COMECAUSE IN*J200 Automated Colony Counter for Food Safety Microbiology Testing
| Brand | COMECAUSE |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Instrument Type | Fully Automated Colony Counter |
| Sample Type | Petri Dishes |
| Sample Diameter Compatibility | 90–110 mm |
| Counting Accuracy | ≤ ±10% |
| Imaging Resolution | 1920 × 1080 (16 MP Color Camera) |
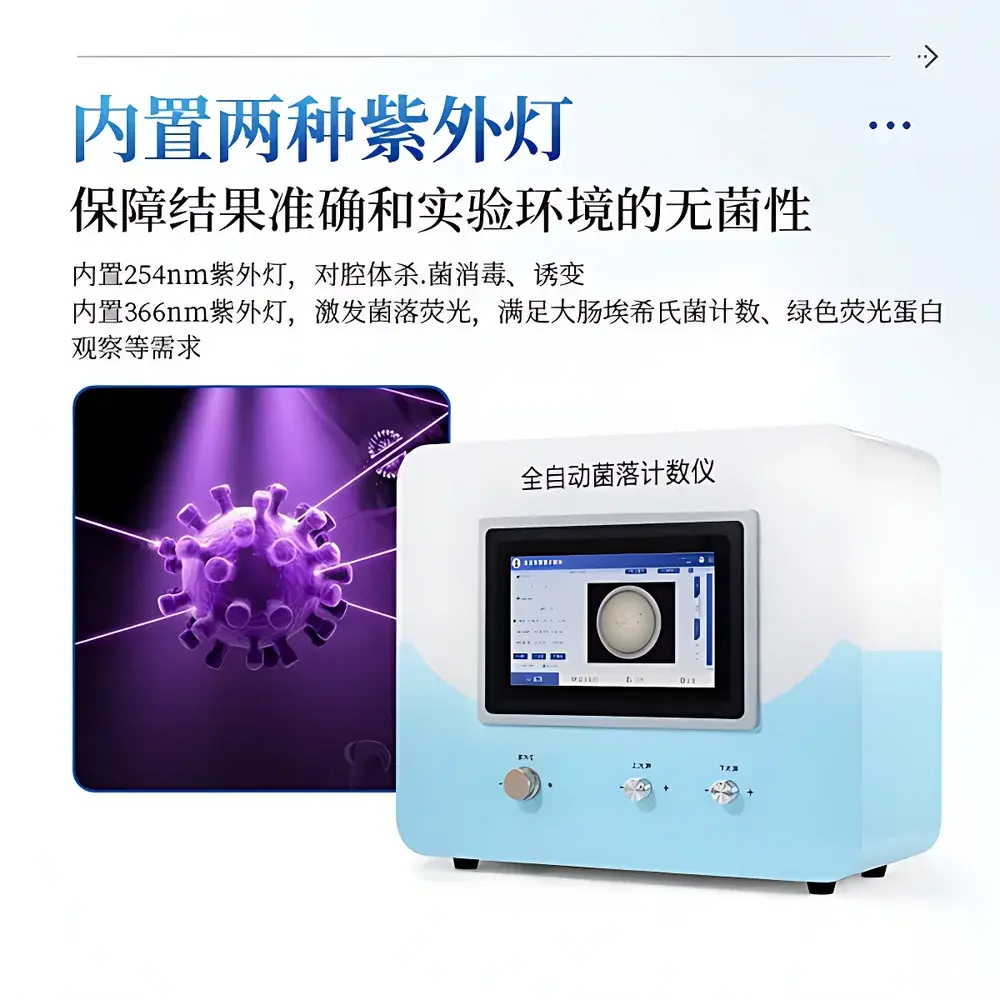
| Key Features | Dual UV Illumination (254 nm & 366 nm), Adjustable Top/Bottom LED Lighting, Touchscreen Interface (15.6″, Full HD), Onboard Windows 10 OS (64-bit, 256 GB SSD), Real-time Image Correction Tools, Multi-Mode Auto-Count Algorithms, Audit-Trail Enabled Data Management |
Overview
The COMECAUSE IN*J200 Automated Colony Counter is an engineered solution for objective, reproducible microbial enumeration in regulated food safety and quality control laboratories. It operates on the principle of high-resolution digital image acquisition combined with adaptive computer vision algorithms—specifically optimized for morphological segmentation, contrast-based thresholding, and spatial clustering analysis of microbial colonies grown on solid agar media. Unlike manual counting or semi-automated systems reliant on fixed illumination or single-mode detection, the IN*J200 integrates a fully enclosed dark chamber with independently controllable top and bottom lighting, dual-wavelength UV excitation (254 nm for surface sterilization and mutagenesis; 366 nm for fluorescence-based colony differentiation), and real-time pixel-level image processing. This architecture ensures consistent capture conditions across operators, shifts, and instruments—addressing a core limitation of subjective visual enumeration defined in ISO 4833-1:2013 and FDA Bacteriological Analytical Manual (BAM) Chapter 3. Designed for compliance-critical environments, it supports traceable, auditable workflows required under ISO/IEC 17025:2017, GMP, and HACCP-aligned quality systems.
Key Features
- Fully integrated imaging station with 16-megapixel color CMOS sensor and precision optical path—capable of resolving colonies as small as 0.1 mm in diameter under optimal contrast conditions.
- Dual UV illumination system: 254 nm lamp for internal chamber decontamination between runs; 366 nm lamp for selective excitation of fluorescent colonies (e.g., E. coli expressing GFP or chromogenic substrates).
- Adjustable dual-direction LED lighting: Independent rotary controls for top and bottom light intensity, enabling dynamic optimization for translucent, opaque, or pigmented media (e.g., VRBA, PCA, TSA, ChromAgar).
- On-device computing platform: 15.6-inch capacitive touchscreen (1920 × 1080), Windows 10 Pro 64-bit OS, 256 GB SSD—eliminates dependency on external PCs and reduces peripheral failure points.
- Multi-algorithm auto-count engine: Eight dedicated counting modes—including Dispersed, Aggregated, Micro-colony (0.1–0.5 mm), 3M PetriFilm™, Compact Dry™, RIDA™ membrane filters, Background-Matched, and Multi-color Segmentation—each configurable via intuitive UI sliders and preview overlays.
- Post-acquisition correction suite: Interactive tools including circular/rectangular ROI selection, freehand drawing, adjustable binarization thresholds, and 25-tier size-classified output tables with contour-based area metrics.
Sample Compatibility & Compliance
The IN*J200 accommodates standard Petri dishes (90–110 mm diameter), including polystyrene, polypropylene, and glass-bottom variants. Its optical design minimizes parallax and distortion across dish curvature, supporting both conventional pour plates and spread plates. The system complies with foundational microbiological standards including ISO 4833-1:2013 (horizontal method for colony count at 30 °C), ISO 6887-1:2017 (preparation of test samples), and AOAC Official Method 2008.01 (for PetriFilm™ applications). All image capture, analysis parameters, and user actions are timestamped and logged with immutable audit trails—meeting data integrity requirements per FDA 21 CFR Part 11 (electronic records/signatures) and EU Annex 11 (computerized systems). Raw images and processed datasets are stored in vendor-neutral TIFF/PNG formats with embedded EXIF metadata, ensuring long-term archival compatibility.
Software & Data Management
The onboard software implements role-based access control with multi-user login (administrator, analyst, reviewer), each maintaining isolated workspaces and encrypted data partitions. Every counting session generates a structured electronic report in PDF/A-1b format, containing: original high-res image, annotated count map, algorithm parameters (thresholds, minimum size, edge sensitivity), operator ID, timestamp, instrument serial number, and calibration status. All raw data remain uneditable post-generation; modifications require versioned annotation with justification. Reports support batch export to network drives or LIMS via SMB/CIFS protocol. Optional CSV export includes per-colony centroid coordinates, equivalent diameter, circularity index, and grayscale intensity—enabling downstream statistical analysis in R, Python, or JMP. Firmware updates are delivered via signed USB packages with SHA-256 verification.
Applications
The IN*J200 serves as a primary enumeration tool across food safety testing workflows: incoming raw material screening (e.g., milk powder, spices, produce wash water), in-process environmental monitoring (swabs from conveyor belts, packaging lines, cleanroom surfaces), and finished product release testing (ready-to-eat meals, dairy, beverages). It is validated for use with chromogenic and fluorogenic media targeting Salmonella, Listeria monocytogenes, Staphylococcus aureus, coliforms, and total viable counts. In research settings, its fluorescence mode supports GFP-tagged strain tracking in biofilm studies or probiotic stability assays. Regulatory labs utilize its audit-ready reporting for accreditation submissions (e.g., CNAS, UKAS) and proficiency testing participation (e.g., FAPAS, NORDVAL).
FAQ
Does the IN*J200 require external calibration or daily verification?
No. The system performs automatic geometric calibration at startup using factory-stored lens distortion profiles and working distance maps. Users may optionally run a quarterly verification with NIST-traceable calibration grids.
Can the software distinguish colonies from debris or agar scratches?
Yes—via configurable morphology filters (circularity, solidity, convexity) and multi-spectral contrast analysis. Debris rejection is enhanced in Fluorescence Mode when colonies emit characteristic emission spectra.
Is raw image data export supported for third-party reanalysis?
Yes. Unprocessed TIFF files (16-bit depth) with full EXIF metadata—including exposure time, gain, white balance, and lens profile—are accessible via file browser or automated export rules.
How does the system ensure compliance with ISO 17025 clause 7.7 (result reporting)?
Each PDF report embeds instrument identification, measurement uncertainty estimates (based on repeatability studies per ISO/IEC 17025:2017 Annex A.3), analyst signature field, and tamper-evident digital watermarking.
What maintenance is required beyond routine UV lamp replacement?
Annual optical alignment verification by certified service engineer; biannual cleaning of lens assembly and chamber interior using isopropyl alcohol and lint-free wipes—no consumables or proprietary reagents required.