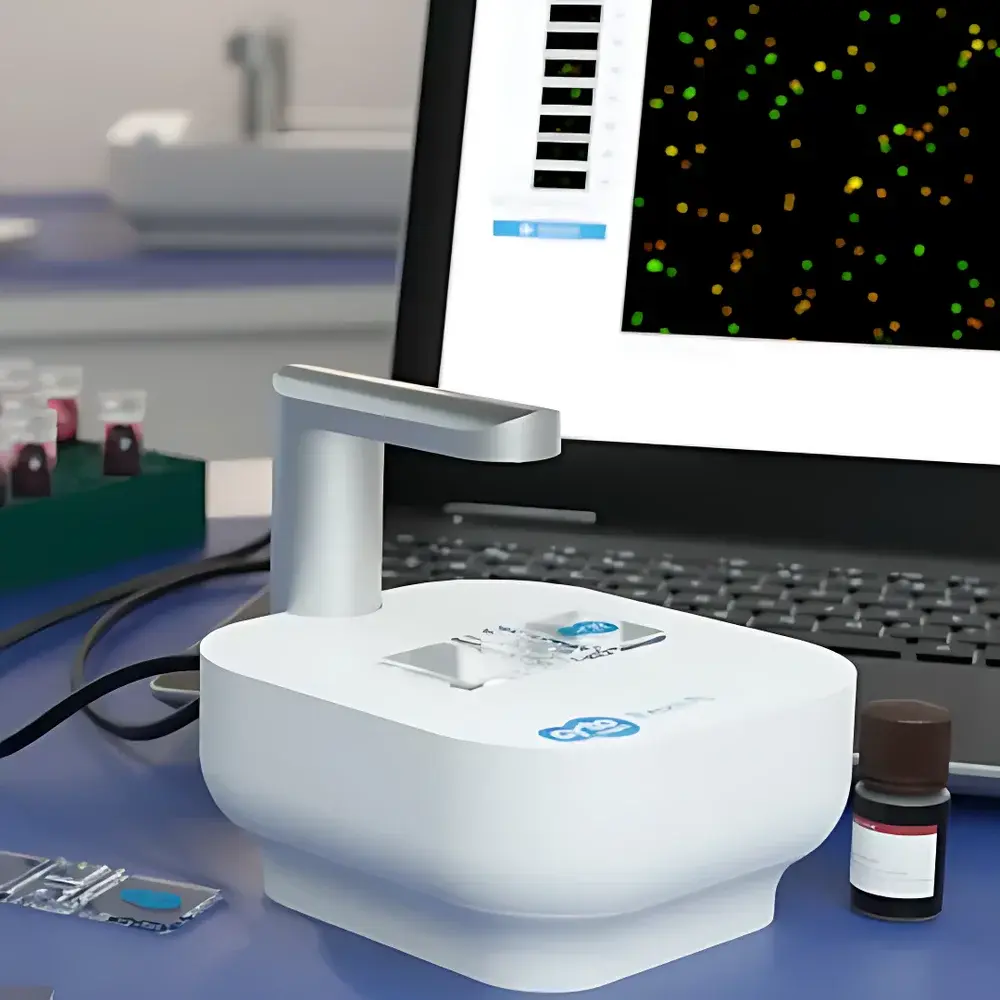
Axion Exact FL Automated Brightfield/Fluorescence Cell Counting Station
| Brand | Axion BioSystems |
|---|---|
| Origin | Netherlands |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Imported Instrument |
| Model | Axion Exact FL |
| Pricing | Available Upon Request |
Overview
The Axion Exact FL Automated Brightfield/Fluorescence Cell Counting Station is a benchtop, AI-powered cell analysis platform engineered for high-precision quantification of 2D monolayer cultures and 3D multicellular structures—including spheroids, organoids, and microtissues. It employs a high-resolution CMOS imaging system coupled with dual-channel fluorescence excitation (470 nm blue LED for green fluorescence; 590 nm amber LED for red fluorescence) and optimized brightfield illumination to capture morphologically rich images across multiple modalities. Unlike conventional hemocytometer-based or flow cytometry–derived counting methods, the Exact FL implements a proprietary deep learning–enhanced image segmentation algorithm trained on diverse primary and immortalized cell types. This enables robust discrimination between intact viable cells, non-viable cells, cellular debris, and clustered aggregates—without reliance on manual gating or threshold tuning. The system operates on the principle of pixel-level object classification and spatial density mapping, delivering statistically validated concentration (cells/mL), viability (%), average diameter (µm), and aggregate count per field—traceable to NIST-traceable reference standards via optional calibration workflows.
Key Features
- Automated dual-mode acquisition: Simultaneous or sequential brightfield and dual-color (green/red) fluorescence imaging in a single workflow
- AI-driven segmentation engine: Trained on >10⁶ annotated cell images; supports adaptive recognition of irregular morphology, partial occlusion, and low-contrast boundaries
- Wide-field imaging: 2.3 mm × 1.7 mm field of view at 10× effective magnification; resolution < 2.5 µm/pixel
- Flexible chamber compatibility: Accepts reusable glass or disposable plastic counting chambers with 0.1 mm depth—no etched grid required
- No optical calibration needed: Factory-aligned optics and LED intensity stabilization ensure inter-unit reproducibility (CV < 3.2% across n = 48 replicate runs)
- Integrated environmental shielding: Includes magnetic-fit light-blocking plate to suppress ambient photonic noise during fluorescence acquisition
Sample Compatibility & Compliance
The Exact FL accommodates standard suspension samples stained with trypan blue (brightfield viability), acridine orange/propidium iodide (AO/PI), calcein-AM/ethidium homodimer-1 (Live/Dead), or any fluorophore pair emitting within 500–550 nm (green) and 600–650 nm (red) detection bands. It is validated for use with adherent cell dissociates, PBMCs, stem cell-derived organoids (diameter range: 20–300 µm), and tumor spheroids. All firmware and software modules comply with ISO 13485:2016 design controls for IVD-related instrumentation. Data integrity features include audit trail logging (per FDA 21 CFR Part 11 requirements), user role–based access control, and encrypted local storage prior to optional CytoSMART Cloud upload. The system supports GLP/GMP-aligned documentation packages including IQ/OQ protocols and raw image retention policies.
Software & Data Management
Acquisition and analysis are managed through the CytoSMART Connect desktop application (Windows 10/11, 64-bit; USB 3.0 host interface required). Image processing occurs locally using GPU-accelerated inference; results—including annotated TIFF stacks, CSV summary reports, and histogram distributions—are exportable without cloud dependency. Optional secure TLS 1.3–encrypted synchronization with CytoSMART Cloud enables centralized data archiving, cross-laboratory comparison, and automated report generation compliant with ISO/IEC 17025 analytical record-keeping guidelines. Software versioning follows semantic versioning (SemVer) with documented change logs and validation summaries available upon request.
Applications
- Quantitative assessment of primary hepatocyte, neuronal, and iPSC-derived organoid batches pre- and post-cryopreservation
- Monitoring proliferation kinetics and cytotoxicity endpoints in 3D co-culture drug screening assays
- Standardization of cell seeding density for CRISPR editing, transfection, or bioprinting workflows
- QC release testing of clinical-grade MSC or CAR-T products under ATMP regulatory frameworks
- Longitudinal tracking of spheroid growth dynamics and necrotic core formation in hypoxia models
FAQ
Can the Exact FL perform viability assessment?
Yes. Viability is determined either via brightfield using trypan blue exclusion (automatically toggled when fluorescence channels are disabled) or via dual-channel fluorescence using AO/PI, Calcein-AM/EthD-1, or similar compatible dyes.
Which counting chambers are compatible?
Any transparent chamber with a defined 0.1 mm depth—regardless of material (glass or polymer), presence of engraved grids, or number of counting areas—is supported.
Is an external light shield required for fluorescence imaging?
Yes. The included magnetic light-blocking plate must be installed over the sample stage to minimize stray light interference and ensure optimal signal-to-noise ratio.
Does the system require a dedicated computer?
Yes. A Windows 10/11 PC or laptop with USB 3.0 port and network connectivity (Wi-Fi or Ethernet) is mandatory for operation and optional cloud integration.
How should the instrument surface be cleaned?
Wipe exterior surfaces with lint-free wipes moistened with 70% ethanol or isopropanol. Do not use acetone, autoclave, or abrasive cleaners.