AS ONE Cell Picking System – Automated High-Throughput Monoclonal Antibody-Secreting Cell Isolation Platform
| Brand | AS ONE |
|---|---|
| Origin | Japan |
| Manufacturer | AS ONE Co., Ltd. |
| Type | Imported Instrument |
| Model | AS ONE Cell Picking System |
| Separation Method | Fully Automated Microfluidic Single-Cell Isolation |
| Cell Viability | 60–90% |
| Chip Format | Array of >100,000 Microwells (Ø ≈ 10 µm) |
| Detection Mode | Fluorescence Intensity + Brightfield Imaging |
| Recovery Mechanism | Glass Capillary-Based Contact-Free Aspiration |
Overview
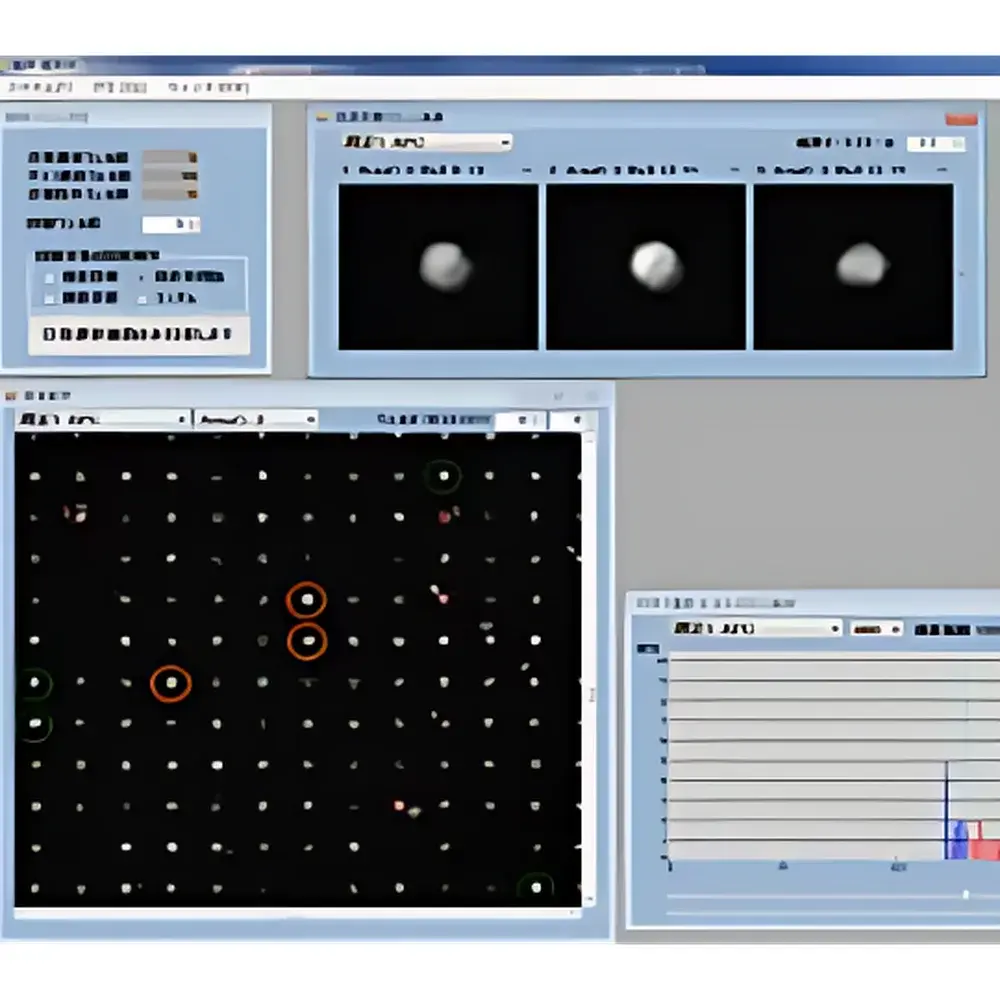
The AS ONE Cell Picking System is an engineered microfluidic platform for automated, high-throughput isolation of monoclonal antibody-secreting cells at single-cell resolution. It operates on the principle of compartmentalized immunoassay—leveraging a proprietary microwell chip with over 100,000 precisely fabricated wells (diameter ~10 µm), each capable of physically isolating one viable cell. Within each well, an immunocompatible surface (e.g., amine-functionalized or antigen-coated) forms a localized “immunochamber,” enabling in situ capture and detection of secreted antibodies. Secreted immunoglobulins bind to immobilized antigens on the well wall; subsequent addition of fluorophore-conjugated secondary antibodies generates spatially resolved fluorescence signals at the periphery of each well. Integrated brightfield and epifluorescence imaging enables real-time phenotypic and functional assessment—correlating morphology, secretion intensity, and viability—without perturbing cellular integrity. This architecture bypasses stochastic dilution and manual handling inherent in limiting dilution assays (LDA), delivering deterministic, traceable, and auditable single-cell recovery for therapeutic antibody discovery.
Key Features
- High-density microwell array (>10⁵ wells/chip) with uniform 10 µm diameter geometry—optimized for single-cell entrapment and minimal cross-contamination.
- On-chip, single-cell ELISA functionality: covalent antigen/antibody immobilization on well surfaces supports quantitative secretion profiling without lysing or detaching cells.
- Automated capillary-based picking: non-contact glass capillary aspiration ensures mechanical gentleness, preserving membrane integrity and post-recovery proliferative capacity.
- Integrated dual-modality imaging: synchronized brightfield and fluorescence acquisition enables concurrent morphological classification and secretion quantification per well.
- Traceable cell lineage tracking: software assigns unique identifiers to each imaged well and logs full acquisition metadata—including time-stamped fluorescence intensity, area, and centroid coordinates—for audit-ready workflows.
Sample Compatibility & Compliance
The system accommodates suspension and semi-adherent mammalian cells including hybridomas, CHO-K1/S, HEK293, primary B lymphocytes, and circulating tumor cells (CTCs). It supports standard culture media (e.g., RPMI-1640, DMEM/F12) and common assay reagents (e.g., anti-IgG Fc Alexa Fluor 488/647, biotinylated antigens). The platform adheres to GLP-aligned operational principles: all image data, pick logs, and environmental timestamps are stored in immutable binary format with SHA-256 checksums. While not FDA-cleared as a diagnostic device, its data structure complies with ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) criteria and supports 21 CFR Part 11–compliant electronic signature integration when deployed with validated LIMS or ELN systems.
Software & Data Management
The AS ONE Control Suite (v4.2+) provides instrument control, multi-channel image acquisition, automated fluorescence thresholding, and pick-list generation. Analysis modules include well-level secretion heatmaps, scatter plots of intensity vs. cell size, and batch-wise viability correlation matrices. Raw TIFF stacks and processed CSV reports are exportable in FAIR-compliant formats (Findable, Accessible, Interoperable, Reusable). Audit trails record user actions, parameter changes, and hardware status events with ISO 8601 timestamps. Data encryption (AES-256 at rest/in transit) and role-based access control align with ISO/IEC 27001 information security frameworks.
Applications
- Therapeutic antibody discovery: Rapid isolation of antigen-specific IgG-secreting B cells from immunized rabbits or human PBMCs—enabling native V(D)J sequencing and recombinant expression within 48 h.
- Cell line development: High-resolution screening of CHO pools for clones exhibiting superior titer, stability, and glycosylation consistency—reducing clone selection timelines from weeks to hours.
- Cancer biology: Enumeration and recovery of CTCs from whole blood lysates with preserved RNA integrity for downstream NGS or ex vivo expansion.
- Regenerative medicine: Functional validation and retrieval of iPSC-derived cardiomyocytes or neural progenitors based on secreted trophic factor profiles (e.g., BDNF, VEGF).
- Single-cell multi-omics: Coupling with scRNA-seq library prep kits via direct lysis-in-well protocols—minimizing batch effects from manual sorting.
FAQ
What is the typical throughput for a single run?
A standard 100,000-well chip processes up to 50,000 viable single cells per 4–6 hour imaging and picking cycle, depending on secretion kinetics and imaging resolution settings.
Can the system be integrated with downstream molecular workflows?
Yes—recovered cells are dispensed into standard 96- or 384-well PCR plates or microfluidic lysis cartridges compatible with commercial single-cell RNA/DNA library kits.
Is surface chemistry customization supported?
AS ONE offers OEM-compatible chip variants with pre-coated streptavidin, Protein A/G, or custom antigen surfaces under NDA; users may also perform in-house coating using standard NHS-ester or silane protocols.
How is cell viability validated post-picking?
Viability is assessed via dual-stain (Calcein AM/Propidium Iodide) fluorescence immediately after recovery and again at 24 h post-culture; reported 60–90% range reflects inter-lot variability in primary samples—not instrument-induced damage.
Does the system support GMP environments?
The hardware meets IP54 enclosure rating and supports IQ/OQ documentation packages; full GMP compliance requires site-specific PQ validation aligned with ICH Q5D and USP guidelines for cell-based products.