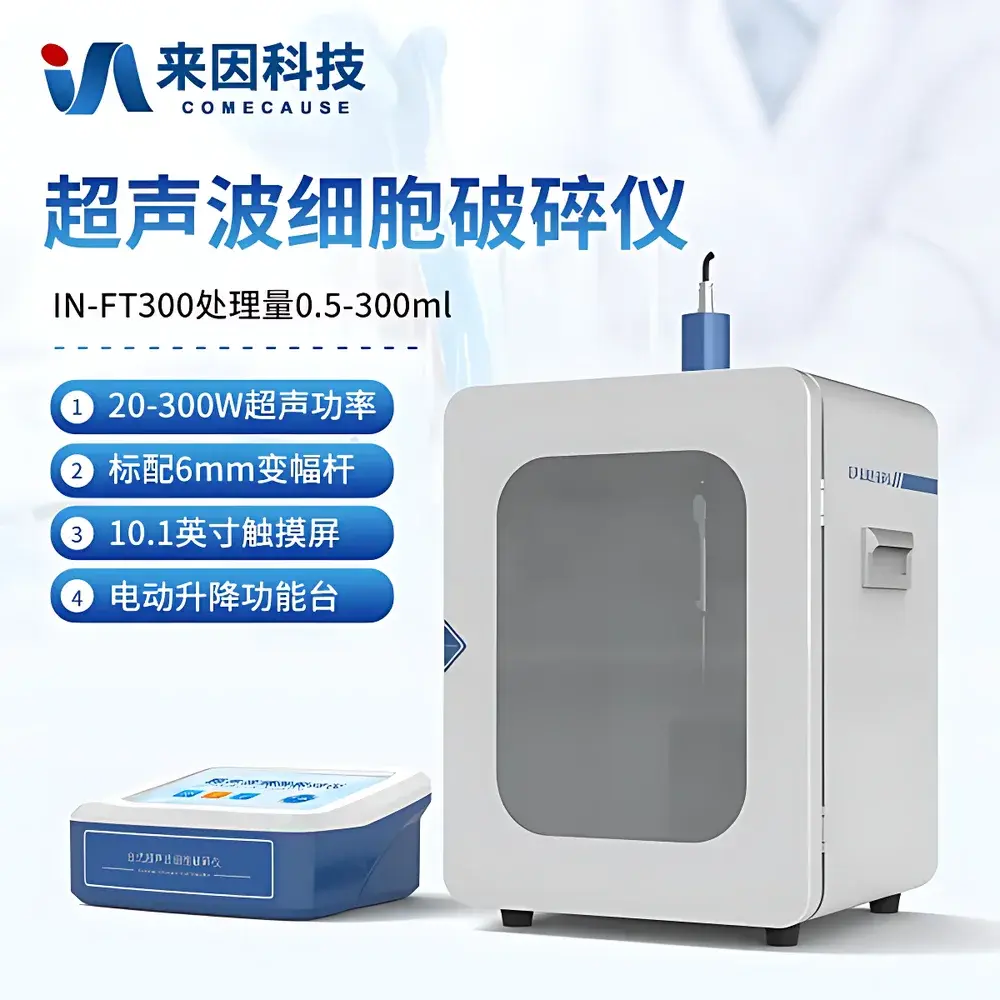
COMECAUSE IN-FT300 Ultrasonic Cell Disruptor with Integrated Soundproof Enclosure
| Brand | COMECAUSE |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Instrument Type | Ultrasonic Cell Disruptor |
| Model | IN-FT300 |
| Operating Frequency | 20–25 kHz |
| Ultrasonic Power Output | 20–300 W |
| Sample Volume Capacity | 0.1–300 mL (with interchangeable titanium alloy probes) |
| Pulse Control Resolution | 0.1 s (on/off/total time) |
| Temperature Monitoring Range | 0–99.9 °C (contact-type sensor) |
| Display | 10.1″ HD capacitive touchscreen |
| Probe Material | TC4 titanium alloy |
| Transducer Housing | Aerospace-grade magnesium-aluminum alloy |
| Data Storage | 256 programmable protocols |
| Compliance | CE-marked design principles |
Overview
The COMECAUSE IN-FT300 Ultrasonic Cell Disruptor with Integrated Soundproof Enclosure is an engineered solution for controlled, reproducible mechanical lysis of biological samples via high-intensity ultrasonic cavitation. It operates on the principle of acoustic cavitation—where rapid pressure fluctuations generated by 20–25 kHz longitudinal vibrations induce transient microbubble formation, growth, and violent collapse within liquid media. This process delivers localized shear forces exceeding 104 Pa and transient temperatures above 5,000 K at bubble collapse sites, sufficient to disrupt cell membranes, organelles, and microbial envelopes without significant thermal denaturation when operated under pulsed conditions. The system integrates a digitally regulated ultrasonic generator, a high-efficiency piezoelectric transducer (based on barium titanate-zirconate ceramics), and a replaceable TC4 titanium alloy probe assembly—all housed within a purpose-built, acoustically damped enclosure. Designed for benchtop deployment in regulated laboratory environments, it supports both routine sample preparation and method development across molecular biology, biopharmaceutical process R&D, and academic life science research.
Key Features
- 10.1″ medical-grade capacitive touchscreen interface with intuitive graphical workflow navigation and real-time parameter visualization (power, temperature, elapsed time, pulse status)
- Modular architecture: physically isolated generator unit and probe assembly minimize cross-contamination risk and simplify maintenance
- Integrated soundproof enclosure constructed from multi-layer composite panels (acrylic viewing window + vibration-damping metal chassis + internal acoustic absorption lining), achieving >30 dB(A) noise reduction versus open-bench operation
- Dual-mode ultrasonic control: continuous or precisely timed pulse sequences (on/off duration adjustable in 0.1 s increments; total runtime up to 999.9 min)
- Intelligent frequency tracking and automatic impedance matching ensure consistent energy delivery across probe wear cycles and sample viscosity changes
- TC4 titanium alloy probe variants (Φ2 mm to Φ10 mm) optimized for volume scalability—from microliter-scale bacterial lysates to 300 mL tissue homogenates
- Onboard contact-type temperature sensor with configurable overtemperature alarm (0–99.9 °C range); optional external chiller integration via rear-panel I/O port
- 256 protocol memory slots with timestamped parameter recall, supporting SOP-driven operation and audit-ready traceability
- Configurable security layer: password-protected access to advanced settings and calibration menus
- Rear-mounted expansion interface (7-pin aviation connector) enables synchronization with external peripherals—including recirculating chillers, magnetic stirrers, or industrial PLCs
Sample Compatibility & Compliance
The IN-FT300 accommodates a broad spectrum of biological matrices: Gram-positive and Gram-negative bacteria (including spores), yeast, mammalian and plant cells, subcellular fractions (e.g., mitochondria, nuclei), and soft-tissue homogenates. Its low-thermal-load pulsing capability preserves labile epitopes and enzymatic activity during lysis—critical for downstream applications such as native co-immunoprecipitation or functional proteomics. While not certified to ISO/IEC 17025 or FDA 21 CFR Part 11 out-of-the-box, the instrument’s deterministic parameter logging, non-volatile protocol storage, and hardware-level interlocks align with core GLP and GMP documentation requirements. Its electrical safety design conforms to IEC 61010-1:2010 standards for laboratory equipment, and electromagnetic compatibility follows EN 61326-1:2013 Class B specifications.
Software & Data Management
All operational parameters—including power setpoint, duty cycle, cumulative exposure time, and recorded temperature history—are stored locally in non-volatile memory with millisecond-level timestamp resolution. Protocols may be exported via USB to CSV or XML format for integration into LIMS or electronic lab notebook (ELN) systems. The interface supports bilingual operation (English/Chinese) with factory-default English configuration. No proprietary cloud service or vendor-hosted software is required; data integrity relies solely on local hardware-based storage and user-controlled export workflows.
Applications
- Preparative lysis for recombinant protein extraction and inclusion body solubilization
- Nucleic acid isolation from fibrous or lipid-rich tissues (e.g., liver, brain, adipose)
- Preparation of antigen-presenting cell lysates for immunological assays
- Disruption of biofilm-embedded microbes for antimicrobial susceptibility testing
- Generation of nanosuspensions and nanoemulsions in formulation development
- Accelerated solvent extraction of phytochemicals and natural products
- Deagglomeration of inorganic nanoparticles (e.g., rare-earth oxides, metal-organic frameworks) in colloidal stabilization studies
- Ultrasonic-assisted crystallization and polymorph screening in solid-state chemistry
FAQ
What safety certifications does the IN-FT300 hold?
The instrument complies with IEC 61010-1:2010 for electrical safety and EN 61326-1:2013 for electromagnetic compatibility. It carries CE marking per EU Directive 2014/30/EU (EMC) and 2014/35/EU (LVD). Formal ISO 13485 or FDA registration is not applicable, as it is classified as general-purpose laboratory equipment.
Can the system operate under refrigerated conditions?
Yes—via optional integration with an external recirculating chiller connected through the rear I/O port. The built-in temperature sensor monitors sample temperature only; active cooling requires external thermal management.
Is probe sterilization supported?
TC4 titanium alloy probes are autoclavable at 121 °C for 20 minutes. Repeated autoclaving may affect surface finish and acoustic coupling efficiency; users should verify performance post-sterilization using standardized test samples.
How is acoustic output calibrated?
Calibration is performed using traceable hydrophone measurements per ASTM E1002-19 (Standard Practice for Calibration of Ultrasonic Medical Equipment). Factory calibration certificates are provided; field recalibration requires OEM-specified instrumentation and firmware tools.
Does the system support remote monitoring or network connectivity?
No Ethernet or Wi-Fi interface is included. Data transfer is limited to USB mass-storage mode. Networked operation requires third-party hardware gateways or custom API development via the serial-over-USB interface (available upon request under NDA).