COMECAUSE IN-LeafClear Non-Destructive Chlorophyll Fluorescence Imaging System
| Brand | COMECAUSE |
|---|---|
| Model | IN-LeafClear |
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Camera Resolution | 1608 × 1104 |
| Pixel Size | 9 µm × 9 µm |
| Frame Rate | 100 fps |
| Bit Depth | 12-bit |
| Interface | USB 3.0 |
| Lens Focal Length | 12 mm |
| Max Aperture | F/2.8 (adjustable) |
| HFOV | 62.11° |
| VFOV | 44.83° |
| Max Imaging Area | 50 cm × 35 cm |
| Excitation Wavelengths | 450 nm (blue), 630 nm (red), 730 nm (far-red) |
| LED Irradiance Range | 90–1440 µmol/(m²·s) |
| OJIP Duration | 0.1–1.0 s (default 1 s) |
| PAM Dark Adaptation | 0–3600 s |
| PAM Saturation Pulse Intensity | 15–100% (450 nm) |
| Fluorescence Dynamic Range | 0–4095 (12-bit) |
| SNR | >100:1 |
| Repeatability (CV) | <3% |
| Fv/Fm Accuracy | ±0.005 |
| ΦPSII Accuracy | ±0.01 |
| Spatial Resolution | ~0.3 mm/pixel (at 50 cm × 35 cm FOV) |
| Temporal Resolution | 10 ms (OJIP), 100 ms (PAM) |
Overview
The COMECAUSE IN-LeafClear is a research-grade, non-destructive chlorophyll fluorescence imaging system engineered for quantitative, spatially resolved analysis of photosynthetic performance in intact plant leaves. It operates on the biophysical principles of chlorophyll a fluorescence induction kinetics—specifically leveraging both the OJIP transient (rapid fluorescence rise after dark adaptation) and pulse-amplitude modulation (PAM) fluorometry. These complementary modalities enable simultaneous assessment of Photosystem II (PSII) structural integrity, electron transport efficiency, photoprotective capacity, and dynamic acclimation responses under controlled or field-simulated light regimes. Unlike point-measurement chlorophyll meters, the IN-LeafClear captures full-field 2D fluorescence maps across defined imaging areas (up to 50 cm × 35 cm), preserving spatial heterogeneity critical for detecting localized stress responses, variegation effects, or treatment gradients. Its design adheres to established biophysical conventions outlined in the JIP-test framework (Strasser et al., 2004) and conforms to standardized PAM protocols referenced in ISO 14244-1:2021 (Plant Physiology — Fluorometric Methods) and ASTM E3292-22 (Standard Guide for Chlorophyll Fluorescence-Based Stress Assessment in Higher Plants).
Key Features
- High-resolution CMOS imaging sensor (1608 × 1104 pixels, 12-bit depth, 100 fps frame rate) with low-noise architecture and >100:1 signal-to-noise ratio for reliable detection of sub-nanomolar chlorophyll fluorescence signals.
- Dual-mode optical excitation system: independently controllable 450 nm blue LEDs (for OJIP induction and saturation pulses), 630 nm red LEDs (actinic light for PAM photoactivation), and 730 nm far-red LEDs (for PSI-driven QA reoxidation during Fo′ determination).
- Precisely calibrated irradiance control (90–1440 µmol/(m²·s), adjustable in 5% increments) with microsecond-level LED response time (<1 ms), ensuring temporal fidelity in kinetic measurements.
- Optimized optical train featuring a fixed 12 mm focal length lens (F/2.8 variable aperture), delivering uniform illumination and high MTF across a 62.11° horizontal × 44.83° vertical field of view.
- Modular hardware architecture with USB 3.0 interface, real-time device status monitoring, and automatic peripheral enumeration (camera + LED controller) at system boot.
- Graphical user interface supporting seamless bilingual operation (English/Chinese), parameter tooltips aligned with IUPAC-recommended nomenclature, and persistent configuration storage per experimental protocol.
Sample Compatibility & Compliance
The IN-LeafClear accommodates intact, unmodified leaves from herbaceous and woody species—including Arabidopsis, maize, wheat, rice, tobacco, and perennial shrubs—without requiring sectioning, staining, or vacuum infiltration. Samples are positioned at a standardized working distance (optimized for 50 cm × 35 cm field coverage), eliminating parallax-induced quantification errors. The system supports compliance workflows required in regulated environments: raw fluorescence time-series data include embedded metadata (timestamp, LED intensity, exposure duration, saturation pulse parameters), enabling traceability per GLP Annex 11 and FDA 21 CFR Part 11 requirements when paired with validated laboratory information management systems (LIMS). All derived parameters—including Fv/Fm, ΦPSII, NPQ, qP, PIABS, and JIP-test indices—are computed using open, peer-reviewed algorithms consistent with the Biophysical Journal’s standardized reporting guidelines for fluorescence kinetics.
Software & Data Management
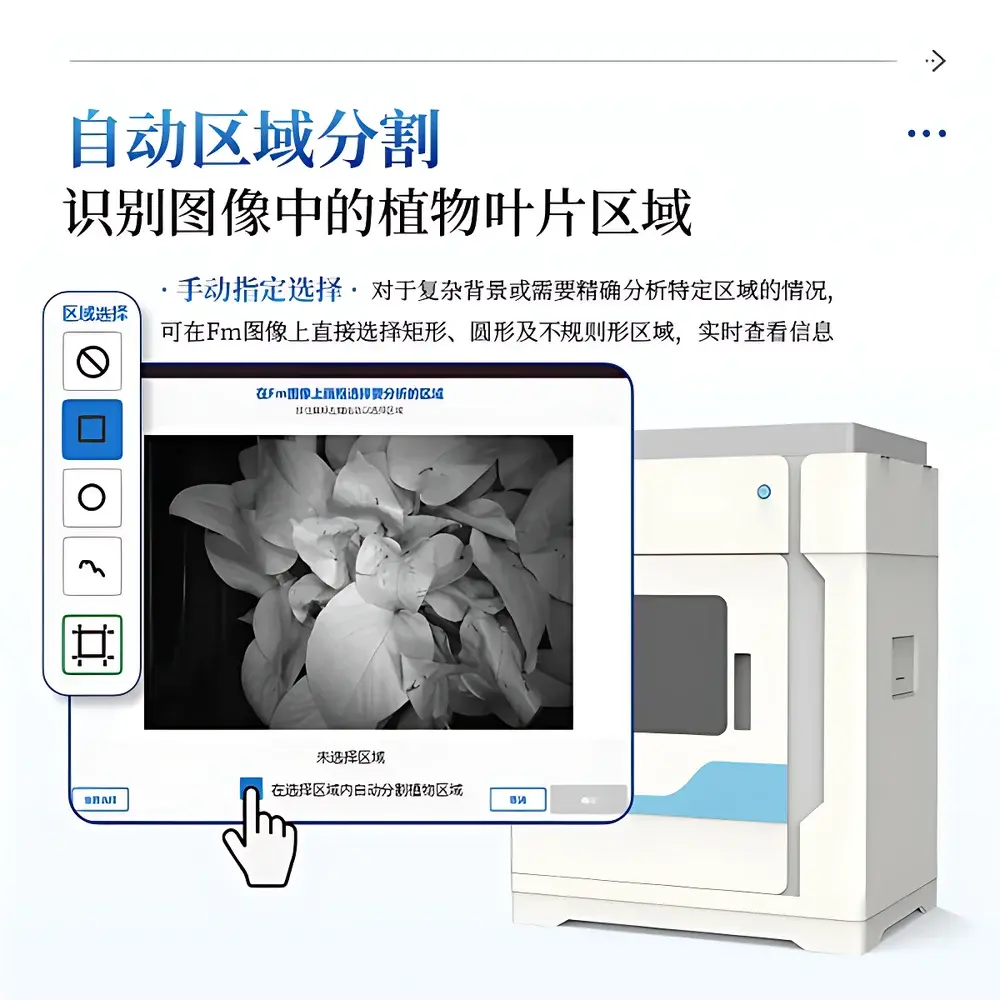
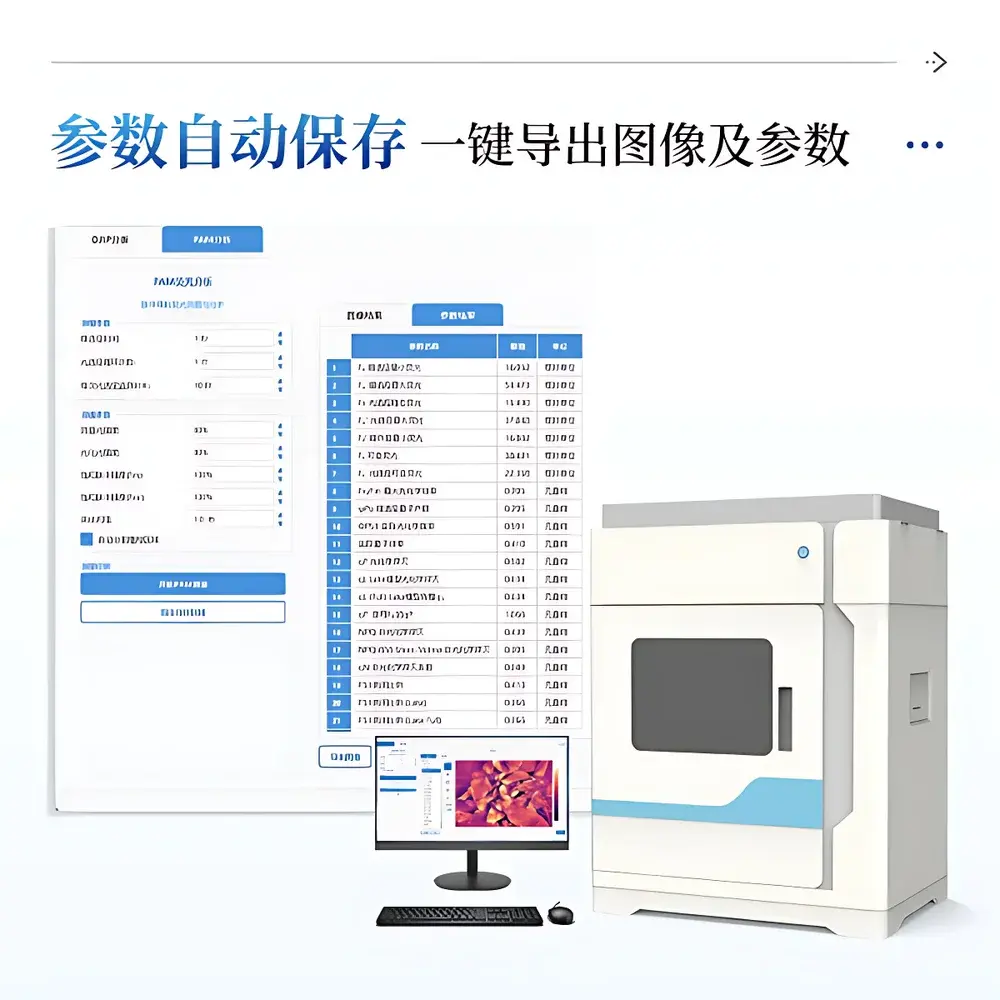
The integrated acquisition and analysis suite provides native support for both OJIP and PAM experimental paradigms. It implements automated region-of-interest (ROI) segmentation via adaptive thresholding (Otsu method), with manual override options for polygonal, rectangular, or circular selections. Each ROI generates spatially averaged parameter values updated in real time. Image processing includes Gaussian noise reduction, contrast-limited adaptive histogram equalization (CLAHE), and scientifically validated pseudo-color mapping (Jet colormap with linear scaling and user-defined min/max bounds). All fluorescence transients are saved as time-stamped, lossless TIFF sequences; derived parameter maps are exportable as PNG (8-/16-bit) with embedded color scale metadata. Numerical outputs—including full JIP-test parameter sets (e.g., Mo, Area, ΦPo, ψEo, PIABS) and PAM-derived metrics (e.g., ETR, Rfd, Y(NO), Y(NPQ))—are exported to CSV or XLSX formats with SI-compliant units and IUPAC-standardized symbols. File naming follows the schema “Mode_SeqNum_ParameterName”, ensuring unambiguous audit trail reconstruction.
Applications
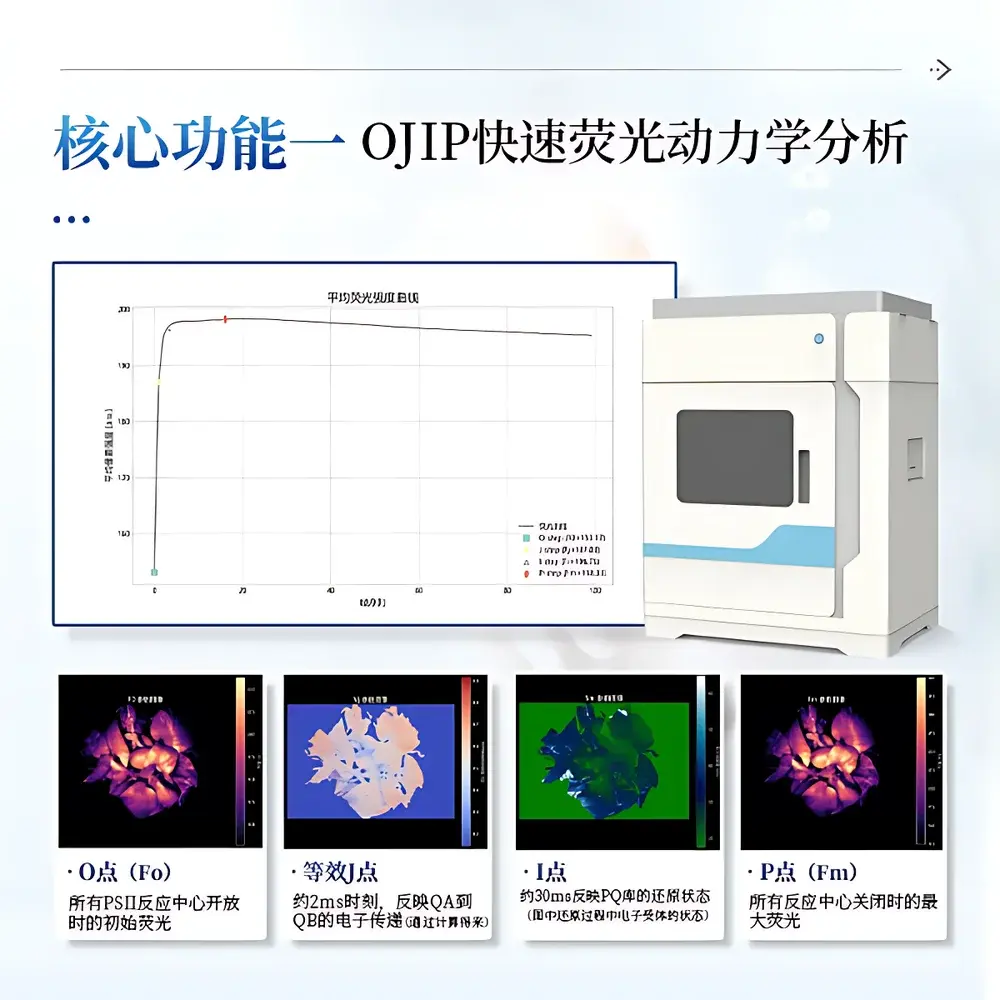
The IN-LeafClear serves as a primary tool in fundamental and applied plant science. In photosynthesis research, it enables high-throughput screening of PSII reaction center density, QA-to-QB electron transfer kinetics (via J-step analysis), and plastoquinone pool redox state (I-step quantification). For environmental stress physiology, it detects early-stage drought-induced declines in Fv/Fm (>5% reduction precedes visible wilting), quantifies thermal damage thresholds via ΦPSII collapse kinetics, and resolves heavy metal–induced NPQ dysregulation patterns. In agronomy, it validates cultivar-specific photoprotection strategies under elevated irradiance, evaluates nitrogen-use efficiency through ABS/RC and TRo/RC stoichiometry, and identifies pesticide phytotoxicity signatures prior to symptom onset. Ecological applications include canopy-level fluorescence heterogeneity mapping in mixed-species stands and longitudinal monitoring of climate change–driven shifts in quantum yield resilience across phenological stages.
FAQ
What is the minimum detectable change in Fv/Fm with this system?
The system achieves ±0.005 absolute accuracy in Fv/Fm, enabling statistically robust detection of ≤2% relative changes under controlled conditions (n ≥ 5 biological replicates, CV < 3%).
Can the system perform simultaneous OJIP and PAM measurements on the same leaf?
No—OJIP and PAM are mutually exclusive acquisition modes due to divergent illumination protocols and kinetic sampling requirements; however, sequential measurements on the same leaf are fully supported with automated dark-adaptation timing.
Is remote operation or scripting support available?
Yes—the software exposes a documented COM/ActiveX API for integration with MATLAB, Python (via pywin32), or LabVIEW, allowing programmable experiment sequencing and external trigger synchronization.
Does the system comply with international standards for fluorescence-based plant phenotyping?
Yes—it implements measurement definitions and calculation routines aligned with ISO 14244-1:2021, the Plant Phenomics Journal’s Minimum Information About a Plant Phenotyping Experiment (MIAPPE) v1.1, and the Fluorescence Kinetics Consensus Guidelines published by the International Society of Photosynthesis Research (ISPR).
What calibration procedures are recommended before daily use?
A blank reference image (no sample, all LEDs off) and dark current frame (all LEDs off, same exposure) should be acquired daily; optional white-reference correction using a Spectralon® panel is supported for absolute radiometric normalization.