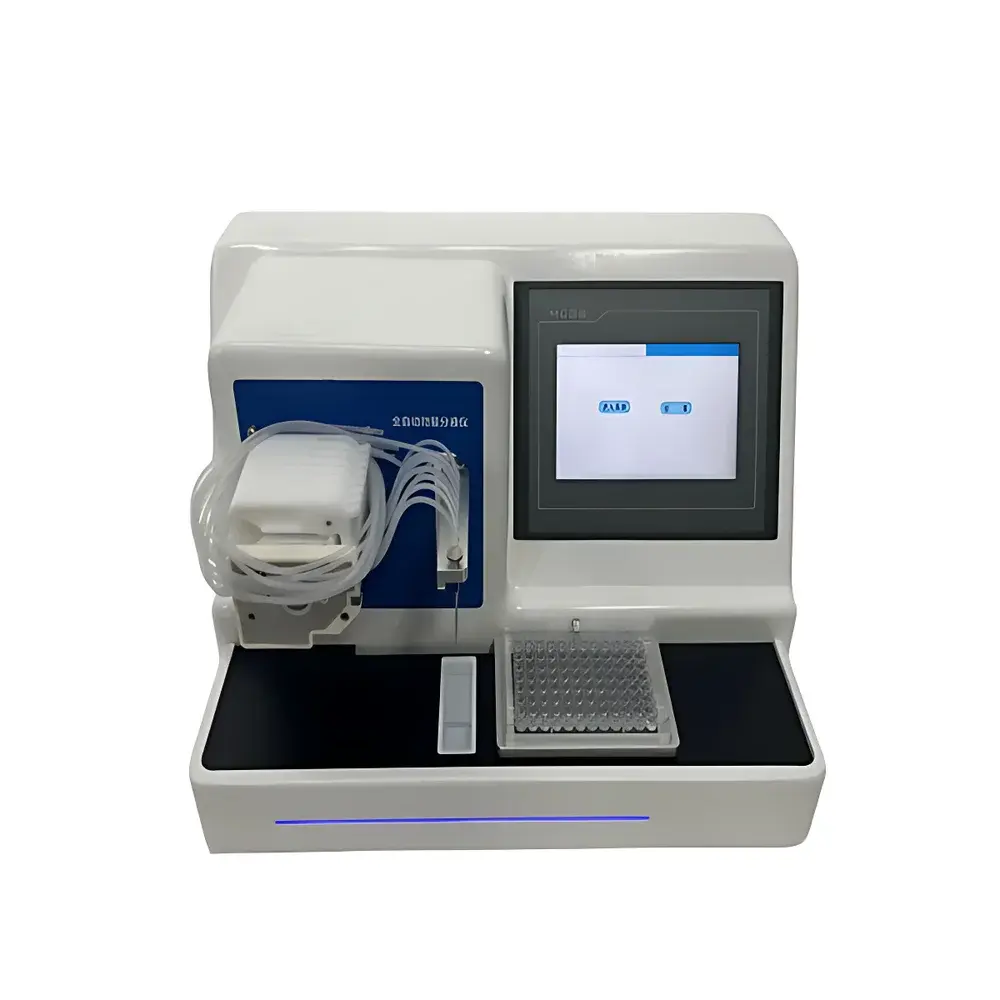
Chuanhong CHFY-96 Single-Channel Automated Microplate Dispenser
| Brand | Chuanhong |
|---|---|
| Origin | Shanghai, China |
| Model | CHFY-96 |
| Instrument Type | Automatic |
| Dispensing Principle | Peristaltic Pump |
| Dispensing Method | Peristaltic Pump |
| Plate Compatibility | 96-well, 384-well, PCR plates, skirted/unskirted microplates, tubes (1.5–15 mL), vials, glass bottles, cuvettes |
| Plate Height Adjustment | Motorized Auto-Adjustment |
| Dispensing Range | 100 µL per well (96-well plate) |
| throughput | 240 plates/hour |
| Dispensing Accuracy | ≤ ±1.5% at 100 µL |
| Liquid Handling Features | Aspirate-reverse dispensing, on-board pre-wash reservoir, programmable row/column/well-specific dispensing, three-speed dispensing modes, auto-cleaning cycle, tube sterilization compatibility (50× autoclave cycles) |
| User Interface | Color TFT touchscreen with Chinese/English bilingual OS |
| Power Supply | AC 220 V, 50 Hz |
| Dimensions (W×D×H) | 360 × 320 × 380 mm |
| Weight | 12.5 kg |
| Regulatory Compliance | Designed for GLP-compliant labs |
Overview
The Chuanhong CHFY-96 Single-Channel Automated Microplate Dispenser is an engineered solution for high-throughput, precision liquid handling in life science laboratories. Based on a calibrated peristaltic pumping mechanism, it delivers consistent volumetric transfer across standardized and non-standard microplate formats without requiring air displacement or positive-displacement pistons. This architecture eliminates carryover risk, avoids aerosol generation, and ensures zero residual volume in tubing during reverse aspiration—critical for expensive reagents such as antibodies, cytokines, and viral vectors. Unlike multi-channel or robotic liquid handlers, the CHFY-96 prioritizes operational simplicity, benchtop footprint efficiency, and rapid deployment in academic core facilities, IVD development labs, and contract research organizations (CROs). Its design targets applications where reproducibility at the microliter scale must be maintained across hundreds of wells per hour, while minimizing operator dependency and technical overhead.
Key Features
- Peristaltic pump system with silicone tubing and stainless-steel dispensing needle—validated for 50 autoclave cycles (121°C, 20 min) and compatible with broad-spectrum solvents including DMSO, ethanol, and aqueous buffers.
- Motorized Z-axis plate height adjustment automatically detects and accommodates plates from standard ANSI/SLAS-compliant 96-well to deep-well 384-well formats, as well as custom racks for cryovials, PCR strips, and conical tubes.
- Three dispensing speed modes (low/medium/high) enable optimization for viscous solutions (e.g., Matrigel®, methylcellulose), particulate suspensions (e.g., magnetic beads, primary cells), and low-surface-tension reagents (e.g., detergents, serum-containing media).
- Programmable dispensing logic supports full-plate, column-wise, or user-defined well patterns—with independent volume assignment per column—enabling gradient dilutions, assay setup with variable standards, or multiplexed reagent addition.
- Integrated pre-wash reservoir with removable, autoclavable design prevents overflow and allows unattended cleaning between runs; reverse aspiration function ensures complete reagent recovery and eliminates dead-volume waste.
- Bilingual (Chinese/English) color touchscreen interface requires no external PC or software installation—configuration, calibration, and run initiation are completed in under 90 seconds.
Sample Compatibility & Compliance
The CHFY-96 accommodates ANSI/SLAS-formatted microplates (96-, 384-, and 1536-well), skirted and unskirted PCR plates, 8-tube strips, 0.5–15 mL centrifuge tubes, 2–50 mL glass or plastic vials, and custom geometries via optional adapter frames. All wetted materials comply with USP Class VI biocompatibility standards. While not certified to ISO 8573-1 for compressed air purity (as no pneumatic system is used), its peristaltic architecture inherently meets ISO 13485 requirements for IVD manufacturing environments. Data logging supports traceable operation records aligned with GLP principles; optional CSV export includes timestamps, operator ID, protocol name, and dispense volume verification logs—facilitating internal audits and method validation documentation.
Software & Data Management
No proprietary software installation is required. All configuration, calibration, and execution occur locally via the embedded Linux-based HMI. Calibration routines include gravimetric verification at multiple setpoints (10, 50, and 100 µL), with results stored internally and exportable via USB. The system maintains a local history of the last 200 executed protocols—including date/time stamp, total dispensed volume, and error flags (e.g., tubing occlusion, insufficient reagent level). For labs operating under FDA 21 CFR Part 11 requirements, third-party electronic lab notebook (ELN) integration is supported via RS-232 or Ethernet (Modbus TCP), enabling secure protocol synchronization and digital signature linkage.
Applications
The CHFY-96 serves as a dedicated platform for routine yet critical workflows: ELISA/EIA plate coating and detection reagent addition; qPCR master mix distribution into 96- and 384-well plates; cell-based assay seeding (adherent and suspension lines); magnetic bead-based immunoassay setup; serial dilution series preparation; microplate washing priming; and IVD kit component filling during R&D scale-up. Its throughput—6 seconds per 96-well plate at 100 µL/well—supports daily processing of >1,400 wells with <2% CV, making it suitable for medium-throughput screening (MTS) pipelines where robotic automation is cost-prohibitive.
FAQ
Does the CHFY-96 support calibration traceability to national standards?
Yes—gravimetric calibration is performed using NIST-traceable analytical balances; calibration certificates can be generated per ISO/IEC 17025 guidelines when conducted by an accredited service provider.
Can the instrument dispense into non-ANSI plates such as custom microfluidic chips or irregularly spaced arrays?
Yes—custom mounting fixtures and Z-height offset parameters allow adaptation to non-standard geometries; mechanical alignment is verified manually prior to first use.
Is tubing replacement a field-serviceable procedure?
Yes—silicone tubing and stainless-steel needle assemblies are tool-free replaceable in <90 seconds; spare kits include tubing, clamps, and sterilization validation documentation.
What maintenance intervals are recommended for long-term accuracy retention?
Daily visual inspection of tubing wear; monthly gravimetric verification at 100 µL; annual pump roller replacement—documented in the included Maintenance Log Template.
Does the system meet electromagnetic compatibility (EMC) requirements for laboratory environments?
Yes—complies with IEC 61326-1:2013 for laboratory equipment, including immunity to electrostatic discharge (±8 kV contact), radiated RF fields (10 V/m), and fast transient bursts (2 kV).