COMECAUSE IN_LeafClear Chlorophyll Fluorescence Imaging System
| Brand | COMECAUSE |
|---|---|
| Model | IN_LeafClear |
| Camera Resolution | 1608 × 1104 |
| Pixel Size | 9 µm × 9 µm |
| Frame Rate | 100 fps |
| Bit Depth | 12-bit |
| Interface | USB 3.0 |
| Lens Focal Length | 12 mm |
| Max Aperture | F/2.8 (adjustable) |
| HFOV | 62.11° |
| VFOV | 44.83° |
| Max Imaging Area | 50 cm × 35 cm |
| Excitation Wavelengths | 450 nm (blue), 630 nm (red), 730 nm (far-red) |
| LED Intensity Range | 1–100% (up to 1440 µmol·m⁻²·s⁻¹) |
| OJIP Acquisition Time | 0.1–1.0 s (min Δt = 10 ms) |
| PAM Sampling Interval | 0.1–10.0 s (min Δt = 100 ms) |
| Fluorescence Dynamic Range | 0–4095 (12-bit), SNR > 100:1, Repeatability CV < 3%, Linearity R² > 0.999 |
| Spatial Resolution | ~0.3 mm/pixel at standard working distance |
| Parameter Accuracy | Fv/Fm ±0.005, ΦPSII ±0.01, qP/qN ±0.02, NPQ ±0.1, PIABS ±0.05 |
Overview
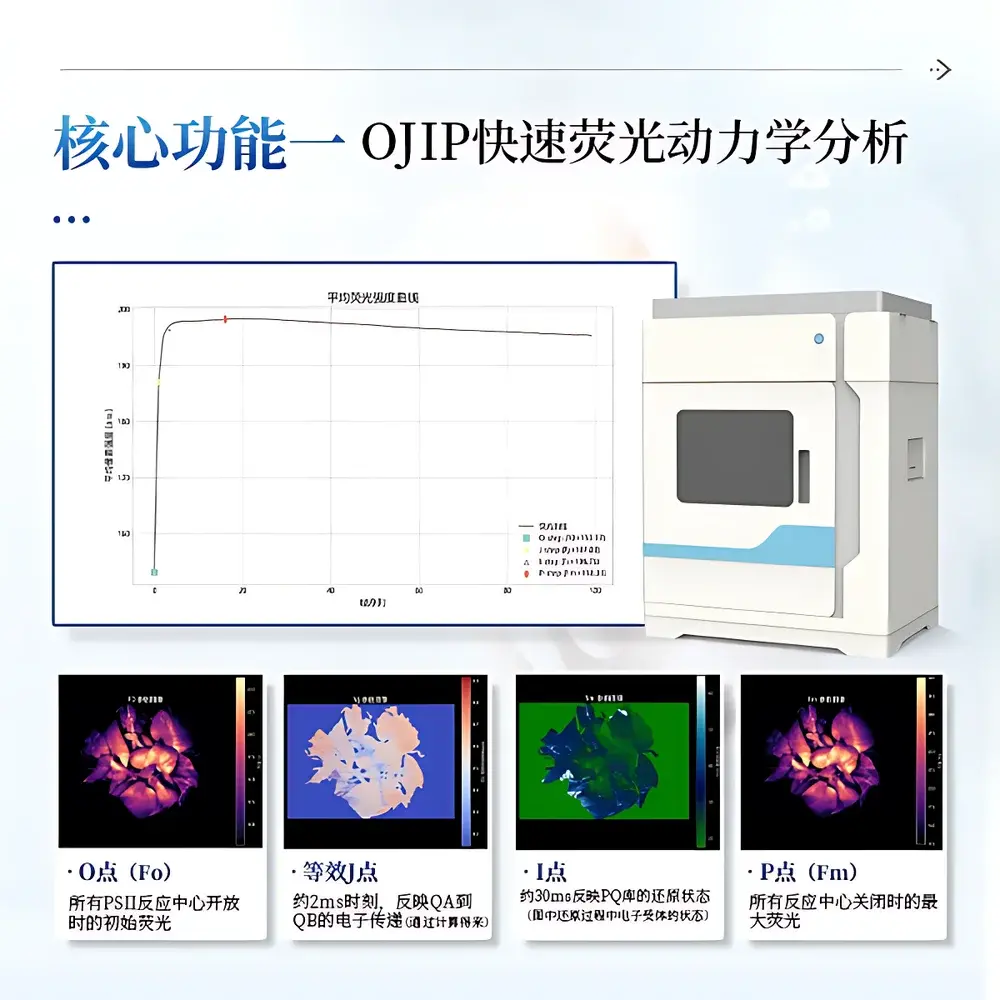
The COMECAUSE IN_LeafClear Chlorophyll Fluorescence Imaging System is a high-resolution, non-invasive plant phenotyping platform engineered for quantitative analysis of photosynthetic performance in intact leaves and whole-plant canopies. It operates on the biophysical principle of chlorophyll a fluorescence induction kinetics—leveraging the intrinsic optical signature of Photosystem II (PSII) reaction centers to report on electron transport efficiency, photochemical quenching, and energy dissipation pathways. The system integrates two complementary measurement paradigms: (1) OJIP transient analysis, capturing millisecond-scale fluorescence rise from Fo to Fm under saturating actinic light; and (2) Pulse-Amplitude Modulated (PAM) fluorometry, enabling dynamic tracking of PSII quantum yield (ΦPSII), non-photochemical quenching (NPQ), and photochemical quenching (qP) under controlled light acclimation regimes. Designed for reproducible, standardized physiological phenotyping, the IN_LeafClear complies with foundational principles outlined in ISO 10211 (optical measurement systems) and supports experimental traceability aligned with GLP-compliant workflows.
Key Features
- High-fidelity CMOS imaging sensor (1608 × 1104 pixels, 12-bit depth, 100 fps frame rate) optimized for low-light fluorescence detection with SNR > 100:1 and repeatability CV < 3%
- Dual-mode excitation architecture: tunable-intensity 450 nm blue LEDs (for OJIP saturation pulses and Fm determination), 630 nm red LEDs (for actinic illumination in PAM mode), and 730 nm far-red LEDs (for QA reoxidation during Fo′ measurement)
- Precise optical calibration: fixed 12 mm lens (F/2.8 adjustable aperture), 62.11° horizontal and 44.83° vertical field of view, delivering uniform illumination and spatial resolution of ~0.3 mm/pixel across a 50 cm × 35 cm imaging area
- Modular hardware control interface with real-time device status monitoring, automatic camera/LED controller enumeration, and fail-safe exposure synchronization (<1 ms LED response time)
- Graphical user interface supporting real-time bilingual operation (English/Chinese), parameter tooltips referencing established photobiological conventions (e.g., Strasser et al. JIP-test nomenclature, Schreiber et al. PAM definitions), and persistent configuration storage
Sample Compatibility & Compliance
The IN_LeafClear accommodates detached leaves, potted seedlings, rosettes, and small canopy segments up to 35 cm in height. Its non-destructive, contactless design enables longitudinal monitoring of individual plants across developmental stages or stress treatments without mechanical perturbation. The system adheres to internationally recognized measurement standards for chlorophyll fluorescence: OJIP acquisition follows the protocol defined in the “OJIP Test” framework (Strasser et al., 2004), while PAM measurements conform to the methodology described by Schreiber et al. (1986) and subsequent ISO/IEC technical reports on modulated fluorometry. All parameter calculations—including Fv/Fm, ΦPSII, NPQ, qP, PIABS, and JIP-test-derived flux ratios (e.g., TRo/RC, ETo/RC)—are implemented using peer-reviewed algorithms published in Photosynthesis Research and Plant Physiology. Data provenance is preserved via embedded metadata (timestamp, LED intensity, exposure duration, ambient temperature logging interface), supporting audit readiness for ISO 17025-accredited laboratories and FDA 21 CFR Part 11–aligned environments where electronic records require integrity controls.
Software & Data Management
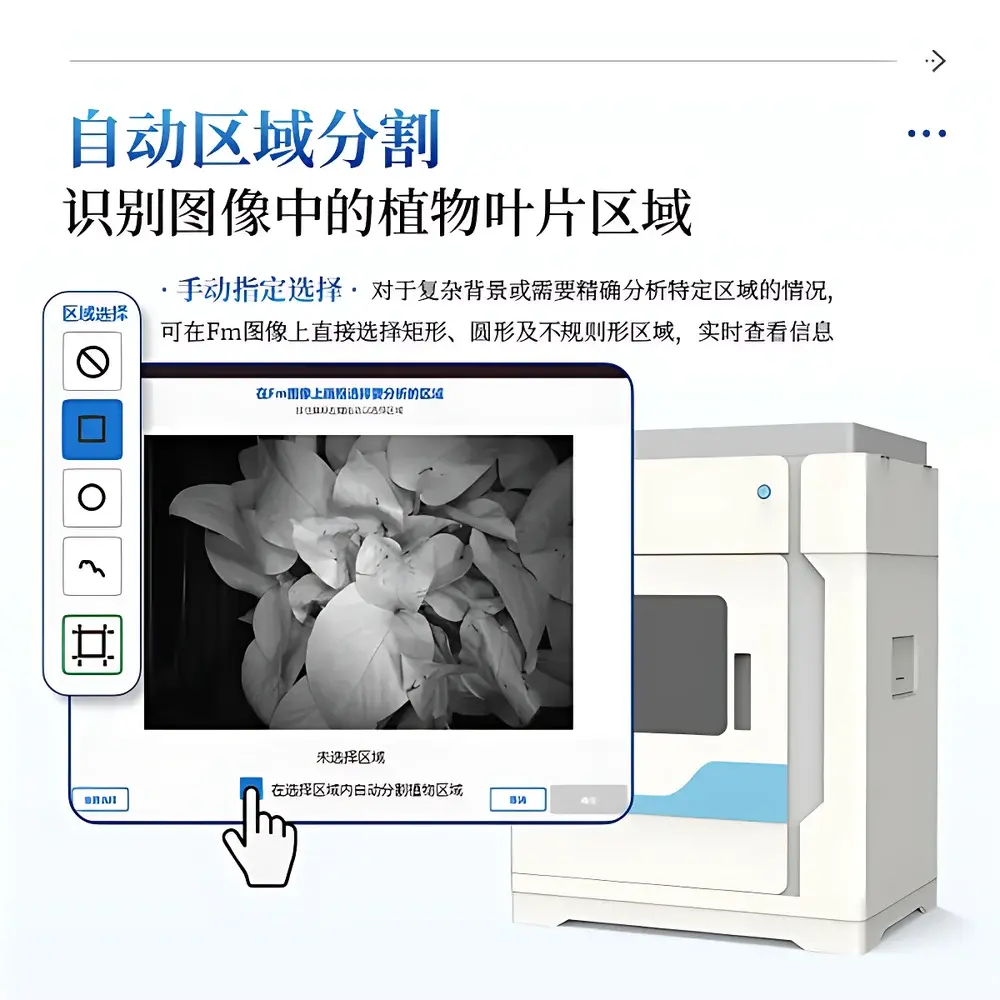
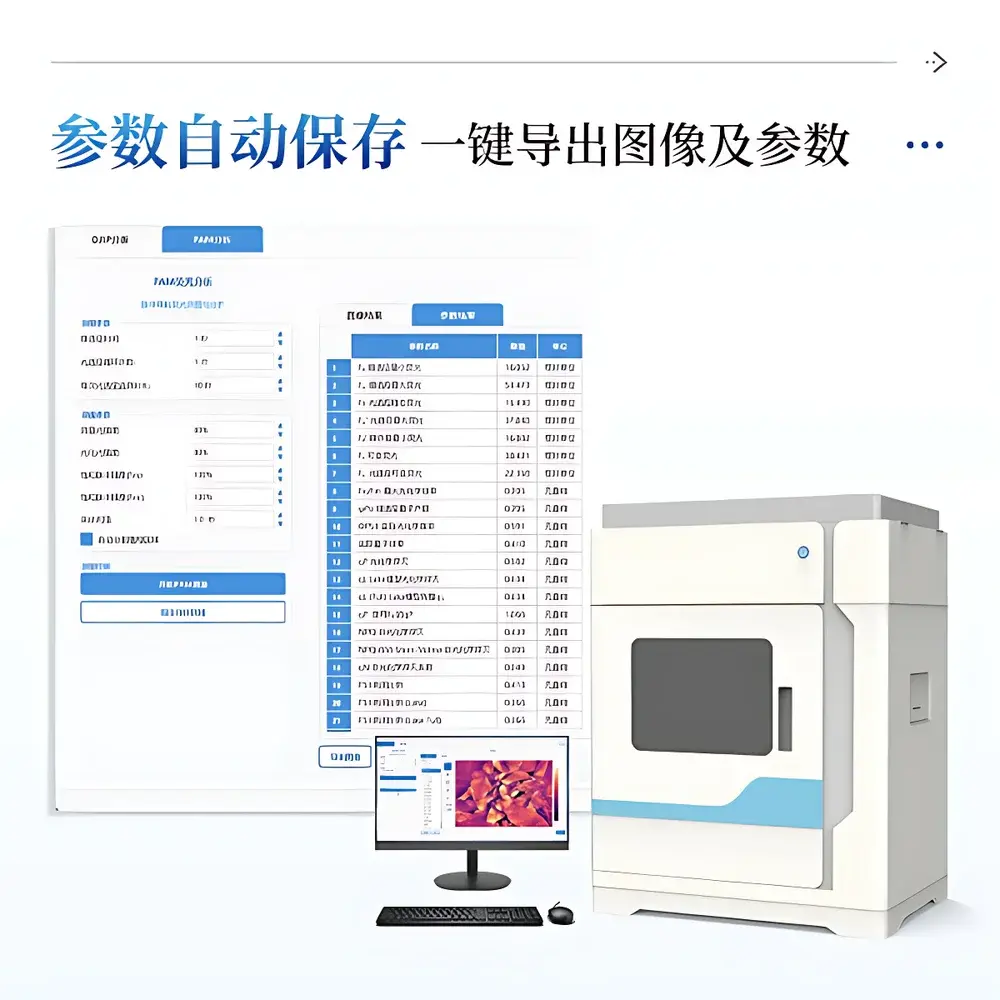
The proprietary IN_LeafClear Analysis Suite provides end-to-end digital workflow support—from acquisition through publication-grade visualization. Core capabilities include: automated leaf segmentation using adaptive thresholding and morphological filtering; manual ROI selection (rectangular, circular, polygonal) with live mean-intensity feedback; multi-parameter image mapping using scientifically validated pseudocolor schemes (Jet, Viridis); synchronized temporal curve plotting for OJIP transients, relative variable fluorescence (Vt), and PAM time-series (Fs, Fm′, Fo′); and export of calibrated images (PNG, 16-bit TIFF), parameter tables (CSV, XLSX), and metadata logs in FAIR-compliant structure. All exported files follow a deterministic naming convention (“Mode_SeqNo_ParameterName”) to ensure unambiguous data lineage. Software architecture includes built-in audit trail functionality: every parameter modification, ROI annotation, or export action is timestamped and logged with operator ID (if configured), satisfying GLP documentation requirements for regulated research.
Applications
- Photosynthetic Mechanism Studies: Quantification of PSII antenna size (ABS/RC), electron transport capacity (ETo/RC), and thermal dissipation efficiency (DIo/RC) via JIP-test analysis; discrimination between photoinhibitory damage and regulatory NPQ responses using kinetic PAM profiling
- Abiotic Stress Phenotyping: Early detection of drought-induced decline in Fv/Fm (>5% reduction precedes visible wilting); thermal tolerance screening via ΦPSII stability and NPQ relaxation kinetics after heat shock; salinity impact assessment through Fo elevation and PIABS suppression
- Agronomic Trait Evaluation: High-throughput varietal screening for nitrogen-use efficiency (correlated with ΦPSII under limiting N), herbicide mode-of-action validation (e.g., DCMU-induced Fv/Fm collapse), and irrigation scheduling optimization based on spatial heterogeneity maps of qP distribution
- Ecophysiological Field Monitoring: Portable deployment for canopy-level fluorescence imaging in controlled-environment chambers or greenhouse modules; integration with environmental sensors (PAR, Tair, RH) for multivariate stress modeling
FAQ
What is the minimum detectable change in Fv/Fm that the IN_LeafClear can resolve?
The system achieves an absolute accuracy of ±0.005 for Fv/Fm, enabling statistically robust discrimination of ≤2% physiological shifts under controlled conditions.
Can the IN_LeafClear be used for time-lapse imaging over multiple days?
Yes—its non-invasive nature and programmable dark-adaptation timing (0–3600 s) support repeated measurements on the same plant at defined intervals, provided consistent positioning and ambient light control are maintained.
Does the software support batch processing of OJIP datasets for statistical comparison?
Yes—the Analysis Suite includes group-level statistical tools (ANOVA, Tukey HSD) with automatic parameter normalization and significance overlay on false-color maps.
Is remote operation possible via network connection?
The current version supports local USB 3.0 operation only; however, exported CSV/XLSX files are fully compatible with third-party analysis platforms (R, Python/Pandas, MATLAB) for cloud-based processing pipelines.
How is calibration traceability maintained across instrument units?
Each system ships with a NIST-traceable fluorescence reference standard (certified Rhodamine B film), and factory calibration coefficients for LED irradiance (µmol·m⁻²·s⁻¹) and pixel gain are embedded in firmware and accessible via diagnostic mode.