ATAGO PAL-03S Clinical & Sports Digital Pocket Refractometer
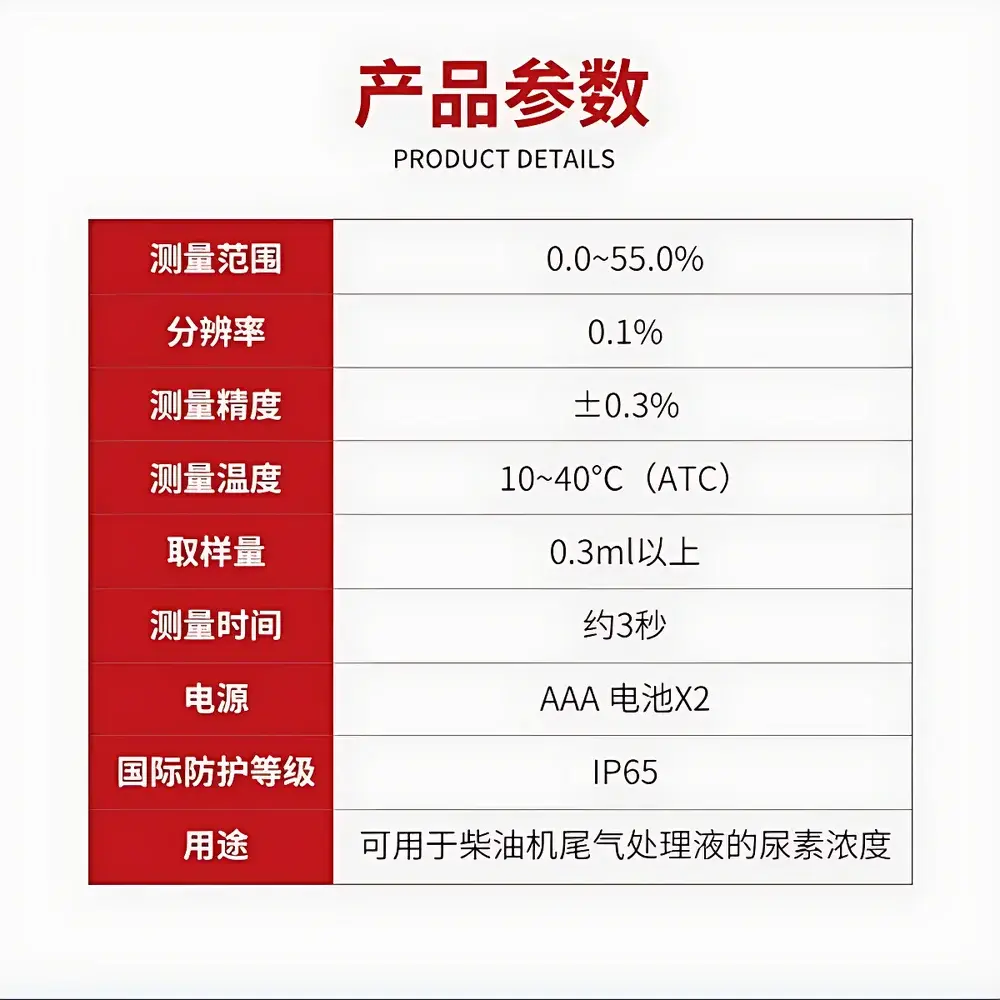
| Brand | ATAGO |
|---|---|
| Origin | Japan |
| Model | PAL-03S (Clinical & Sports Series) |
| Type | Handheld Digital Refractometer |
| Measurement Range | 0.0–45.0% (w/w NaCl equivalent) |
| Accuracy | ±0.4% |
| Temperature Control | None |
| Display | Digital LCD |
Overview
The ATAGO PAL-03S is a compact, handheld digital refractometer engineered for rapid, on-site quantification of sodium chloride (NaCl) concentration in clinical and sports-related biological fluids—primarily human sweat, oral rehydration solutions, and isotonic saline preparations. It operates on the principle of critical-angle refractometry: incident light undergoes total internal reflection at the prism-sample interface, with the resulting shadow line position directly correlated to the sample’s refractive index. This index is converted in real time to mass percent concentration (% w/w) using a factory-calibrated NaCl-specific algorithm traceable to JIS Z 8013 and ISO 21748 standards. Designed for field use in athletic training facilities, physical therapy clinics, and point-of-care environments, the PAL-03S delivers repeatable measurements without external power sources beyond its built-in battery, eliminating dependency on lab infrastructure.
Key Features
- Prism-based optical system with high-stability sapphire-coated measurement surface, resistant to abrasion and chemical degradation from repeated contact with biological samples.
- Digital LCD display with automatic backlighting for low-light conditions; resolution of 0.1% across full 0.0–45.0% range.
- Auto-zero function activated via START ZERO button—eliminates baseline drift caused by ambient temperature fluctuations or prism residue between measurements.
- Compact ergonomic housing (130 × 60 × 35 mm), IP65-rated for dust and water resistance during active field deployment.
- Battery-powered operation (2 × AAA alkaline cells) supporting >5,000 measurements per set; low-battery indicator ensures data continuity.
- No moving parts or consumables—maintenance limited to periodic prism cleaning with lens tissue and distilled water.
Sample Compatibility & Compliance
The PAL-03S is validated for aqueous NaCl solutions within physiological and exercise-relevant ranges (0–28 g/100 g, equivalent to 0.0–45.0%). It accommodates viscous or particulate-laden samples such as unfiltered sweat, provided volume exceeds 0.3 mL and surface tension permits uniform prism coverage. While not intended for serum or urine electrolyte profiling (which require ion-selective electrodes or ICP-MS), it meets ASTM D1152–19 requirements for refractometric salt concentration verification in non-viscous aqueous media. Device firmware and calibration certificates comply with ISO/IEC 17025 traceability frameworks through ATAGO’s Tokyo Calibration Laboratory (JCSS-accredited). No regulatory submission (e.g., FDA 510(k), CE-IVD) applies, as the instrument is classified as a general-purpose analytical tool—not an in vitro diagnostic device under IVDR Annex XVI.
Software & Data Management
The PAL-03S operates as a standalone instrument with no embedded memory or Bluetooth/Wi-Fi connectivity. All measurements are displayed in real time only; no internal storage or export capability is provided. For documentation purposes, users may manually record values alongside contextual metadata (e.g., subject ID, collection time, environmental temperature) in accordance with GLP-compliant logbooks. When integrated into institutional workflows, the device supports audit-ready manual entry into LIMS or electronic health records (EHRs) compliant with 21 CFR Part 11—provided user-defined procedures include secondary verification, timestamp annotation, and reviewer sign-off.
Applications
- Sweat sodium analysis: Quantifying NaCl loss during endurance exercise to inform individualized hydration and electrolyte replacement strategies.
- Clinical hydration assessment: Supporting differential diagnosis in cases of suspected hyponatremia or hypernatremia where rapid screening precedes confirmatory serum testing.
- Sports medicine quality control: Verifying concentration accuracy of pre-packaged oral rehydration salts (ORS) and intravenous saline solutions prior to administration.
- Research field studies: Enabling longitudinal monitoring of electrolyte dynamics in ambulatory cohorts without requiring centrifugation or refrigerated transport.
- Educational laboratories: Demonstrating fundamental principles of solution thermodynamics and optical property–concentration relationships in physiology curricula.
FAQ
Is the PAL-03S suitable for measuring serum sodium concentration?
No. Serum requires precise ion-selective detection due to protein interference and matrix effects; this device measures total dissolved solids via refractive index and is calibrated exclusively for NaCl in aqueous buffer.
Does the instrument require temperature compensation?
It incorporates a built-in thermistor for ambient temperature sensing and applies algorithmic correction based on JIS Z 8013 reference curves—but does not regulate sample temperature. Measurements should be performed at 10–40°C for optimal accuracy.
Can it be recalibrated by the end user?
Calibration is factory-set and non-adjustable. Users perform daily verification using ATAGO-certified 0.0% and 15.9% NaCl standard solutions (sold separately); deviation >±0.4% warrants service return.
What is the expected service life under routine clinical use?
With proper prism cleaning and battery replacement, mechanical and optical performance remains stable for ≥5 years, consistent with ATAGO’s published MTBF specifications for PAL-series instruments.