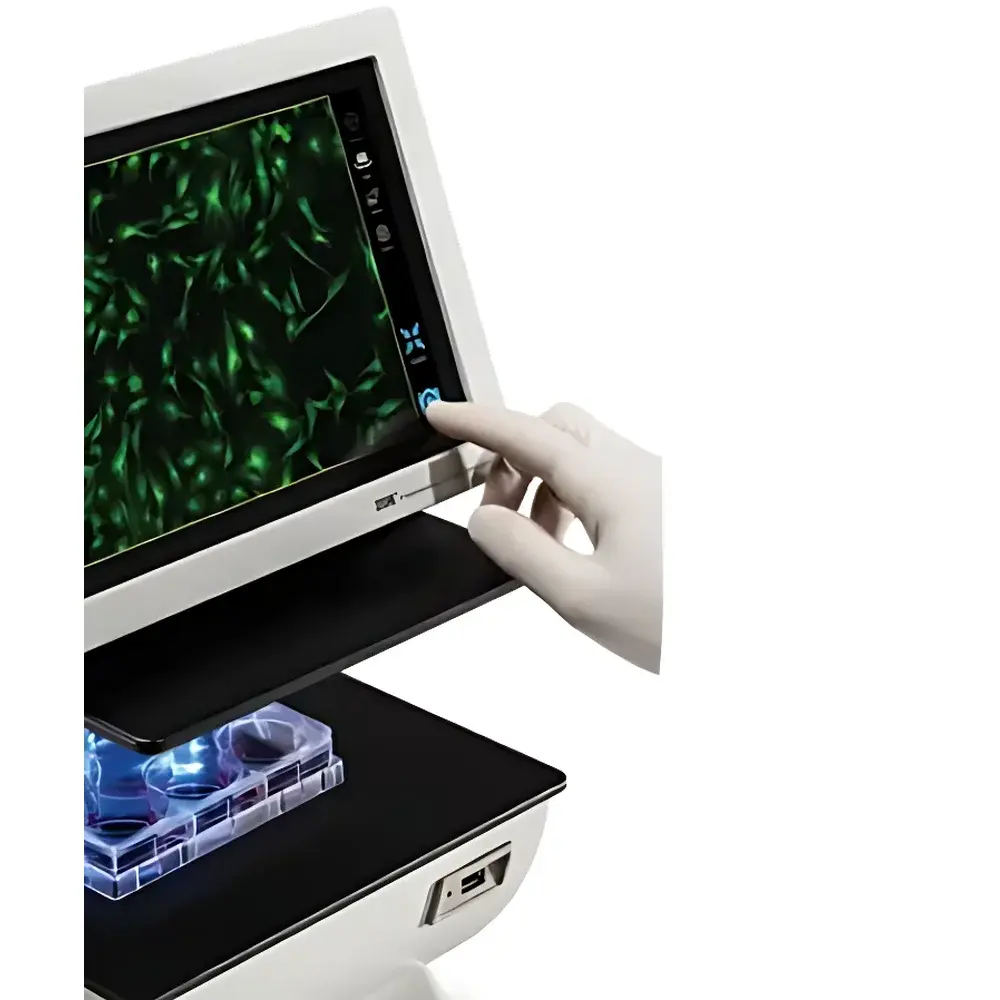
Bio-Rad ZOE Fluorescent Cell Imager
| Brand | Bio-Rad |
|---|---|
| Origin | USA |
| Model | ZOE |
| Product Category | Integrated Fluorescent Cell Imaging System |
| Excitation/Emission Filters (Blue Channel) | 355/40 nm / 433/36 nm |
| Excitation/Emission Filters (Green Channel) | 480/17 nm / 517/23 nm |
| Excitation/Emission Filters (Red Channel) | 556/20 nm / 615/61 nm |
| Imaging Sensor | 5 MP, 12-bit CMOS monochrome camera |
| Display | 10.1-inch high-resolution LCD touchscreen (1280 × 768) |
| Objective | 20× achromatic, wide-field mounting (0.70 mm² FOV, ~180% larger than standard 20×) |
| Stage | Motorized XY stage with ±6 mm travel, touch-controlled navigation |
| Illumination | LED-based brightfield (adjustable green LED ring with quadrant masking for oblique illumination) and fluorescence excitation (no warm-up, >10,000 h lifetime) |
| Image Storage | 16 GB internal memory (~2,500 JPEG images) |
| Output Formats | JPEG, TIFF, RAW |
| Software Features | Real-time gain, exposure time, LED intensity, and contrast adjustment |
Overview
The Bio-Rad ZOE Fluorescent Cell Imager is an integrated, benchtop cell imaging platform engineered for routine cell culture monitoring and multichannel fluorescence visualization without the complexity of traditional upright or inverted microscope systems. It operates on a fixed-optics, LED-illuminated inverted architecture—combining a high-sensitivity 5-megapixel 12-bit monochrome CMOS sensor with three precisely aligned fluorescence channels (DAPI/FITC/TRITC-equivalent), a motorized XY stage, and a calibrated 20× achromatic objective mounted in a proprietary wide-field configuration. Unlike conventional microscopes requiring Köhler illumination alignment, mercury lamp replacement, or darkroom operation, the ZOE delivers stable, reproducible imaging through solid-state LED excitation (355/40–615/61 nm), cold uniform illumination, and real-time z-axis feedback via on-screen focus assist. Its design prioritizes operational robustness, minimal maintenance, and compliance-ready documentation—making it suitable for academic core facilities, biomanufacturing QC labs, and GLP-regulated preclinical workflows where traceability, repeatability, and ease of training are critical.
Key Features
- Integrated 10.1-inch anti-glare, fingerprint-resistant LCD touchscreen (1280 × 768 resolution) enabling collaborative sample review and intuitive one-touch image capture
- Three optimized fluorescence channels with hard-coated, long-life dichroic filters: Blue (355/40 nm ex / 433/36 nm em), Green (480/17 nm ex / 517/23 nm em), Red (556/20 nm ex / 615/61 nm em)
- Patented adjustable green LED ring illumination for brightfield—supports quadrant masking to generate controlled oblique lighting, enhancing edge contrast without phase optics
- Motorized XY stage with ±6 mm travel range, fully controllable via touchscreen interface for rapid multi-region scanning (e.g., confluency assessment across wells)
- Wide-field 20× achromatic objective delivering 0.70 mm² field of view—~180% larger than standard 20× objectives—and supporting 4×–20× digital zoom with sub-micron resolution retention (≥1 µm)
- LED illumination system with >10,000-hour lifetime, zero warm-up delay, and programmable intensity control per channel—reducing phototoxicity and eliminating mercury lamp replacement cycles
- Onboard image processing engine supporting real-time adjustment of gain, exposure time, LED intensity, and contrast—plus post-capture brightness/contrast correction and RGB channel fusion
Sample Compatibility & Compliance
The ZOE imager accommodates standard tissue culture vessels including 6–96-well plates, Petri dishes (up to 100 mm), flasks (T25–T75), and chamber slides—all without coverslip removal or immersion oil. Its enclosed optical path and built-in light shield eliminate ambient interference, permitting fluorescence imaging under ambient lab lighting—no darkroom required. The system complies with ISO 13485-aligned manufacturing standards and supports audit-ready documentation: each acquired image embeds timestamp, objective ID, exposure parameters, LED intensities, and user-defined metadata. When deployed in regulated environments, the firmware enables optional password-protected user roles and export logs compatible with FDA 21 CFR Part 11 requirements for electronic records and signatures—facilitating integration into GMP/GLP quality management systems.
Software & Data Management
The embedded Linux-based imaging software provides full local control—no external PC required. Images are stored internally (16 GB flash memory, ~2,500 JPEGs) with lossless RAW capture capability. Export options include JPEG (for quick sharing), TIFF (for quantitative analysis), and RAW (for third-party deconvolution or spectral unmixing). USB 2.0 ports support direct transfer to external drives or network-attached storage. Metadata—including objective magnification, filter set used, exposure duration, gain setting, and stage coordinates—is automatically appended to each file header in EXIF-compatible format. Batch export preserves folder hierarchy and naming conventions, enabling seamless ingestion into LIMS or ELN platforms such as LabArchives or Benchling.
Applications
- Quantitative confluence estimation in adherent cell lines during passaging or bioreactor inoculation
- Morphological assessment of primary cells, stem cells, or iPSC-derived cultures under both brightfield and fluorescence
- Time-resolved monitoring of GFP/RFP-tagged protein expression dynamics in transient transfection assays
- Immunofluorescence validation of subcellular localization (e.g., nuclear vs. cytoplasmic markers)
- Transfection efficiency scoring using co-transfected fluorescent reporters (e.g., GFP + RFP dual-labeling)
- Routine QC of CRISPR-edited clones via fluorescent reporter readout or viability staining (e.g., Calcein AM/PI)
- Documentation of cellular responses to compound treatment in phenotypic screening workflows
FAQ
Does the ZOE support live-cell time-lapse imaging?
Yes—via programmable interval capture (1 sec to 24 hr intervals) with temperature-stable LED illumination and minimal photobleaching. External environmental chambers (e.g., stage-top incubators) are required for CO₂ and humidity control.
Can I perform quantitative fluorescence intensity measurements?
The system provides relative intensity values per pixel in RAW mode; absolute quantification requires external calibration with NIST-traceable fluorescent standards and background subtraction protocols.
Is the ZOE compatible with third-party analysis software like ImageJ or CellProfiler?
Yes—TIFF and RAW exports retain full bit-depth and spatial metadata, enabling direct import and batch processing in open-source or commercial analysis pipelines.
What maintenance is required beyond routine cleaning?
None. Solid-state LEDs, sealed optical paths, and factory-aligned filter cubes eliminate recalibration, lamp replacement, or alignment procedures throughout the instrument’s service life.
How does the ZOE handle autofluorescence in mammalian samples?
The narrow-band excitation/emission filters and high optical rejection ratios (>OD6) minimize bleed-through; users may further suppress background by optimizing exposure time and applying rolling-ball background subtraction during post-processing.