COMECAUSE IN-CL1 Intelligent Airborne Particle Counter
| Brand | COMECAUSE |
|---|---|
| Model | IN-CL1 |
| Sampling Flow Rate | 2.83 L/min (0.1 cfm) |
| Particle Size Channels | 0.3, 0.5, 1.0, 3.0, 5.0, 10.0 µm |
| Counting Efficiency | ≥50% at 0.3 µm, 100% ±30% at 0.5 µm and 5.0 µm |
| Accuracy (Particle Concentration) | ±30% at 0.5 µm |
| Repeatability (Relative Standard Deviation) | ≤±10% |
| Zero Count Level | ≤1 count per 5 minutes |
| Self-Cleaning Time | ≤10 min |
| Operating Temperature | 15–35 °C |
| Relative Humidity | 10–85% RH (non-condensing) |
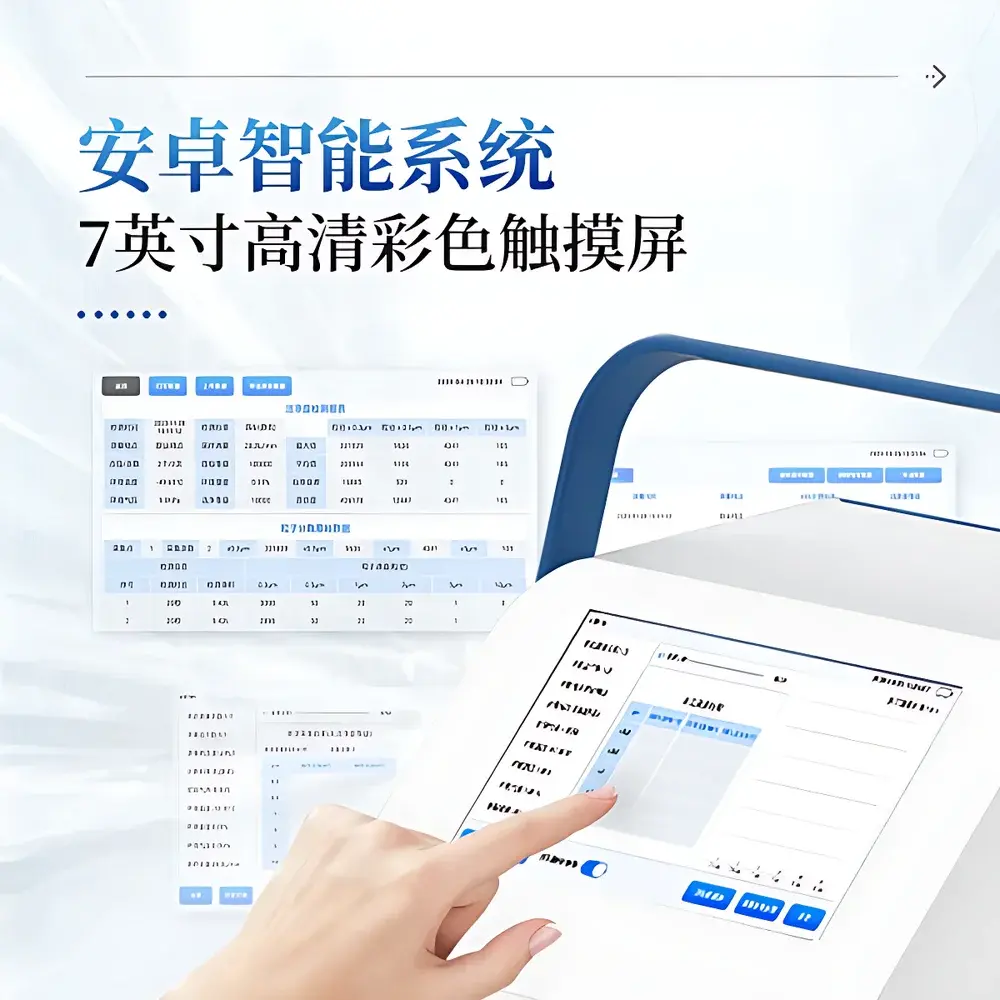
| Display | 7-inch color capacitive touchscreen |
| Data Storage Capacity | Up to 100,000 records |
| Connectivity | Wi-Fi 802.11 b/g/n, Bluetooth 4.2, USB Type-A (driver-free U-disk export) |
| Power Supply | Rechargeable Li-ion battery (16.8 V / 5 Ah, 74 Wh) |
| Battery Runtime | ≥6 hours continuous sampling |
| Charging Time | ≤2 hours |
| Printer | Built-in high-speed thermal printer (selectable channel output) |
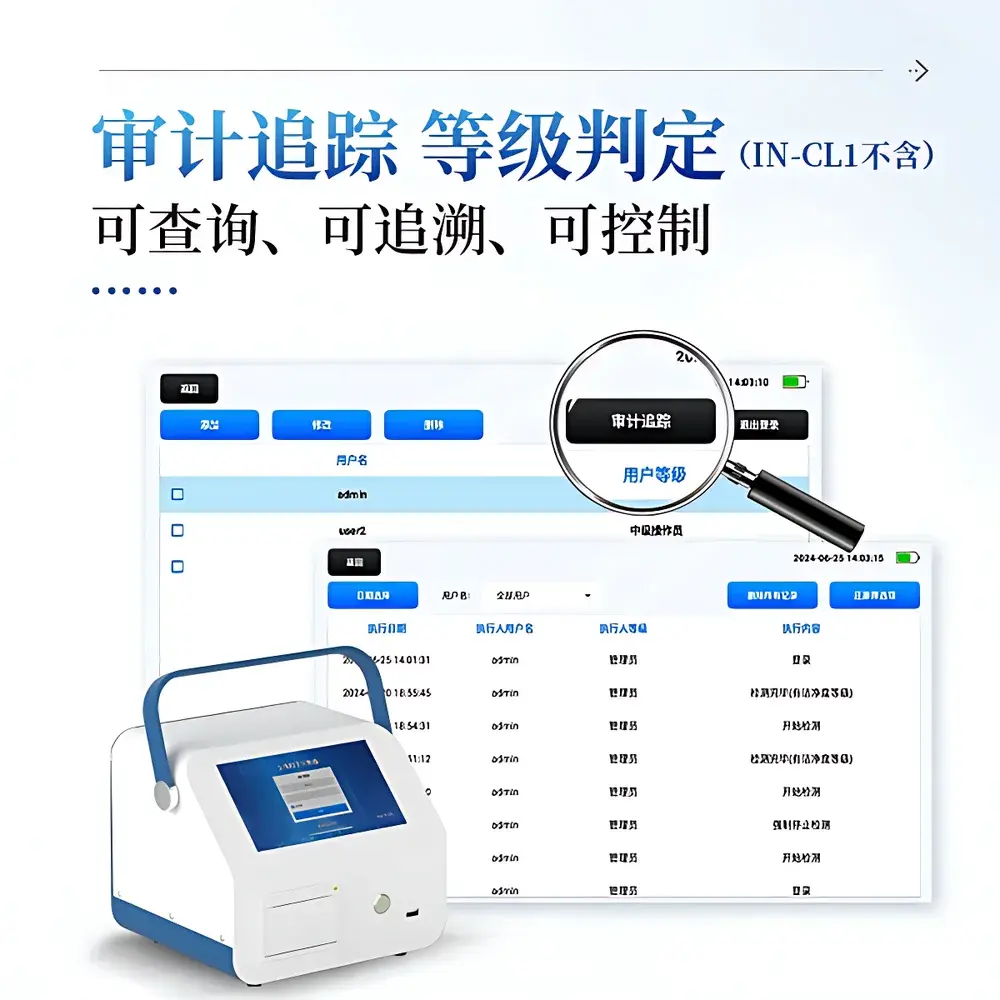
| Compliance | GB/T 6167–2007, JJF 1190–2008, ISO 14644-1, Fed Std 209E, Annex 1 of EU GMP (2022), USP <788>, FDA 21 CFR Part 11 (audit trail enabled in platform mode) |
Overview
The COMECAUSE IN-CL1 Intelligent Airborne Particle Counter is a portable, microprocessor-controlled optical particle counter engineered for real-time quantification of airborne particulate contamination in controlled environments. It operates on the principle of single-particle light scattering: aerosol samples are drawn through a precisely defined sensing volume illuminated by a high-stability imported semiconductor laser diode (rated lifetime >30,000 hours). Scattered light pulses from individual particles are detected by a low-noise photodetector assembly; pulse height analysis enables discrimination across six calibrated size channels—0.3, 0.5, 1.0, 3.0, 5.0, and 10.0 µm—with counting efficiency meeting ISO 21501-4 requirements. The instrument implements 95% Upper Confidence Limit (UCL) statistical evaluation per ISO 14644-1 Annex D, enabling automated cleanroom classification under static and dynamic conditions per EU GMP Annex 1 and FDA guidance. Its compact form factor, integrated environmental sensors (temperature, relative humidity, barometric pressure), and dual AC/battery operation support unattended monitoring in Grade A–D cleanrooms, isolators, and critical process zones.
Key Features
- Android 7.1 operating system with intuitive graphical interface and multi-touch responsive 7-inch color display
- Optimized optical cavity design minimizing stray light interference—enhanced signal-to-noise ratio ensures improved counting accuracy and stability
- Real-time simultaneous acquisition and visualization of all six particle size channels with time-series trend overlay
- Automated ISO 14644-1 cleanroom classification with UCL calculation per sampling location and duration
- Integrated environmental parameter logging (temperature, RH, pressure) synchronized with particle data for contextual traceability
- Dual-mode connectivity: Wi-Fi and Bluetooth for live transmission to local networks or cloud-based regulatory platforms; driver-free USB mass storage for rapid data extraction
- Built-in thermal printer supporting selective channel output, timestamped reports, and configurable header/footer fields
- Extended field operation enabled by 74 Wh rechargeable lithium-ion battery—supports ≥6 hours of continuous 2.83 L/min sampling
- Ergonomic telescoping tripod mount with 360° rotation and fine-height adjustment for reproducible probe positioning
- Comprehensive onboard memory (up to 100,000 records) with searchable timestamp, location ID, and user-defined batch tagging
Sample Compatibility & Compliance
The IN-CL1 is validated for use with ambient air, compressed gases (filtered via 0.1 µm membrane), and HEPA/ULPA-filtered laminar airflow streams. It meets metrological requirements specified in JJF 1190–2008 (Calibration Specification for Airborne Particle Counters) and performance testing criteria outlined in GB/T 6167–2007. Regulatory alignment includes ISO 14644-1:2015 (cleanroom classification), ISO 21501-4:2018 (light-scattering particle counter calibration), Fed Std 209E (withdrawn but referenced in legacy protocols), and Annex 1 of the EU Guidelines to Good Manufacturing Practice (2022 revision). When deployed with the optional COMECAUSE Regulatory Cloud Platform, the system supports 21 CFR Part 11-compliant electronic records—including audit trails, role-based access control, electronic signatures, and immutable data archiving—satisfying GLP and GMP data integrity requirements.
Software & Data Management
Data acquisition, visualization, and reporting are managed via embedded firmware and the optional COMECAUSE Regulatory Cloud Platform. On-device software enables customizable sampling protocols (cycle duration: 1 s–99 min; delay: ≤99 s; interval: ≤999 s), user-defined location mapping, and real-time UCL-based classification alerts. Export formats include CSV (for Excel/Statistica), PDF (print-ready reports), and XML (LIMS integration). The cloud platform provides centralized fleet management, cross-site comparative analytics, long-term trend modeling (e.g., filter decay rate estimation), and automated compliance dashboards aligned with ISO 14644-2 surveillance schedules. All data transfers employ TLS 1.2 encryption; platform audit logs record every data modification, user login, and configuration change with ISO-compliant timestamps.
Applications
- Pharmaceutical manufacturing: Monitoring Grade A isolators, Grade B background areas, and aseptic filling lines per EU GMP Annex 1 and USP
- Biotechnology facilities: Environmental qualification of cell culture suites, fill-finish suites, and lyophilization corridors
- Semiconductor fabrication: Cleanroom certification in photolithography, etching, and packaging zones per SEMI F21 standards
- Medical device production: Validation of Class 100–100,000 environments during sterile barrier system assembly
- Food & beverage processing: Verification of hygiene-critical zones in dairy, infant formula, and ready-to-eat meal production
- HEPA/ULPA filter testing: In-situ efficiency verification and leak detection using upstream/downstream differential counting
- Aerospace & optics manufacturing: Monitoring ISO Class 5–8 assembly enclosures for precision optics and avionics integration
FAQ
Does the IN-CL1 comply with FDA 21 CFR Part 11 for electronic records?
Yes—when operated in conjunction with the COMECAUSE Regulatory Cloud Platform, the system enforces electronic signature workflows, audit-trail generation, and data immutability per Part 11 Subpart B requirements.
What is the minimum detectable particle size and its counting efficiency?
The instrument detects particles ≥0.3 µm with ≥50% counting efficiency at 0.3 µm and ≥95% efficiency at ≥0.5 µm, verified per ISO 21501-4.
Can the device perform sequential multi-point sampling automatically?
Yes—users can predefine up to 99 sampling locations with independent cycle parameters; the unit executes fully autonomous sequences with geotagged metadata.
Is external calibration required annually?
Per JJF 1190–2008, annual recalibration by an accredited laboratory is recommended; the unit supports NIST-traceable check particle verification between calibrations.
How is data integrity ensured during wireless transmission?
All Wi-Fi and Bluetooth transmissions utilize AES-128 encryption and certificate-pinned TLS 1.2 handshakes; failed transfers trigger automatic retry with CRC-32 validation.