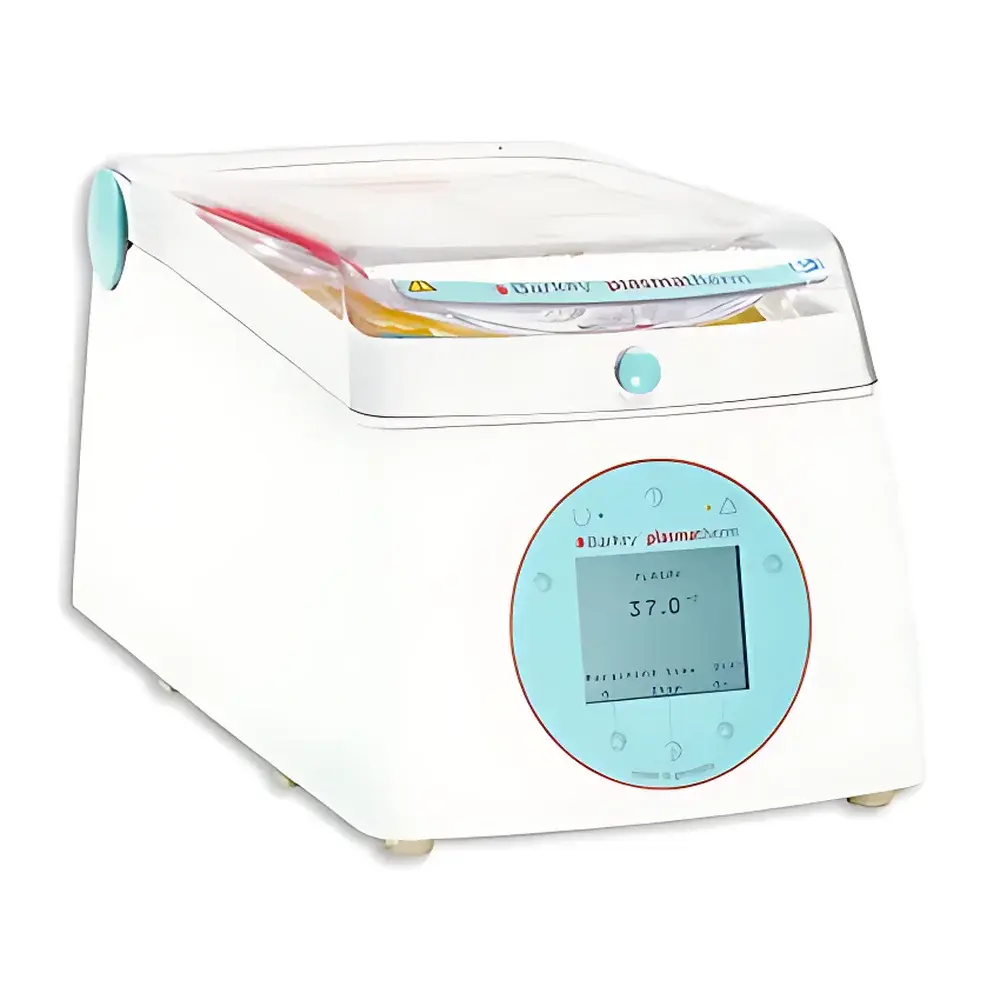
Barkey Plasmatherm Dry-Phase Cell Thawing and Blood Warming System
| Brand | Barkey |
|---|---|
| Origin | United Kingdom |
| Model | Barkey Plasmatherm |
| Instrument Type | Dry-phase (non-contact, closed-loop thermal transfer) thawing system |
| Regulatory Classification | Class II medical device per MDR 2017/745 (EU), compliant with ISO 23500-1:2023 for blood component warming |
| Software Compliance | Audit-trail enabled, 21 CFR Part 11–ready data logging |
| Certification | CE-marked, UKCA-certified, GMP-aligned design |
Overview
The Barkey Plasmatherm Dry-Phase Cell Thawing and Blood Warming System is an engineered thermal management platform designed for the controlled, reproducible, and traceable warming of cryopreserved biological materials—including peripheral blood mononuclear cells (PBMCs), hematopoietic progenitor cells (HPCs), fresh frozen plasma (FFP), cryoprecipitate (AHF), whole blood, and packed red blood cells (RBCs). Unlike conventional water-bath thawers, the Plasmatherm employs a sealed, recirculating glycol–water thermal fluid loop that transfers heat indirectly through conductive aluminum heating plates. This dry-phase architecture eliminates direct liquid contact with blood bags or cryovials, thereby mitigating risks of bag rupture, microbial ingress, cross-contamination, and operator exposure to biohazards. The system operates on validated thermal kinetics aligned with ISO 23500-1:2023 and AABB Standard 3.12.0, ensuring uniform temperature distribution across sample surfaces (±0.3 °C spatial deviation at setpoint) and compliance with clinical transfusion safety requirements.
Key Features
- Dry-phase thermal transfer: Closed-loop heated plate system—no immersion, no condensation, no waterborne pathogens.
- Precision temperature control: Setpoint range from 30 °C to 42 °C; ±0.2 °C stability over full operational cycle.
- Five pre-validated warming protocols: BLOOD (for RBCs/whole blood), PLASMA (FFP/cryoprecipitate), HPC (hematopoietic progenitor cells), USER (customizable ramp-hold profiles), and CONTINUOUS (for high-throughput batch processing).
- Integrated real-time monitoring: Dual NTC sensors per chamber, continuous logging of plate surface temperature, ambient air temperature, and lid-open status.
- GMP-aligned data integrity: Onboard non-volatile memory stores complete thaw logs (timestamp, protocol ID, start/end time, max/min/mean temperature, operator ID, alarm events) with export via USB or Ethernet to LIMS/HIS.
- Passive safety architecture: Triple-redundant overtemperature cutoff, automatic lid-open interruption, leak detection via capacitive fluid-sensing array, and audible/visual alarm escalation.
- Low-maintenance engineering: Chemically resistant polymer housing (ISO 10993-5 cytocompatible), wipe-clean surface compatible with 70% ethanol, IPA, and hydrogen peroxide vapor; thermal fluid replacement interval: once per 12 months.
Sample Compatibility & Compliance
The Plasmatherm accommodates standard blood bag configurations (up to 600 mL CPDA-1 or additive solution–containing units), cryovials (1.8–5.0 mL), and cell therapy cryobags (e.g., CryoBag™, MACS® GMP Bag). It meets the thermal performance criteria defined in FDA Guidance for Industry: “Cryopreservation and Thawing of Hematopoietic Progenitor Cells” (2022), EU Annex 1 (sterile processing), and CLSI GP44-A6. All warming protocols are validated per ISO 15189:2022 clause 5.5.2 for measurement uncertainty and traceability. Device firmware supports electronic signature capture and ALCOA+ data governance principles.
Software & Data Management
The embedded Plasmatherm Control Suite provides password-protected user roles (Operator, Supervisor, Administrator), configurable audit trail settings (including event type, timestamp, user ID, and before/after values), and encrypted CSV log export. Optional integration with enterprise LIMS via HL7 v2.5 or ASTM E1384-compliant messaging enables automatic ingestion of thaw metadata—including barcode-scanned bag ID, lot number, expiration date, and thaw completion flag—ensuring alignment with China’s Ministry of Health Order No. 85 (“Administrative Measures for Clinical Blood Use in Medical Institutions”) and EU MDR post-market surveillance obligations.
Applications
- Clinical transfusion services: Rapid, standardized FFP and cryoprecipitate thawing meeting AABB Standard 3.12.0.
- Cell therapy manufacturing: GMP-compliant HPC and CAR-T cell recovery with documented thermal history per ICH Q5D and USP .
- Blood bank QC laboratories: Reproducible RBC warming prior to compatibility testing or hemolysis assessment.
- Biobanking operations: High-fidelity PBMC recovery from long-term vapor-phase LN₂ storage.
- Research core facilities: Multi-sample parallel thawing with protocol locking and inter-lab comparability.
FAQ
Does the Plasmatherm require consumables such as disposable warming sleeves or water bath inserts?
No. The system operates without single-use components, eliminating recurring supply costs and reducing environmental waste in accordance with national healthcare material reform policies.
Can the device be calibrated in-house?
Yes—calibration is performed using NIST-traceable PT100 reference probes; procedures and acceptance criteria are documented in the IQ/OQ/PQ validation kit supplied with each unit.
Is remote monitoring supported?
Optional Ethernet connectivity enables SNMP-based network alerts and integration with centralized facility monitoring systems (e.g., Siemens Desigo, Honeywell Experion).
What is the maximum sample throughput per cycle?
Up to six standard 200–400 mL blood bags or twelve 5 mL cryovials, depending on selected protocol and thermal load.
How is software validation handled for regulated environments?
The Plasmatherm Control Suite includes a fully documented 21 CFR Part 11 validation package (risk assessment, test scripts, evidence reports) supporting GxP deployment in FDA- and EMA-inspected sites.