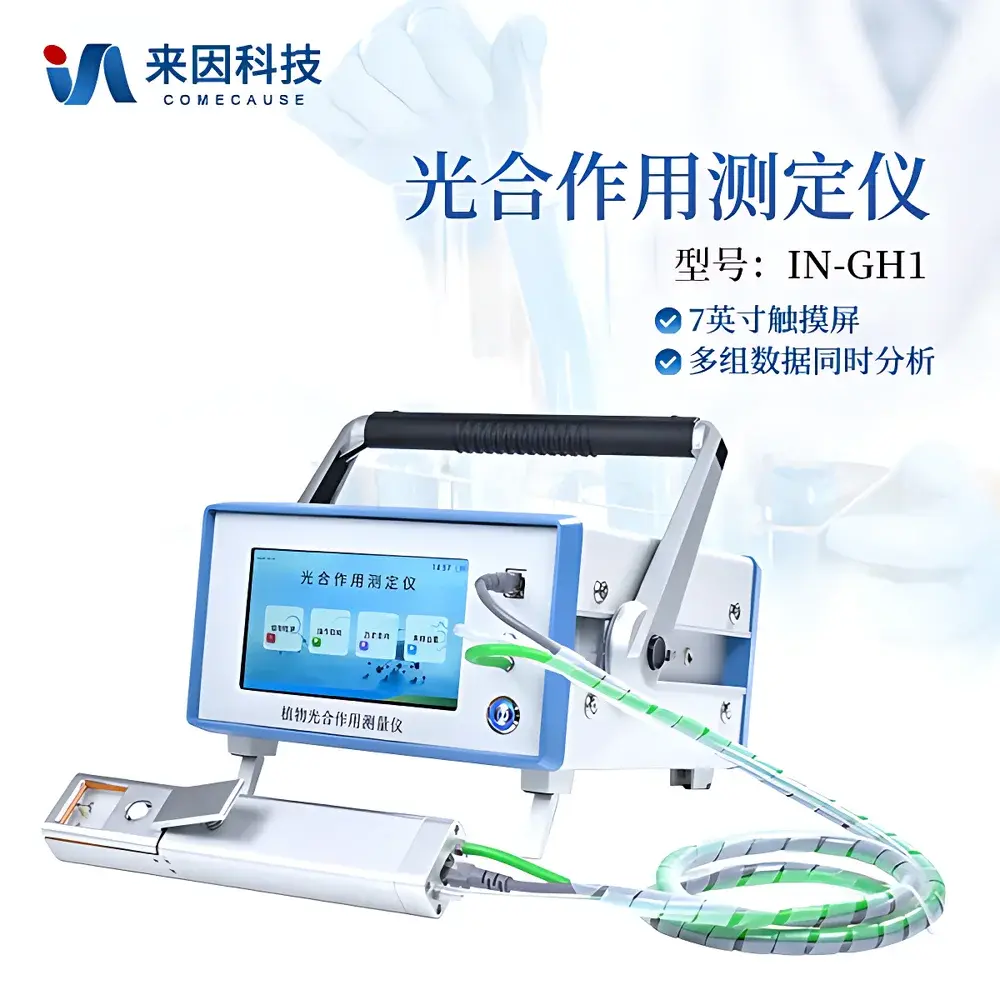
COMECAUSE IN-GH1 Portable Photosynthesis Measurement System
| Brand | COMECAUSE |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Model | IN-GH1 |
| CO₂ Measurement Range | 0–3000 μmol/mol (ppm) |
| CO₂ Accuracy | ≤3% FS |
| Operating System | Android |
| Display | 7-inch capacitive touchscreen |
| Battery Life | 10–12 hours (8000 mAh Li-ion) |
| Weight | 4.0 kg (main unit) + 0.7 kg (handheld chamber handle) |
| Dimensions | 312.3 × 308.5 × 186 mm (main unit) |
| Data Export | USB flash drive (plug-and-play, no driver required) |
| Environmental Sensors | Air/leaf temperature (0–50 °C, ±0.2 °C), RH (0–100% RH, ±1% RH), atmospheric pressure (30–110 kPa, ±0.06 kPa), PAR (0–3000 μmol·m⁻²·s⁻¹, ±5 μmol·m⁻²·s⁻¹) |
| Leaf Chamber | Standard 3.3 × 3.3 cm |
| Core Analyzer | Dual-wavelength NDIR CO₂ sensor with integrated temperature compensation and barometric pressure correction |
| Calculated Parameters | Net photosynthetic rate (Pn), stomatal conductance (Gs), transpiration rate (Tr), intercellular CO₂ concentration (Ci), water use efficiency (WUE), dark respiration rate (Rd), transpiration ratio (TR) |
Overview
The COMECAUSE IN-GH1 Portable Photosynthesis Measurement System is a field-deployable, open-system gas exchange analyzer engineered for high-fidelity, non-destructive quantification of leaf-level photosynthetic physiology. It operates on the principle of infrared gas analysis (NDIR) coupled with real-time environmental monitoring to compute key biophysical parameters—including net photosynthetic rate (Pn), stomatal conductance (Gs), transpiration rate (Tr), intercellular CO₂ concentration (Ci), and water use efficiency (WUE)—based on mass balance calculations of CO₂ and H₂O vapor fluxes across a sealed leaf chamber. The system employs dual-wavelength NDIR detection to minimize spectral interference and drift, while integrated temperature stabilization and barometric pressure compensation ensure measurement integrity under variable ambient conditions. Designed for both controlled-environment studies (growth chambers, greenhouses) and in situ field deployments (agricultural plots, forest understories, grasslands), the IN-GH1 delivers reproducible physiological data essential for plant ecophysiology, crop improvement, and ecosystem carbon modeling.
Key Features
- Android-based embedded operating system with intuitive 7-inch capacitive touchscreen interface—enabling real-time visualization of all measured and derived parameters during acquisition.
- Dual-wavelength non-dispersive infrared (NDIR) CO₂ analyzer with active thermal regulation and built-in barometric pressure sensor—reducing CO₂ signal drift caused by ambient temperature fluctuations and altitude-dependent pressure shifts.
- Comprehensive environmental sensing suite: simultaneous measurement of air/leaf temperature (±0.2 °C), relative humidity (±1% RH), atmospheric pressure (±0.06 kPa), and photosynthetically active radiation (PAR; ±5 μmol·m⁻²·s⁻¹).
- Modular handheld leaf chamber (3.3 × 3.3 cm aperture) with ergonomic handle design—optimized for single-operator field use and minimal mechanical stress on target foliage.
- Onboard ARM Cortex-A7 (RK3288, quad-core, 1.88 GHz) processor with 1 GB RAM and 16 GB internal storage—supporting multi-session logging, timestamped metadata tagging, and user-defined experimental notes.
- Extended battery autonomy: 10–12 hours of continuous operation on a rechargeable 8000 mAh Li-ion battery; status-indicated charging (red = charging, green = full).
- Plug-and-play USB data export—no drivers required; compatible with standard FAT32-formatted USB flash drives for immediate offline analysis.
Sample Compatibility & Compliance
The IN-GH1 accommodates broad leaf morphology across herbaceous and woody species—including monocots (e.g., maize, wheat), dicots (e.g., Arabidopsis, soybean), and perennial broadleaves (e.g., poplar, oak)—via its standardized 3.3 cm² chamber geometry and adjustable sealing gasket. Chamber clamping force is calibrated to avoid epidermal damage or stomatal occlusion while maintaining gas-tight integrity. All sensor calibrations adhere to traceable reference standards compliant with ISO 17025-accredited metrology practices. While not certified for GLP or FDA 21 CFR Part 11 out-of-the-box, the system supports audit-ready data logging (with time-stamped raw sensor outputs, user annotations, and session IDs), enabling integration into validated workflows under ISO 14001, ISO 9001, or institutional QA/QC protocols. Raw data files are exported in CSV format for interoperability with third-party statistical and modeling platforms (e.g., R, Python, MATLAB, SigmaPlot).
Software & Data Management
Data acquisition and visualization are managed through a proprietary Android application with multilingual UI support (English default). Each measurement session automatically logs synchronized time-series records for all 15 primary parameters—including CO₂ concentration (μmol/mol), Pn (μmol CO₂·m⁻²·s⁻¹), Gs (mmol H₂O·m⁻²·s⁻¹), Tr (mmol H₂O·m⁻²·s⁻¹), Ci (μmol/mol), WUE (μmol CO₂·mol⁻¹ H₂O), Rd (μmol CO₂·m⁻²·s⁻¹), and TR (μmol H₂O·mmol⁻¹ CO₂)—alongside environmental metadata (PAR, Tₐᵢᵣ, Tₗₑₐf, RH, Pₐₜₘ). Post-acquisition, users may import multiple CSV datasets for side-by-side comparative analysis, generate response curves (A–Q, A–Ci), and export annotated graphs in PNG or PDF. No cloud dependency: all processing occurs locally; no telemetry or remote data transmission is enabled by default.
Applications
- Plant Ecophysiology: Characterization of light-response (A–Q) and CO₂-response (A–Ci) curves to estimate Vcmax, Jmax, and mesophyll conductance using established Farquhar–von Caemmerer–Berry model frameworks.
- Crop Improvement: High-throughput phenotyping of photosynthetic efficiency across breeding populations under controlled or field conditions—supporting selection for enhanced WUE, drought resilience, or canopy-level carbon gain.
- Abiotic Stress Physiology: Quantitative assessment of photosynthetic inhibition kinetics under drought, heat, salinity, or ozone exposure—enabling early detection of stomatal vs. non-stomatal limitations.
- Ecosystem Carbon Flux Modeling: Ground-truthing of eddy-covariance tower data via leaf-scale measurements across dominant species in forests, grasslands, and wetlands—improving upscaling accuracy for regional carbon budgets.
- Controlled Environment Agriculture: Optimization of lighting spectra, CO₂ enrichment strategies, and irrigation scheduling based on real-time Pn and Tr feedback in vertical farms and greenhouse production systems.
FAQ
What is the measurement principle used by the IN-GH1?
The IN-GH1 uses an open-path, differential infrared gas analysis (NDIR) method to measure CO₂ and H₂O vapor concentrations upstream and downstream of a sealed leaf chamber, applying Fick’s law of diffusion to calculate net photosynthetic rate, stomatal conductance, and transpiration.
Can the instrument operate without external power in the field?
Yes—the integrated 8000 mAh lithium-ion battery supports 10–12 hours of continuous measurement, including PAR logging, environmental monitoring, and real-time computation.
Is calibration required before each use?
Zero calibration using ambient air is recommended prior to deployment; span calibration with certified CO₂ gas (e.g., 350 ppm or 1000 ppm) is advised weekly or after transport-induced shock to maintain ≤3% FS accuracy.
Does the system support automated light-response curve generation?
Yes—the Android interface allows programmable stepwise PAR ramping (e.g., 0 → 2000 μmol·m⁻²·s⁻¹ in 100 μmol increments) with user-defined dwell times per step.
How is data traceability ensured for regulatory or publication purposes?
Each CSV export includes full sensor timestamps, firmware version, chamber ID, operator ID (if entered), and raw analog-to-digital conversion values—enabling full reconstruction of derived parameters per established biophysical equations.