Chuanyu Qi3536 Hemocytometer with 58-Cell Classification & Four Counting Modes
| Brand | Chuanyu Instruments (Shanghai) |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | PRC |
| Model | Qi3536 |
| Price Range | USD 490 – 560 |
Overview
The Chuanyu Qi3536 Hemocytometer is a dedicated clinical laboratory instrument engineered for manual morphological classification and quantitative enumeration of hematopoietic cells in peripheral blood smears, bone marrow aspirates, megakaryocyte preparations, and histochemical staining specimens. Unlike automated hematology analyzers that rely on impedance or flow cytometry, the Qi3536 supports visual microscopic assessment—enabling trained technologists to apply standardized morphologic criteria per WHO and ICSH guidelines. It implements a structured, keyboard-driven counting workflow aligned with established hematologic differential protocols, ensuring traceable, auditable, and repeatable manual counts across shifts and operators. Designed specifically for routine use in hospital hematology labs, reference laboratories, and academic teaching facilities, the device integrates ergonomic human factors with regulatory-ready data handling.
Key Features
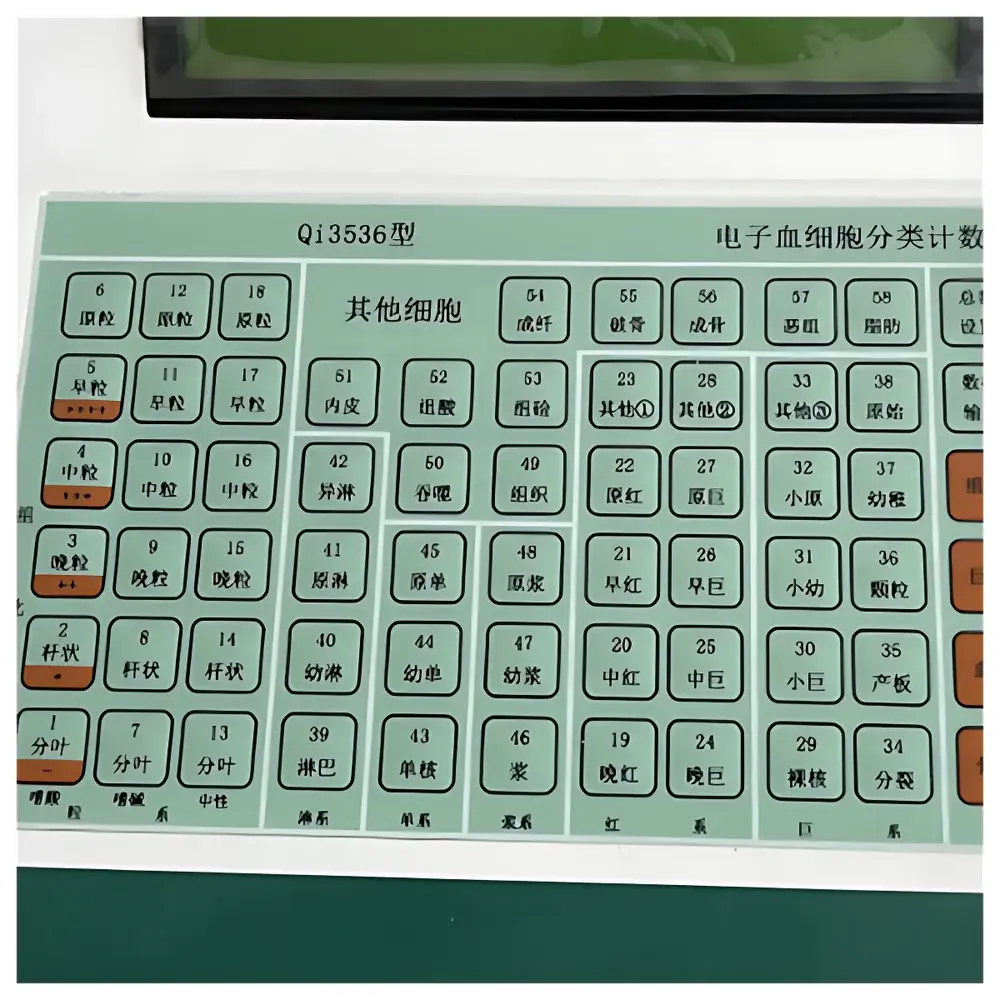
- Dedicated 58-cell classification matrix covering granulocytic, erythroid, megakaryocytic, plasmacytic, monocytic, lymphoid, histiocytic, stromal, and cytochemical categories—including differential subtypes (e.g., neutrophilic, eosinophilic, basophilic segmented forms)
- Four validated counting modes: Peripheral Blood Smear, Bone Marrow Aspirate, Megakaryocyte Enumeration, and Histochemical Staining Interpretation (with qualitative intensity scoring: −, +, ++, +++, ++++)
- 20×2-character backlit LCD display optimized for low-glare, high-contrast viewing under standard lab lighting conditions
- Non-volatile memory with power-loss protection retains all session data—including absolute counts, percentage distributions, five clinically relevant ratios (e.g., myeloid:erythroid), six lineage totals, and non-erythroid cell summaries
- Keyboard layout organized by hematopoietic lineage to minimize cognitive load and reduce transcription errors during prolonged counting sessions
- Compact benchtop form factor (240 × 170 × 85 mm) with integrated control unit—no external PC or host required for basic operation
Sample Compatibility & Compliance
The Qi3536 is intended for use with standard glass microscope slides prepared using Wright–Giemsa, May–Grünwald, or Prussian blue stains. It supports both capillary and smear-based specimen formats compliant with CLSI H20-A2 and ISO 15189:2022 requirements for manual differential counts. While not an IVD-certified analyzer under EU IVDR or FDA 510(k), its operational logic mirrors internationally accepted morphology-based classification frameworks referenced in WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues (5th ed.) and the Bethesda Consensus Guidelines. Data output meets GLP documentation standards for internal quality control logs and educational recordkeeping.
Software & Data Management
The device stores up to 99 counting sessions locally, each timestamped and identifiable by operator ID (manually entered). Export is supported via RS-232 serial interface to LIMS or Excel-compatible spreadsheets. No proprietary software installation is required; raw count files are plain-text CSV with column headers aligned to ISBT 128 terminology where applicable. Audit trail functionality includes session start/end time, total counted cells, and mode selection—sufficient for internal QA review and CAP/CLIA inspection readiness. Firmware updates are delivered via USB-to-serial adapter (optional accessory).
Applications
- Routine peripheral blood differential counts in outpatient and inpatient hematology workflows
- Bone marrow differential analysis for leukemia subtyping, MDS evaluation, and treatment response monitoring
- Megakaryocyte quantification in thrombocytopenia workups and myeloproliferative neoplasm assessment
- Histochemical interpretation support (e.g., myeloperoxidase, esterase, iron staining) with semi-quantitative intensity grading
- Medical student and resident training in hematologic morphology recognition and systematic enumeration methodology
- Reference lab verification of automated analyzer outliers requiring manual review per CLIA §493.1253(c)(1)
FAQ
Does the Qi3536 support automated image capture or AI-based cell recognition?
No. The Qi3536 is a manual counting aid only—it does not integrate digital microscopy, camera modules, or machine learning algorithms. It assumes user-initiated visual identification and keypress entry.
Is the device compliant with FDA 21 CFR Part 11 or EU IVDR?
No. It is classified as a Class I general-purpose laboratory instrument under Chinese NMPA regulations and is not marketed as an IVD device in regulated jurisdictions.
Can count data be exported directly to LIS without third-party software?
Yes—via RS-232 serial output in ASCII-delimited format compatible with most legacy LIS interfaces.
What maintenance is required?
No consumables or calibration kits are needed. Routine cleaning of the keypad and display with isopropyl alcohol is recommended per manufacturer guidelines.
Is technical support available in English?
Yes—Chuanyu Instruments provides English-language documentation and remote troubleshooting via authorized regional distributors.