COMECAUSE IN&LeafClear Modular Chlorophyll Fluorescence Imaging System
| Brand | COMECAUSE |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Model | IN&LeafClear |
| Camera Resolution | 1608 × 1104 pixels |
| Pixel Size | 9 µm × 9 µm |
| Frame Rate | 100 fps |
| Bit Depth | 12-bit |
| Interface | USB 3.0 |
| Lens Focal Length | 12 mm |
| Max Aperture | F/2.8 (adjustable) |
| HFOV | 62.11° |
| VFOV | 44.83° |
| Max Imaging Area | 50 cm × 35 cm |
| Excitation Wavelengths | 450 nm (blue), 630 nm (red), 730 nm (far-red) |
| LED Irradiance Range | 1–100% (up to 1440 µmol·m⁻²·s⁻¹) |
| OJIP Measurement Duration | 0.1–1.0 s (default 1 s) |
| PAM Dark Adaptation | 0–3600 s |
| PAM Light Cycles | 1–100 |
| Sampling Interval | 0.1–10.0 s |
| Temporal Resolution | 10 ms (OJIP), 100 ms (PAM) |
| Fluorescence Dynamic Range | 0–4095 (12-bit), SNR > 100:1 |
| Repeatability | CV < 3% |
| Linearity | R² > 0.999 |
| Spatial Resolution | ~0.3 mm/pixel at 50 cm × 35 cm FOV |
| Parameter Accuracy | Fv/Fm ±0.005, ΦPSII ±0.01, qP/qN ±0.02, NPQ ±0.1, PIABS ±0.05 |
Overview
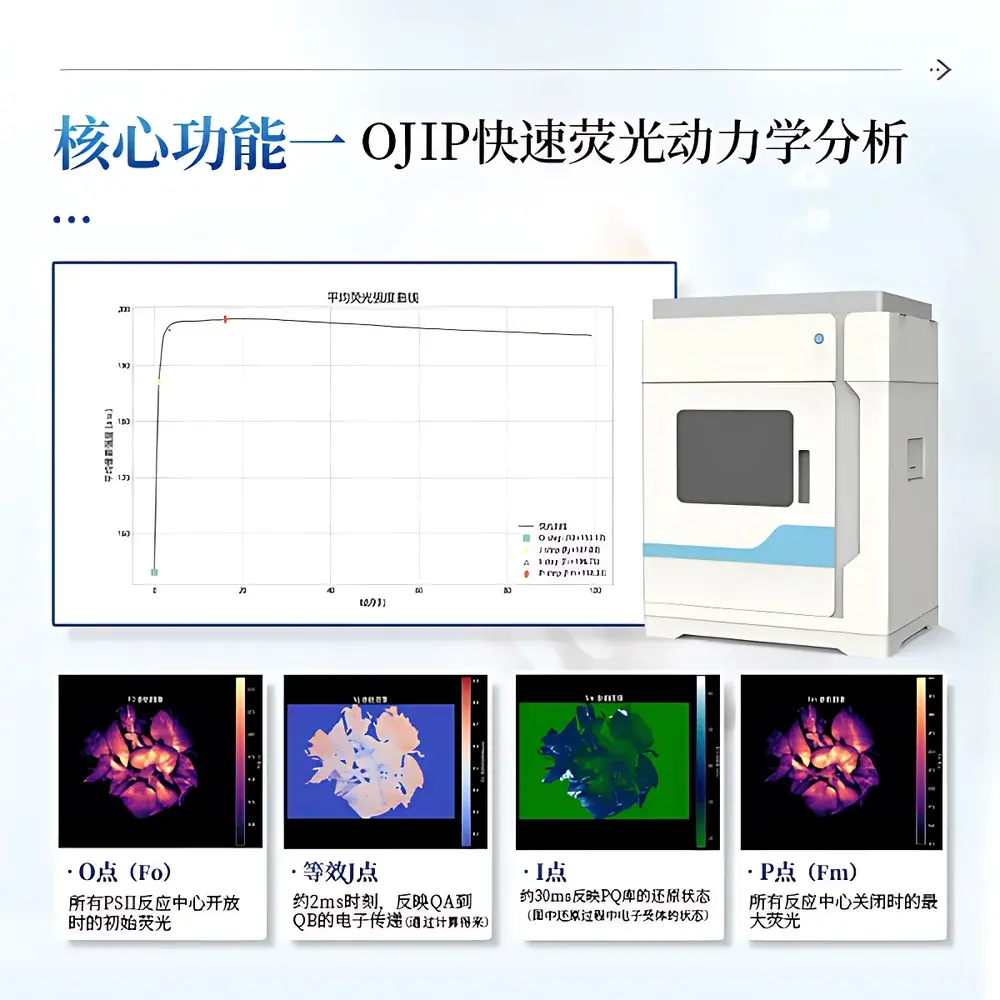
The COMECAUSE IN&LeafClear Modular Chlorophyll Fluorescence Imaging System is a research-grade, non-invasive optical platform engineered for quantitative spatial mapping of chlorophyll a fluorescence kinetics in intact plant leaves. It operates on the biophysical principles of photosystem II (PSII) photochemistry—specifically, the light-induced redox state transitions of the primary quinone acceptor QA—and captures fluorescence emission dynamics with high temporal and spatial fidelity. The system integrates two complementary measurement paradigms: (1) OJIP transient analysis, which records the millisecond-scale rise of chlorophyll fluorescence following a saturating pulse of blue light (450 nm), and (2) Pulse-Amplitude-Modulated (PAM) fluorometry, which uses modulated measuring light and saturating pulses under actinic illumination (630 nm) to resolve photochemical and non-photochemical quenching components. Designed for reproducible, high-throughput physiological phenotyping, the IN&LeafClear enables pixel-wise calculation of over 40 biologically validated parameters—including Fv/Fm, ΦPSII, qP, NPQ, PIABS, and JIP-test-derived energy flux ratios—without tissue disruption or chemical labeling.
Key Features
- Modular architecture supporting hardware expansion (e.g., additional spectral LEDs, environmental chambers, or gas exchange modules) via standardized electrical and mechanical interfaces.
- Dual-mode acquisition engine: synchronized OJIP transient capture (10 ms minimum sampling interval) and PAM time-series monitoring (100 ms minimum sampling interval), both fully configurable via software-defined protocols.
- High-sensitivity CMOS imaging sensor (1608 × 1104, 12-bit, 100 fps) with low-noise readout and >100:1 signal-to-noise ratio, optimized for weak fluorescence detection under controlled ambient light conditions.
- Triple-wavelength LED illumination system: 450 nm (blue, for PSII excitation and saturating pulses), 630 nm (red, for actinic light in PAM mode), and 730 nm (far-red, for PSI-driven QA reoxidation during Fo′ determination).
- Optical design featuring a fixed 12 mm lens (F/2.8 adjustable aperture) delivering uniform irradiance across a 50 cm × 35 cm field of view at standard working distance, with calibrated spatial resolution of ~0.3 mm per pixel.
- Graphical user interface with real-time preview, dual-language support (English/Chinese), parameter tooltips aligned with IUPAC and Plant Physiology Society nomenclature, and GLP-compliant audit trail logging for all instrument settings and acquisition events.
Sample Compatibility & Compliance
The IN&LeafClear accommodates intact, unmodified leaves from angiosperms, gymnosperms, ferns, and bryophytes—ranging from small model species (e.g., Arabidopsis thaliana) to large agricultural crops (e.g., maize, wheat, soybean). Sample placement requires no mounting media or vacuum fixation; measurements are performed under ambient atmospheric conditions or within optional climate-controlled enclosures. The system complies with ISO 14040/14044 for environmental impact assessment workflows and supports experimental designs aligned with ASTM E2912-22 (Standard Guide for Chlorophyll Fluorescence Measurements in Plants). All fluorescence parameters adhere to the consensus definitions established by the International Workshop on Chlorophyll Fluorescence Kinetics (2011) and the Photosynthesis Research journal’s reporting standards. Data export formats (CSV, PNG) meet FAIR principles (Findable, Accessible, Interoperable, Reusable), and configuration files retain full metadata traceability for regulatory audits.
Software & Data Management
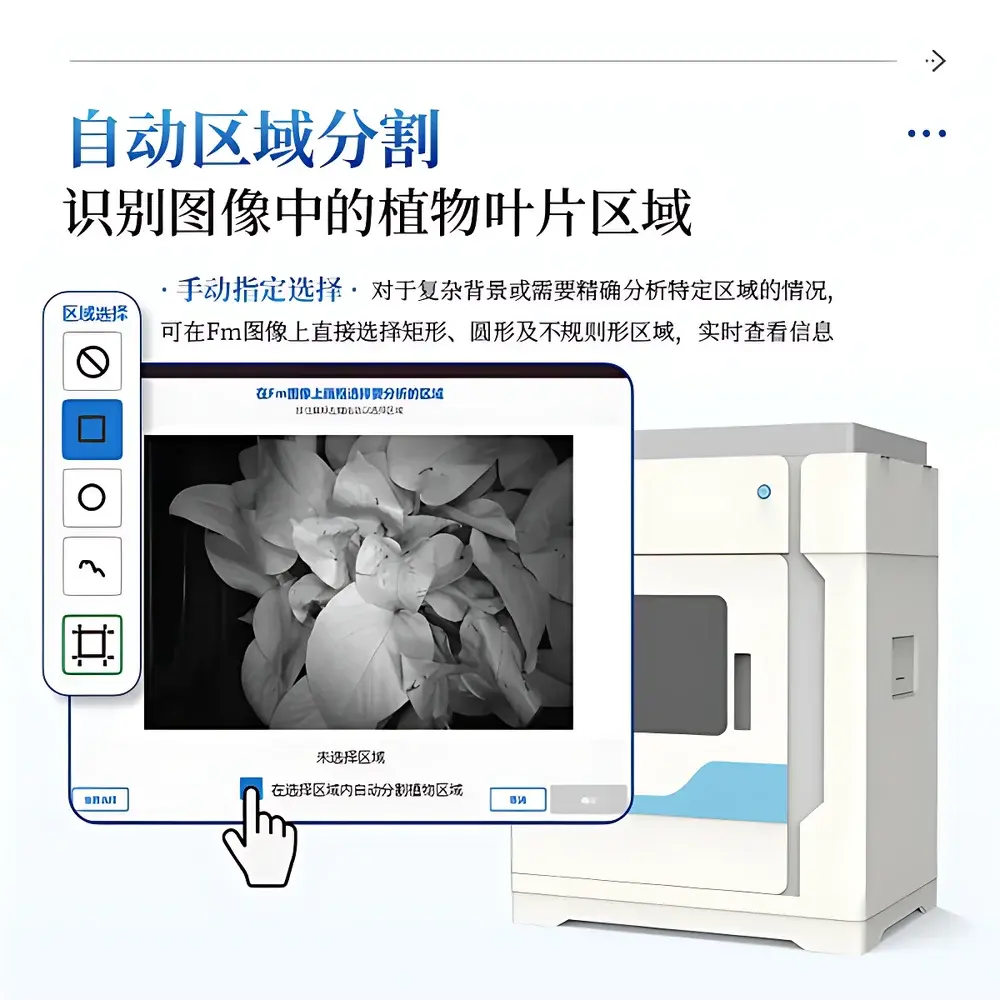
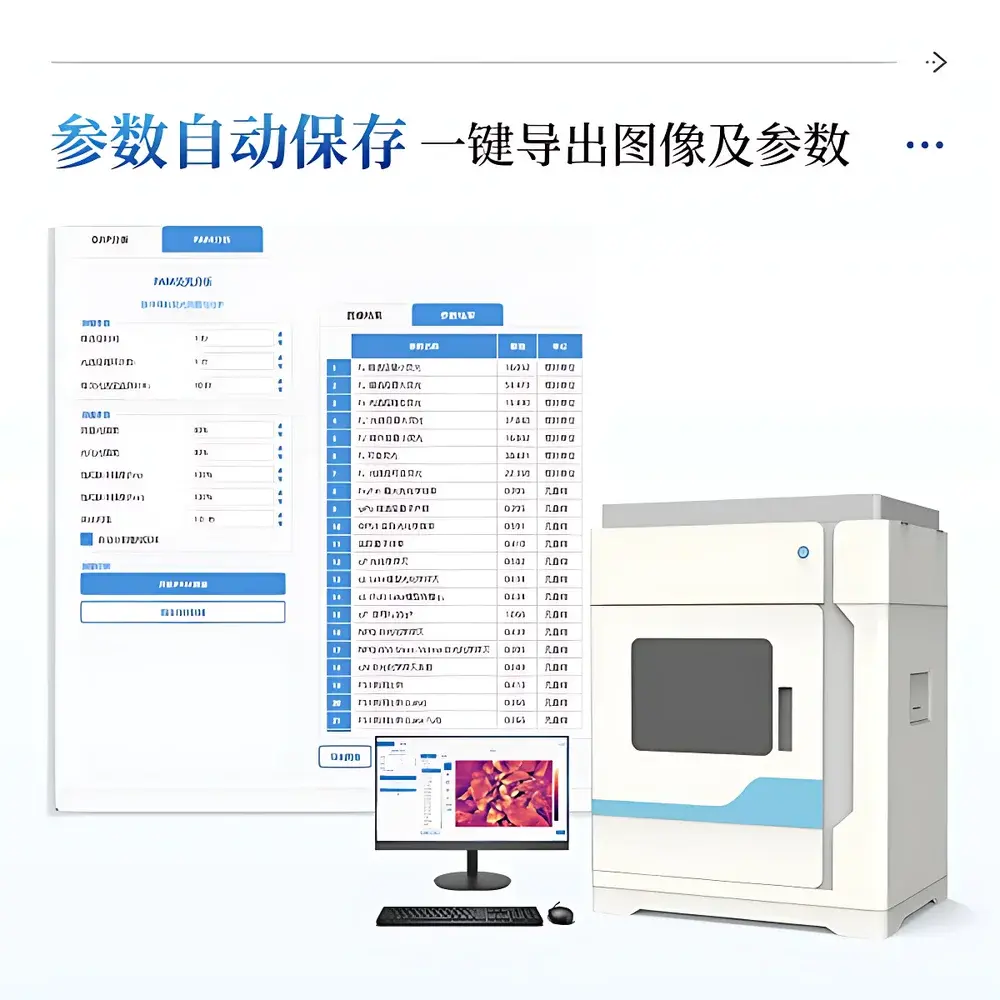
The proprietary IN&LeafClear Control Suite provides end-to-end data handling—from acquisition protocol definition to publication-ready visualization. Measurement parameters (e.g., dark adaptation duration, actinic intensity, saturation pulse amplitude, sampling frequency) are stored in human-readable JSON configuration files with automatic versioning and timestamped backups. Image processing includes adaptive Gaussian noise reduction, region-of-interest (ROI) segmentation using Otsu thresholding or manual polygon selection, and per-pixel parameter computation using validated algorithms derived from the JIP-test formalism and Kramer et al.’s PAM theory. Fluorescence images are rendered using perceptually uniform colormaps (e.g., viridis, plasma) with embedded colorbars showing absolute numerical ranges. All results—including kinetic curves (OJIP, relative variable fluorescence), time-series plots (ΦPSII, NPQ), and tabular outputs—are exportable in CSV (for statistical analysis in R or Python) or Excel (for GLP/GMP documentation). The software enforces FDA 21 CFR Part 11 compliance through electronic signatures, role-based access control, and immutable audit logs tracking every parameter change, image acquisition, and export action.
Applications
The IN&LeafClear serves as a core analytical tool in plant stress physiology, crop science, and ecosystem monitoring. In controlled-environment studies, it quantifies sub-lethal drought responses via early declines in Fv/Fm (<5% reduction preceding visible wilting) and altered PIABS kinetics reflecting impaired electron transport capacity. Under thermal stress assays, it discriminates thermotolerant genotypes by sustained ΦPSII (>0.5) and regulated NPQ induction (2–3 range) during prolonged heat exposure. In agronomic trials, it evaluates nitrogen-use efficiency through correlations between Fv/Fm recovery rates post-fertilization and grain yield proxies. Field-deployable variants (with portable power and weatherproof housing) enable canopy-level fluorescence mapping for ecological assessments—such as monitoring photoinhibitory effects of ozone pollution or tracking seasonal shifts in photosynthetic vigor across forest understories. Its capacity for longitudinal, non-destructive monitoring also supports early disease detection: pathogen-induced reductions in Fo heterogeneity and J-I-P phase distortions precede symptom onset by 36–48 hours in fungal infection models.
FAQ
What is the minimum detectable change in Fv/Fm that the IN&LeafClear can reliably resolve?
The system achieves ±0.005 absolute accuracy in Fv/Fm, enabling statistically robust discrimination of ≤1% physiological differences across biological replicates under controlled conditions.
Can the system operate under ambient laboratory lighting?
Yes—its narrow-band excitation LEDs (450/630/730 nm) and synchronized gated acquisition eliminate interference from broad-spectrum room lighting; however, direct sunlight must be excluded during measurement.
Is remote operation supported for long-term experiments?
The software includes TCP/IP-based network control, scheduled acquisition triggers, and automated data archiving to network-attached storage (NAS), facilitating unattended overnight or multi-day monitoring.
How does the system ensure calibration stability across extended use?
Each LED channel incorporates factory-calibrated irradiance sensors; users may perform routine verification using NIST-traceable reference standards (e.g., Spectralon® reflectance tiles) and validate camera linearity via neutral density filter series.
Are third-party data analysis scripts compatible with exported datasets?
All CSV exports follow MIAME-compliant column headers and include metadata headers (instrument ID, timestamp, protocol name, ROI coordinates); MATLAB, Python (NumPy/Pandas), and R packages provide native loaders for batch processing and machine learning pipelines.