COMECAUSE IN-J200 Automated Colony Morphology Analyzer & Colony Counter
| Brand | COMECAUSE |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Instrument Type | Fully Automated Colony Counter |
| Sample Type | Standard Petri Dishes (90–110 mm diameter), 3M Petrifilm™, Compact Dry™, RIDA™ Filter Membranes, Fluorescent & Non-fluorescent Microbial Colonies |
| Imaging System | Sealed Light-Tight Dark Chamber with Stray-Light Elimination |
| Illumination | Dual-Independent Adjustable Top/Bottom LED Light Sources |
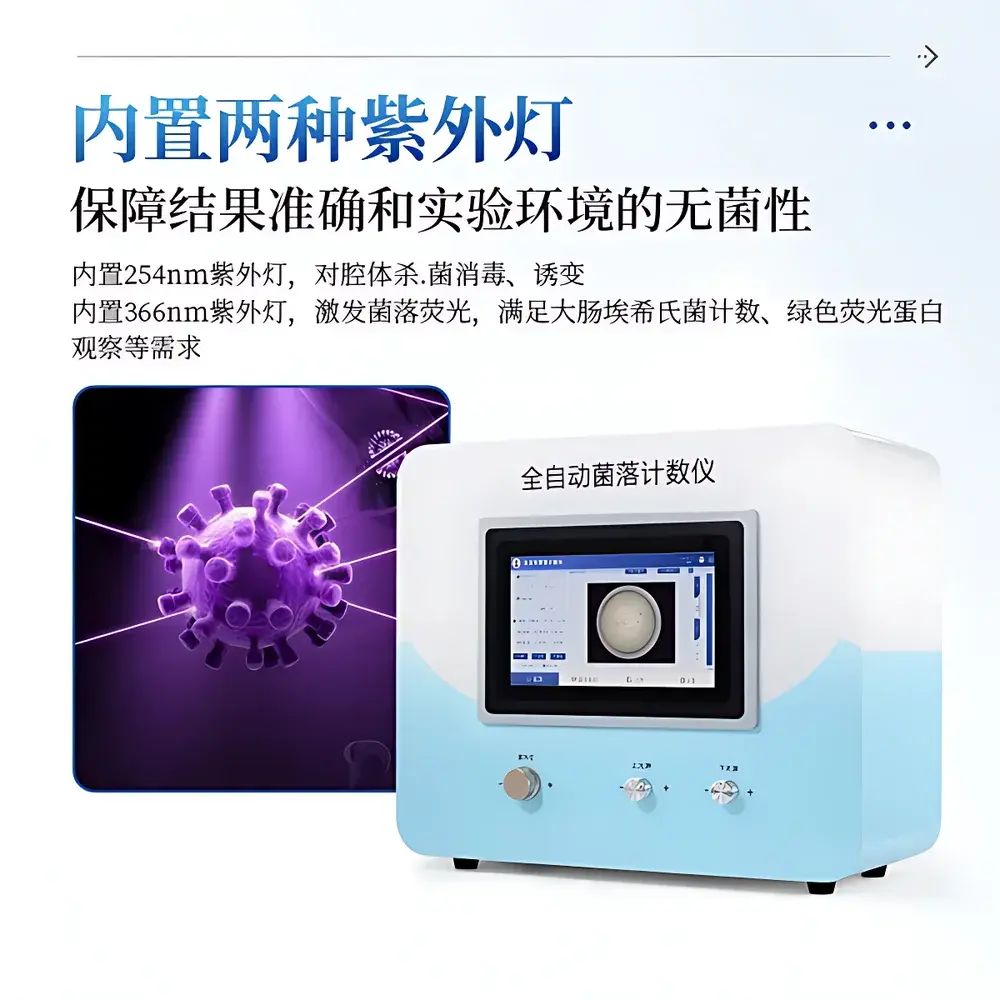
| UV Modules | 254 nm (sterilization & mutagenesis), 366 nm (fluorescence excitation for *E. coli*, GFP, etc.) |
| Camera | 16 MP Full-Color CMOS Sensor |
| Minimum Detectable Colony Size | 0.1 mm |
| Counting Accuracy | ≤ ±10% |
| Display | 15.6″ Touchscreen (1920 × 1080), Windows 10 64-bit OS, 256 GB Internal Storage |
| Software | Integrated Colony Recognition Engine with Multi-Mode Segmentation Algorithms |
| Compliance | Supports GLP-compliant audit trails, user-level access control, immutable PDF reporting, bilingual (EN/CN) UI |
Overview
The COMECAUSE IN-J200 Automated Colony Morphology Analyzer is an integrated digital imaging platform engineered for high-fidelity microbial colony enumeration and morphological characterization in regulated and research-grade microbiology laboratories. It operates on the principle of high-resolution optical image acquisition followed by adaptive pixel-based segmentation and feature extraction—leveraging calibrated illumination geometry, spectral discrimination, and morphology-aware machine vision algorithms to distinguish colonies from background agar or membrane substrates. Unlike manual or semi-automated counters reliant on threshold-based binary segmentation, the IN-J200 implements multi-modal recognition logic—including size distribution profiling, edge contrast optimization, color-space clustering, and fluorescence channel separation—to resolve overlapping, low-contrast, micro-colonies (≥0.1 mm), and mixed-species populations on heterogeneous media. Its sealed dark chamber architecture eliminates ambient light interference, ensuring reproducible photometric conditions across repeated assays—a prerequisite for longitudinal monitoring and inter-laboratory comparability.
Key Features
- Sealed optical dark chamber with vibration-damped stage and integrated 254 nm UV lamp for internal cavity sterilization between runs, minimizing cross-contamination risk.
- Dual independently controllable LED illumination system: top-light (reflected mode) and bottom-light (transmitted mode), each with continuous brightness adjustment via rotary encoder—enabling optimal contrast enhancement for opaque colonies, translucent molds, or membrane-filtered samples.
- Dedicated 366 nm UV excitation source for fluorescent colony detection, validated for *Escherichia coli* enumeration on chromogenic media and GFP-expressing recombinant strains.
- 16-megapixel color CMOS sensor with fixed-focus macro lens; factory-calibrated for consistent spatial resolution across 90–110 mm Petri dishes and standardized test films (3M Petrifilm™, Compact Dry™, RIDA™).
- On-device embedded computing: Intel Core i5 processor, 8 GB RAM, 256 GB SSD, Windows 10 IoT Enterprise OS—no external PC required; touchscreen interface supports glove-compatible operation.
- Automated geometric calibration: system self-validates optical path parameters (lens focal length, working distance, sensor pitch) at boot; enables traceable colony size measurement in millimeters without user intervention.
Sample Compatibility & Compliance
The IN-J200 accommodates standard microbiological sample formats including poured agar plates (90–110 mm), spread plates, membrane filtration assemblies (e.g., Millipore HA, Sartorius SM11192), and commercial rapid-test films (3M Petrifilm™ Aerobic Count, Compact Dry™ TC, RIDA®COUNT). Its algorithm suite includes dedicated modules for each substrate type—applying optimized noise suppression, dynamic range normalization, and region-of-interest masking to maintain counting fidelity across variable backgrounds. From a regulatory perspective, the instrument supports Good Laboratory Practice (GLP) and current Good Manufacturing Practice (cGMP) workflows through role-based user authentication, electronic signature-capable audit logs, immutable PDF report generation with embedded raw images and metadata (timestamp, operator ID, plate ID, analysis parameters), and full data retention history. While not FDA 21 CFR Part 11-certified out-of-the-box, its architecture complies with foundational requirements for electronic records integrity—making it suitable for pre-validation deployment in pharmaceutical QC, food safety labs, and academic core facilities conducting ISO/IEC 17025-aligned testing.
Software & Data Management
The proprietary COMECAUSE ColonyVision™ software provides a unified workflow—from image capture to statistical export—within a single interface. It features eight distinct counting modes: Dispersed Colony Mode (for isolated colonies with sharp edges), Aggregated Colony Mode (morphology-invariant watershed segmentation), Micro-Colony Mode (enhanced sensitivity for 0.1–0.5 mm entities), 3M Petrifilm™ Mode (grid-aligned colony isolation), Compact Dry™ Mode (black-line contrast inversion), RIDA™ Filter Mode (membrane pore compensation), Chromogenic Mode (RGB-channel selective thresholding), and Multi-Color Mode (interactive hue-range selection for differential enumeration). Post-processing tools include adjustable binarization thresholds, manual correction via brush/ellipse/rectangle ROI tools, and automatic colony size classification into 25 discrete diameter bins. All results are stored locally with SHA-256 hash verification; reports export as PDF with embedded EXIF metadata and optional CSV exports for LIMS integration. The bilingual UI (English/Chinese) supports seamless switching without restart.
Applications
The IN-J200 serves critical functions across multiple domains requiring quantitative microbial assessment. In food and beverage quality control, it enables rapid aerobic plate count (APC), coliform screening, and probiotic viability assessment—reducing turnaround time from hours to minutes while improving inter-operator consistency. In environmental microbiology, it quantifies heterotrophic plate counts (HPC) in water samples processed via membrane filtration, supporting compliance with EPA Method 1603 and ISO 9308-1. For fermentation process development, temporal colony imaging allows growth kinetics modeling—tracking changes in mean colony diameter, circularity, and dispersion index as proxies for population vitality and phenotypic stability. In agricultural biotechnology, researchers use it to quantify rhizobacterial inoculant persistence in soil extracts plated on selective media, informing dosing regimens under ISO 11092. Clinical and pharmaceutical labs apply it for environmental monitoring (ISO 14644-1 cleanroom settle plates), sterility test membrane enumeration (USP <71>), and antimicrobial efficacy testing (ASTM E2149, ISO 22196).
FAQ
Does the IN-J200 require external calibration standards or daily maintenance beyond UV sterilization?
No. The system performs automated geometric calibration at startup using embedded reference patterns; only periodic cleaning of the glass viewport and UV lamp quartz sleeve is recommended.
Can the software distinguish between bacterial and fungal colonies based on morphology alone?
It classifies colonies by measurable features (diameter, circularity, texture, edge gradient) but does not perform taxonomic identification; morphological clusters may correlate with broad categories (e.g., yeast vs. mold) when trained on representative reference sets.
Is raw image data export supported for third-party analysis?
Yes—TIFF and PNG formats are available via USB or network share, preserving full 16-bit depth and unprocessed pixel values.
What is the maximum throughput per hour under standard operating conditions?
Approximately 40–50 standard Petri dishes (90 mm), depending on colony density and selected analysis mode; batch processing queues support unattended overnight operation.
How does the system handle plates with condensation or scratches on the lid?
The dark chamber design minimizes lid reflection artifacts; software includes dew detection filters and scratch-aware background subtraction to suppress false positives from surface imperfections.