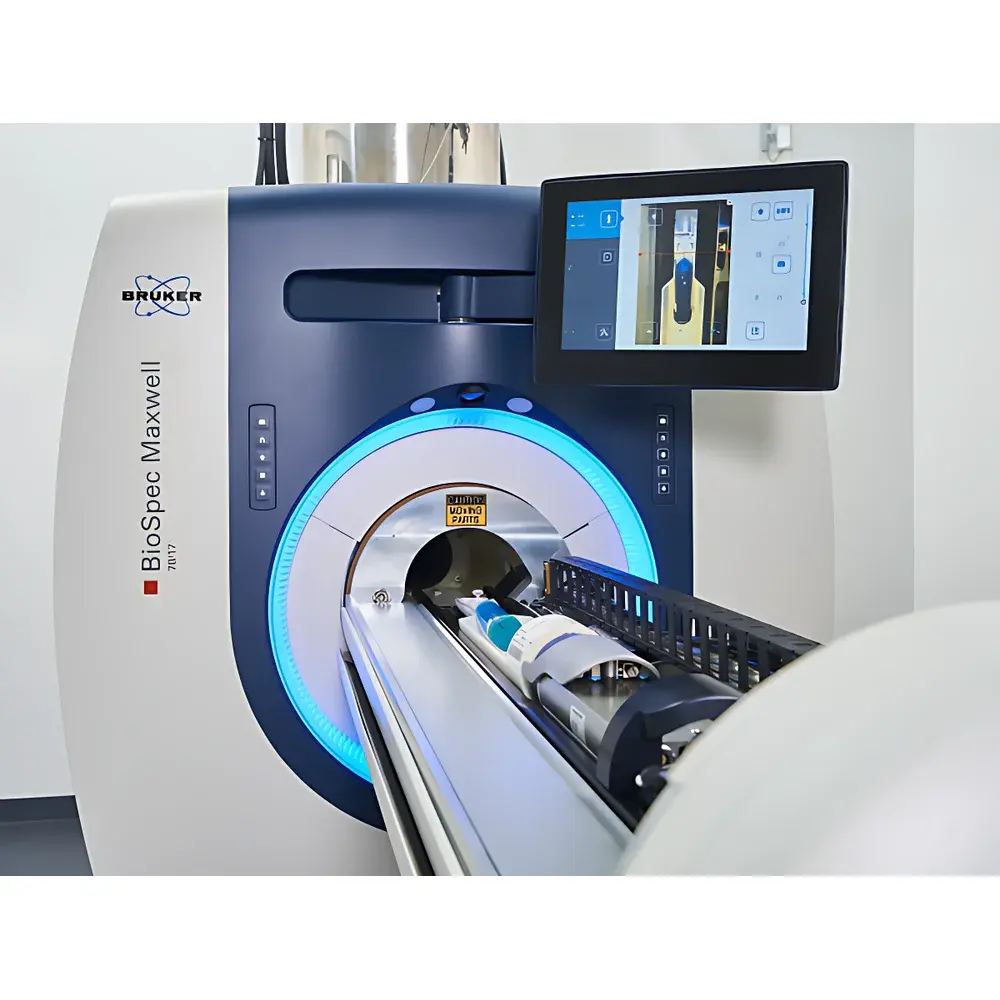
Bruker BioSpec Maxwell MRI System
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | Maxwell |
| Instrument Type | Preclinical Magnetic Resonance Imaging (MRI) System |
| Energy Resolution | 1000 (arbitrary units, typical for spectral stability metric) |
| Spatial Resolution | 0.05 mm (isotropic, in vivo) |
| Scan Time per Sequence | <1 min (typical for rapid anatomical protocols) |
| Simultaneous Sample Capacity | 2 animals |
Overview
The Bruker BioSpec Maxwell MRI System is a next-generation preclinical magnetic resonance imaging platform engineered for high-fidelity, longitudinal in vivo studies in small animal models. Unlike conventional superconducting MRI systems requiring periodic liquid helium refills and complex cryogenic infrastructure, the Maxwell platform integrates Bruker’s proprietary Maxwell magnet technology — a fully self-contained, cryogen-free superconducting magnet operating at 3T, 7T, or 9.4T field strengths. This architecture eliminates dependence on external liquid helium supply chains, removes the need for RF-shielded rooms or quench pipes, and enables installation in standard laboratory environments without structural reinforcement. The system employs a closed-cycle pulse tube cooler with advanced vascular cold head design to maintain thermal stability over extended periods, ensuring sub-micron gradient timing fidelity (<1 µs resolution), minimal vibration during fMRI and diffusion-weighted acquisitions, and robust long-term field homogeneity. Its core measurement principle relies on nuclear magnetic resonance (NMR) signal detection from hydrogen nuclei (¹H) in biological tissues under precisely controlled static magnetic (B₀), radiofrequency (B₁), and time-varying gradient fields — enabling non-invasive, quantitative assessment of anatomy, perfusion, metabolism, functional activation, and microstructural integrity.
Key Features
- Cryogen-free Maxwell magnet technology: Zero liquid helium consumption, >6-hour hold time during cooling interruption, no quench pipe or RF shielding required
- Compact footprint and lightweight design: Enables deployment in existing lab spaces, including multi-modality suites adjacent to PET, CT, or optical imaging systems
- Motorized, glove-compatible touchscreen animal positioning system with precision sub-millimeter reproducibility
- Modular animal handling platform: Includes quick-lock anesthesia gas exhaust ports, IV tracer injection pathways, and dual-temperature (water- and air-based) physiological support
- High-performance gradient subsystem: Up to 900 mT/m maximum amplitude and 4200 T/m/s slew rate, with hardware-synchronized channels for artifact-minimized EPI and DWI
- AVANCE NEO spectrometer architecture: Fully digital, high-dynamic-range RF transmission and reception with built-in B₁ field optimization (dual-channel base, expandable to 8-channel)
- Advanced thermal management: Vascular cold head and hydraulic isolation feet minimize mechanical drift and acoustic noise during sensitive functional or diffusion experiments
Sample Compatibility & Compliance
The BioSpec Maxwell accommodates rodents (mice and rats) across multiple configurations: single-animal beds optimized for high-resolution neuroimaging, dual-animal beds for comparative or pharmacokinetic studies, and a triple-mouse bed option for throughput-oriented screening. All animal interfaces comply with AAALAC International standards for humane handling and physiological monitoring. The system supports integration with FDA 21 CFR Part 11–compliant audit trails via ParaVision 360 software, and its operational parameters adhere to ISO/IEC 17025 traceability frameworks when used in GLP-certified preclinical research facilities. Gradient performance meets IEC 62704-1 safety thresholds for peripheral nerve stimulation (PNS) limits in rodent-sized bore geometries.
Software & Data Management
ParaVision 360 serves as the unified acquisition, reconstruction, and analysis environment. It includes over 1,000 validated pulse sequences — spanning T₁/T₂ mapping, ASL perfusion, DTI, fMRI, MRS, and angiography — with more than 100 pre-optimized, publication-ready protocols for oncology, neurology, and inflammation research. Each protocol embeds automated quantification modules (e.g., tumor volume segmentation, cortical thickness estimation, ADC histogram analysis) to ensure inter-user reproducibility. The Pulse Sequence Development Environment (PSDE) allows researchers to implement custom k-space trajectories, RF pulse shapes, and real-time feedback loops using MATLAB-based scripting. All data are stored in DICOM-compliant format with embedded metadata (field strength, gradient specs, coil configuration), facilitating PACS integration and FAIR (Findable, Accessible, Interoperable, Reusable) data practices.
Applications
The BioSpec Maxwell MRI supports a broad spectrum of translational research applications, including but not limited to: longitudinal monitoring of orthotopic brain tumor growth and treatment response; high-resolution hippocampal volumetry in Alzheimer’s disease models; cardiac function assessment via cine-MRI and late gadolinium enhancement; renal perfusion mapping using arterial spin labeling; and whole-brain functional connectivity analysis via resting-state fMRI. Its compatibility with sequential or integrated PET/MRI acquisition expands molecular imaging capabilities — enabling simultaneous assessment of glucose metabolism (¹⁸F-FDG PET) and structural/functional MRI biomarkers within the same imaging session. The system’s stability and automation make it particularly suited for multi-site collaborative studies requiring cross-platform harmonization.
FAQ
Does the BioSpec Maxwell require liquid helium refills?
No — the Maxwell magnet uses a closed-cycle cryocooler and does not rely on liquid helium or liquid nitrogen.
Can the system be installed without an RF-shielded room?
Yes — its integrated active shielding and automatic RF compensation eliminate the need for external RF cages.
What is the minimum spatial resolution achievable in vivo?
Isotropic resolution down to 0.05 mm is routinely achieved in high-SNR anatomical scans of mice under optimal coil and sequence conditions.
Is ParaVision 360 compliant with regulatory data integrity requirements?
Yes — optional 21 CFR Part 11 compliance packages include electronic signatures, audit trails, and role-based access control.
How many animals can be scanned simultaneously?
The standard configuration supports two animals; optional triple-mouse beds are available for parallel throughput studies.
Can the system be upgraded to PET/MRI capability?
Yes — Bruker offers both sequential PET insert modules and fully integrated PET/MRI solutions compatible with the Maxwell platform.