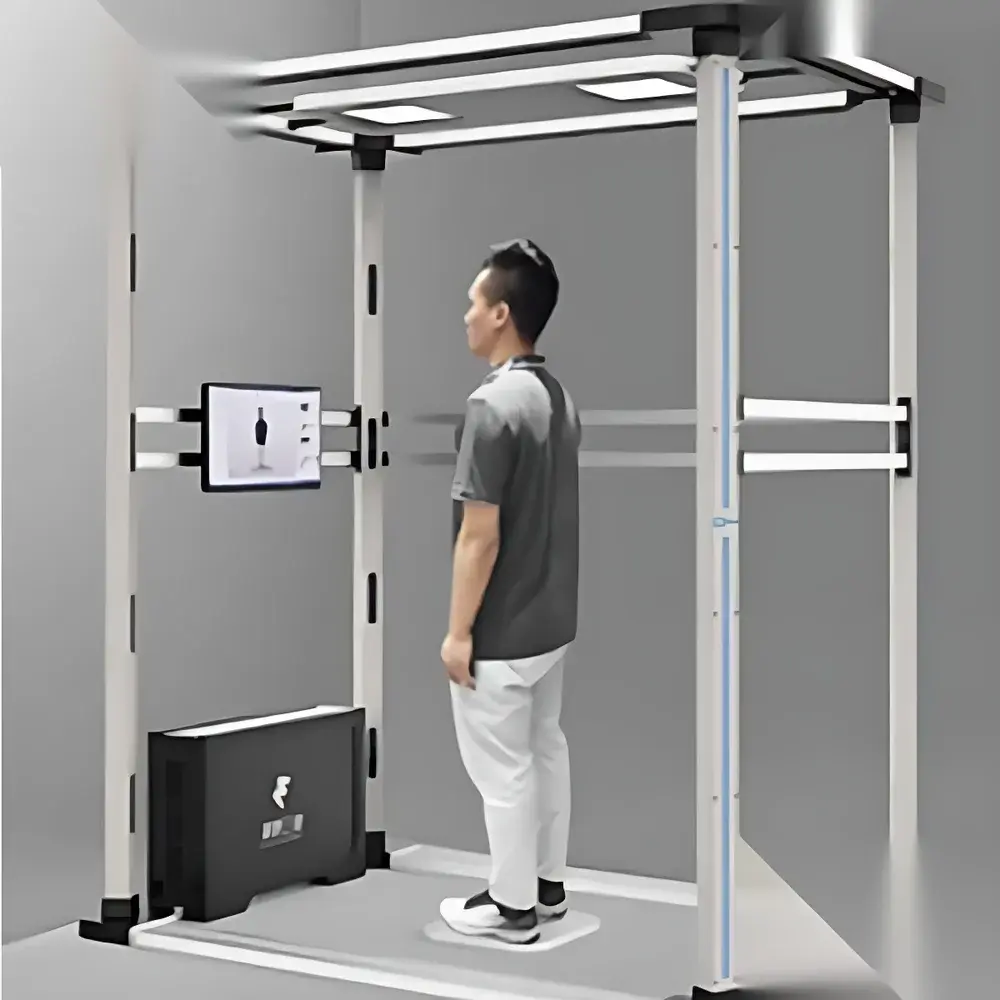
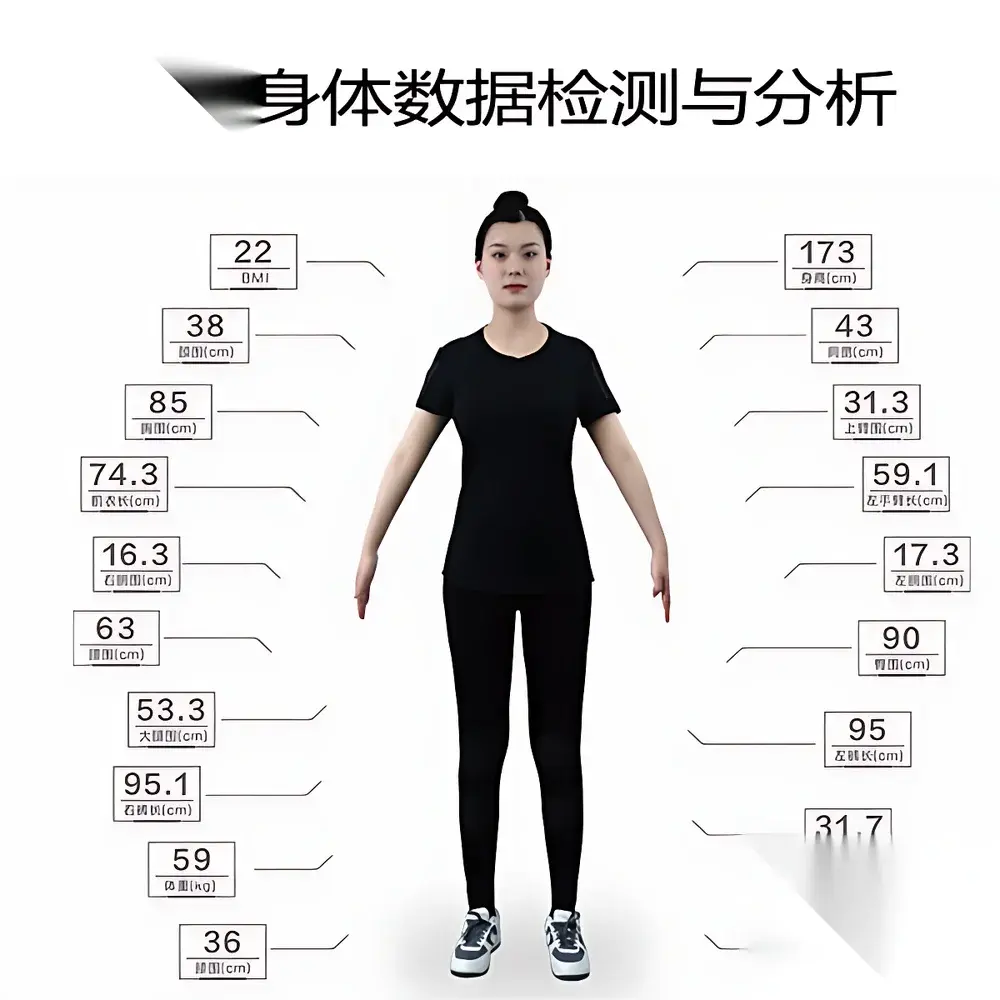
Artec BodyScan Smart 3D Full-Body Scanning System
| Brand | Artec |
|---|---|
| Origin | Imported |
| Manufacturer Type | Authorized Distributor |
| Quotation | Upon Request |
| Depth Resolution | 1280×1024 @ 7 fps / 640×480 @ 30 fps / 320×240 @ 30 fps |
| Color Resolution | 1920×1080 |
| Depth Range | 0.6–2 m |
| Measurement Time | 2 s |
| Measurement Volume | 1200 mm × 1200 mm × 2000 mm (L×W×H) |
| Point Density | 120 pts/cm² |
| Depth of Field | 800 mm |
| Accuracy | ±1 mm |
| Operating Voltage | AC 220 V |
| Minimum Installation Footprint | 1.5 m × 2 m |
Overview
The Artec BodyScan Smart 3D Full-Body Scanning System is a non-contact, infrared-based optical measurement instrument engineered for high-fidelity, sub-millimeter-resolution 3D anthropometric acquisition. Leveraging structured-light-assisted time-of-flight infrared sensing and synchronized multi-angle depth capture, the system reconstructs complete human surface geometry—including topology, curvature, and spatial proportions—within two seconds. Unlike traditional tape-based or caliper-driven anthropometry, BodyScan eliminates inter-operator variability, physical contact artifacts, and subjective landmark identification. Its core architecture complies with ISO 20685:2010 (Anthropometric procedures for the assessment of body dimensions), enabling traceable, repeatable, and statistically robust body shape quantification. Designed for integration into clinical, commercial, and industrial workflows, it serves as a foundational data acquisition layer for digital twin generation, biomechanical modeling, and personalized product development.
Key Features
- Quad-sensor modular array: Four synchronized infrared depth cameras mounted at orthogonal azimuthal positions (front, left, right, rear) ensure full circumferential coverage without occlusion gaps.
- Real-time point cloud registration: Proprietary automatic co-registration algorithm aligns multi-view scans into a unified coordinate frame using iterative closest point (ICP) refinement and feature-based alignment—no manual post-processing required.
- True-color texture mapping: Optional RGB overlay integrates high-definition 1920×1080 color imaging with geometric data, preserving skin tone, garment texture, and anatomical contrast for visualization and downstream rendering.
- Robust dark-surface handling: Advanced infrared illumination modulation and adaptive exposure control enable reliable capture of low-reflectivity surfaces—including black hair, matte fabrics, and synthetic textiles—addressing a known limitation in consumer-grade 3D scanning.
- Integrated anthropometric engine: Preconfigured measurement library computes 120+ standardized body dimensions (e.g., bust circumference, waist-to-hip ratio, shoulder slope angle, leg length asymmetry) per scan, compliant with ASTM D5585 and ISO 8559 anthropometric definitions.
- Modular deployment architecture: Self-contained upright column design supports rapid installation in retail environments, clinics, fitness centers, or research labs; requires only standard AC 220 V power and minimal floor space (1.5 m × 2 m).
Sample Compatibility & Compliance
BodyScan accommodates standing adult subjects (height range: 120–200 cm; weight capacity: ≤150 kg) wearing standard clothing—including layered garments and accessories—without requiring pose correction or marker placement. It meets electromagnetic compatibility (EMC) requirements per IEC 61326-1 and laser safety Class 1 compliance (IEC 60825-1), ensuring safe operation in public-facing locations. All measurement outputs are timestamped and digitally signed to support auditability under GLP and GMP frameworks. Data export formats include OBJ, STL, PLY, and CSV—enabling interoperability with CAD platforms (e.g., Autodesk Fusion 360), PLM systems, and FDA-regulated health informatics pipelines (e.g., DICOM-SR extensions for clinical morphology tracking).
Software & Data Management
The proprietary BodyScan Studio software provides a validated, FDA 21 CFR Part 11–ready environment for acquisition, analysis, and archival. Role-based access control (RBAC), electronic signatures, and immutable audit trails log every scan event—including operator ID, calibration status, environmental metadata (ambient temperature/humidity), and raw sensor diagnostics. Batch processing supports automated report generation in PDF/HTML with customizable templates aligned to brand-specific KPIs (e.g., fit deviation indices, growth percentile tracking). Cloud synchronization (AWS-hosted optional module) enables secure, encrypted transmission of de-identified datasets for federated learning applications in apparel sizing optimization or longitudinal health monitoring—fully compliant with GDPR and HIPAA data minimization principles.
Applications
- Fashion & Apparel: Automated size recommendation engines, virtual try-on integration (WebGL/Unity), made-to-measure pattern grading, and real-time fit analytics for e-commerce returns reduction.
- Clinical Research: Longitudinal monitoring of scoliosis progression, post-bariatric surgery morphological changes, lymphedema volumetric assessment, and prosthetic socket fitting validation.
- Wellness & Fitness: Objective baseline profiling for personal training programs, posture analysis via skeletal joint angle derivation, and metabolic risk stratification using waist-to-height ratio trends.
- Digital Content Creation: High-fidelity avatar generation for gaming, film VFX, and metaverse avatars; archival scanning for heritage preservation (e.g., dance movement documentation).
- Industrial Design: Ergonomic workstation evaluation, PPE (personal protective equipment) customization, and anthropometric database enrichment for automotive seating and aerospace cabin design.
FAQ
What is the minimum recommended computing configuration for real-time reconstruction?
A Windows 10/11 64-bit system with Intel Core i7-8700K or equivalent, 32 GB RAM, NVIDIA GeForce RTX 3060 or higher GPU, and ≥512 GB NVMe SSD is required for sub-30-second mesh reconstruction and interactive model navigation.
Does BodyScan support integration with existing ERP or PLM systems?
Yes—via RESTful API and configurable CSV/XML export schemas. Pre-built connectors are available for SAP S/4HANA, Oracle Agile PLM, and Centric PLM; custom middleware development is supported under NDA.
How is measurement traceability maintained across multiple devices and sites?
Each unit undergoes factory calibration against NIST-traceable anthropometric phantoms. On-site verification kits and quarterly self-diagnostic routines generate calibration certificates compliant with ISO/IEC 17025 requirements.
Can the system operate in ambient lighting conditions typical of retail stores?
Yes—the infrared sensors are spectrally filtered to reject visible-light interference; performance remains stable under illuminance up to 10,000 lux (equivalent to direct daylight through skylights).
Is patient or user consent management built into the software?
Yes—integrated digital consent workflow includes versioned text display, biometric signature capture, and opt-in/opt-out toggles for data sharing, fully aligned with GDPR Article 7 and CCPA Section 1798.100 requirements.