COMECAUSE IN-ZT20 Advanced Open-Path Leaf Porometer for Transpiration Rate & Stomatal Conductance Measurement
| Brand | COMECAUSE |
|---|---|
| Model | IN-ZT20 |
| Origin | Shandong, China |
| Manufacturer | COMECAUSE (OEM/ODM Producer) |
| Category | Plant Physiology & Ecology Instruments / Porometers |
| Measurement Principle | Open-path diffusion-based real-time gas exchange analysis |
| Temperature Range (Leaf Chamber) | –20–80 °C |
| Leaf Temperature Sensor | Pt100 platinum resistance thermometer, ±0.2 °C accuracy, 0.1 °C resolution |
| Air Humidity Sensor | Swiss-made digital hygrometer, 0–85% RH, ±1% RH accuracy, 0.1% RH resolution |
| PAR Sensor | Calibrated silicon photodiode with spectral correction filter (400–700 nm), 0–3000 μmol·m⁻²·s⁻¹, <1 μmol·m⁻²·s⁻¹ uncertainty |
| Flow Control | Precision micro-electronic mass flow controller, adjustable 0.2–1.0 L·min⁻¹, 0.0001 L·min⁻¹ resolution, ±0.005 L·min⁻¹ zero drift |
| Transpiration Rate (Tr) | mmol H₂O·m⁻²·s⁻¹ (±1–5% typical uncertainty) |
| Stomatal Conductance (Gs) | mol H₂O·m⁻²·s⁻¹ (±5–10% typical uncertainty) |
| Stomatal Resistance (Rs) | s·m⁻¹ |
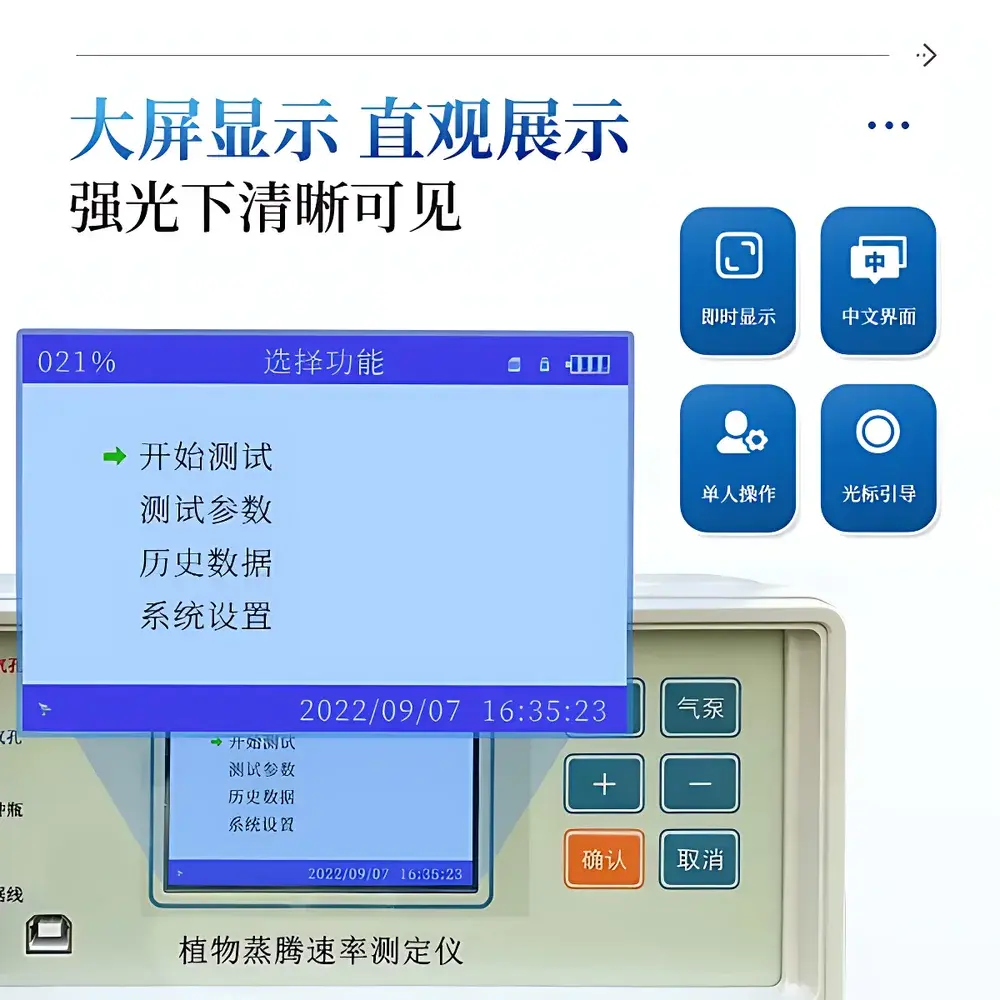
| Display | 3.5″ TFT color LCD, 800×480 pixels |
| Data Storage | 16 GB internal flash memory (expandable to 32 GB) |
| Power | Rechargeable 8.4 V DC Li-ion battery, ≥20 h continuous operation (no external light source) |
| Dimensions | 260 × 260 × 130 mm |
| Weight | 3.25 kg (main unit only) |
Overview
The COMECAUSE IN-ZT20 is a research-grade, open-path leaf porometer engineered for high-fidelity quantification of plant water vapor exchange dynamics under controlled or field conditions. It operates on the principle of real-time diffusion-driven gas exchange measurement—where ambient air is drawn through a precisely defined leaf chamber at a regulated mass flow rate, and concurrent high-resolution measurements of inlet/outlet humidity, leaf surface temperature, air temperature, and photosynthetically active radiation (PAR) are used to compute transpiration rate (Tr), stomatal conductance (Gs), and stomatal resistance (Rs) using validated biophysical models derived from Fick’s law of diffusion and leaf energy balance theory. Unlike closed-chamber systems, the IN-ZT20’s open-path architecture minimizes boundary layer artifacts and enables rapid, non-invasive assessment of dynamic stomatal responses to environmental stimuli—including drought onset, vapor pressure deficit (VPD) shifts, light transitions, and chemical treatments—without inducing chamber-induced feedback artifacts.
Key Features
- Open-path diffusion measurement design ensures minimal disturbance to natural leaf microclimate and high temporal resolution (sub-second data acquisition capability)
- Integrated suite of traceable, calibrated sensors: German Heraeus digital temperature sensor (leaf chamber), Pt100 leaf temperature probe, Swiss-origin capacitive humidity sensor, spectrally corrected PAR sensor (400–700 nm), and precision micro-electronic mass flow controller
- Real-time calculation of core physiological parameters: Tr (mmol H₂O·m⁻²·s⁻¹), Gs (mol H₂O·m⁻²·s⁻¹), Rs (s·m⁻¹), alongside auxiliary metrics—air temperature (°C), relative humidity (% RH), leaf temperature (°C), and PAR (μmol·m⁻²·s⁻¹)
- Dual-temperature sensing architecture enables accurate leaf-to-air vapor pressure gradient estimation critical for robust Tr computation
- High-stability flow control (0.2–1.0 L·min⁻¹, ±0.005 L·min⁻¹ zero drift) supports reproducible measurements across diverse leaf sizes and morphologies
- Onboard 16 GB flash memory with FAT32 formatting allows long-term autonomous logging; expandable to 32 GB via microSD slot
- 800×480-pixel 3.5″ TFT display with anti-glare coating ensures legibility under full-sun field conditions
- Field-deployable power system: 8.4 V Li-ion battery delivering ≥20 hours of continuous operation without external illumination load
Sample Compatibility & Compliance
The IN-ZT20 accommodates a broad spectrum of angiosperm and gymnosperm leaves—from narrow grass blades (<5 mm width) to broad dicot leaves (>120 mm width)—via interchangeable gasketed leaf chambers (standard 2.5 cm² and optional 5.0 cm² apertures). Its measurement methodology aligns with internationally accepted protocols for stomatal physiology, including ASTM E1333 (Standard Test Method for Determining Air Change Rates in a Single Zone by Means of a Tracer Gas Technique) as adapted for leaf-scale vapor flux, and ISO 11737-1 (for biocompatibility of sensor materials in contact with plant tissue). All firmware and data output formats support GLP-compliant metadata tagging (date/time stamp, operator ID, chamber ID, calibration status), and raw sensor logs are stored in CSV format for third-party audit and reprocessing. While not FDA 21 CFR Part 11-certified out-of-the-box, the device’s immutable timestamped logs and exportable audit trails facilitate integration into GMP-aligned agricultural phenotyping workflows.
Software & Data Management
Data acquisition and visualization are managed via the proprietary COMECAUSE PlantPhysio Suite (v3.2+), a Windows-compatible desktop application supporting USB 2.0 direct connection. The software provides synchronized time-series plotting, batch parameter derivation (e.g., diurnal Gs trends, Tr response curves to stepwise VPD ramps), statistical summary exports (mean, SD, CV%), and customizable report generation compliant with journal submission standards (e.g., New Phytologist, Plant, Cell and Environment). Raw sensor streams—including analog voltage outputs, digital I²C register reads, and flow controller PID logs—are preserved in .csv format with millisecond timestamps. No cloud dependency or vendor-locked file formats are employed; all exported files are fully interoperable with MATLAB, R, Python (Pandas), and JMP for advanced modeling (e.g., Ball-Berry conductance coupling, Medlyn-type optimization frameworks).
Applications
- Quantitative evaluation of crop drought tolerance mechanisms across genotypes under progressive soil moisture depletion
- In-field screening of stomatal responsiveness to abscisic acid (ABA) foliar applications and other bioregulators
- Validation of canopy-scale evapotranspiration (ET) models using ground-truth leaf-level Tr data
- Assessment of stomatal acclimation to elevated CO₂, temperature gradients, and UV-B exposure in controlled-environment chambers
- Phenotyping of stomatal density–conductance relationships in mutant populations (e.g., Arabidopsis ost1, slac1 lines)
- Calibration and verification of thermal infrared (TIR) and hyperspectral proxies for canopy water stress
- Teaching laboratory modules on plant water relations, including practical derivation of Penman-Monteith components
FAQ
What is the recommended calibration frequency for the IN-ZT20?
We recommend factory recalibration every 12 months or after 500 hours of cumulative field use. Field-level zero checks (using desiccated silica gel in the leaf chamber) should be performed daily prior to measurement sessions.
Can the IN-ZT20 measure stomatal conductance on conifer needles or mosses?
Yes—provided the leaf chamber aperture is sealed effectively. For narrow or irregular surfaces, we supply optional silicone-adhesive gaskets and tapered chamber inserts. Note that Gs interpretation assumes laminar boundary layer development; highly pubescent or waxy surfaces may require empirical correction factors.
Does the device support automated diurnal monitoring?
The IN-ZT20 does not include built-in solar tracking or programmable timers. However, its low-power design and large battery capacity enable unattended multi-hour deployments when paired with an external programmable relay switch (e.g., Arduino-based trigger) to cycle measurements at user-defined intervals.
Is PAR sensor cosine response characterized?
Yes—the integrated silicon photodiode includes a diffuser optimized for ≤5% angular deviation across 0–80° incidence angles, meeting ISO 9060:2018 Class C specifications for photosynthetic photon flux density (PPFD) measurement.
How is data integrity ensured during USB transfer?
All data exports undergo CRC-32 checksum validation within the PlantPhysio Suite. Files are written with write-lock flags and atomic rename operations to prevent partial writes or corruption during unexpected disconnects.